 Case Report
Case Report
Death from Acute Hyponatremia Following Uneventful Abdominal Hysterectomy: Lessons from a Case Report
George A Vilos*, Angelos G Vilos and Basim Abu Rafea
London Health Sciences Centre, Canada
George A Vilos, Professor, Obstetrics and Gynecology, London Health Sciences Centre, Canada.
Received Date: May 31, 2019; Published Date: June 10, 2019
Abstract
A 38-year-old woman underwent a routine/uneventful abdominal hysterectomy for abnormal uterine bleeding (AUB) and uterine fibroids. Approximately 24 hours later, she developed severe hyponatremia (Na = 118 mEq/L), brain edema and coma, and she died 3 days later. The mechanism of hyponatremia was, likely, due to excessive and/or inappropriate fluid infusion of 2/3:1/3 solution. The inability to regulate her fluids and electrolytes (Na and K), was, likely, related to addback estrogen therapy given for 3 months preoperatively in conjunction with a GnRH agonist to optimize the patient’s hemoglobin and shrink the uterus/fibroids. It has been known that postmenopausal women and men can regulate excessive hyponatremic fluid absorption and electrolytes better than premenopausal women implicating an estrogen role. Estrogen has been found to inhibit the Na/K-ATPase pump in a variety of tissues/organs including the brain. This woman’s pathology of the endometrium was reported as proliferative indicating that enough estrogen may have been available to inhibit her Na/K-ATPase pump resulting in inability to regulate her fluids and electrolytes leading to her death.
Keywords: Hysterectomy; Hyponatremia; Death; GnRH agonist; Addback therapy
Introduction
Hysterectomy is the most frequently performed gynecologic procedure worldwide with a lifetime risk for hysterectomy in the US of 45% [1]. Uterine fibroids, abnormal uterine bleeding (AUB) and chronic pelvic pain with or without endometriosis are the three major indications for hysterectomy and together, they account for 55% - 85% of all hysterectomies [2].
However, in spite of its common practice and advancements in techniques and technologies, hysterectomy is still associated with considerable risks and complications resulting in significant morbidity (15%-30%) and mortality (0.03%-0.04%) [3]. In 2018, a population-based, retrospective cohort study of 93 059 adult women who underwent abdominal hysterectomy from April 1, 2003, to October 1, 2014, in Ontario, Canada, reported a 30-day mortality of 59 (0.06%) [4].
Although the cause of death was not specified in any of the above studies, the most frequent associated morbidities listed were wound complications, venous thromboembolism (VTE), urinary tract infection (UTI), sepsis and blood transfusion. Herein we present a unique case of uneventful abdominal hysterectomy for AUB and uterine fibroins who died from acute dilutional hyponatremia.
Case
Pre-operative care & informed consent
A 38-year-old woman, P1G1, BMI 27kg/m2 presented with abnormal uterine bleeding (AUB), severe dysmenorrhea and known uterine fibroids. On examination, the uterus measured up to her umbilicus (20 weeks size). Ultrasound measured the uterus at 15 x 12 x 8cm with multiple fibroids. Fertility was not desired, and she was using condoms for birth control. She had no significant current medical and no past surgical issues.
Risks and benefits of treatment options including uterine artery embolization (UAE) and total abdominal hysterectomy (TAH) with or without bilateral salpingo-oophorectomy (BSO) were discussed. The patient opted for TAH +/- BSO and risks including injury to ureters, bladder and bowel were discussed.
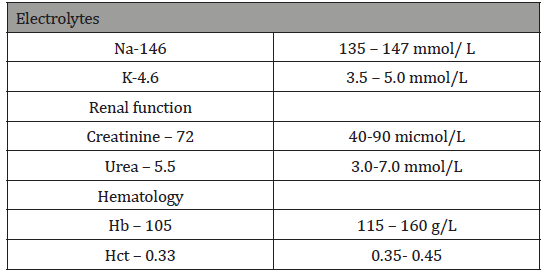
Pre-operatively, to optimize the patient’s hemoglobin and shrink the uterus and fibroids for a possible ‘bikini’ incision, the patient was prescribed leuprolide acetate (3.75mg x 3 months; Abbvie, Saint-Laurent, Quebec, Canada) and Estradot patch (Novartis pharmaceuticals, Dorval, Quebec, Canada) for addback therapy to minimize the hypoestrogenic effects of the leuprolide acetate. The patient’s immediate preoperative hematology indices are shown in Table 1.
Table 1: Immediate preoperative hematology indices.
Conduct of surgery
The surgery (TAH + BSO) was completed uneventfully in 1 hour and 36 minutes, through a Pfannenstiel incision and the estimated blood loss was 100mL.
The postoperative orders included: IV [intravenous] 2/3:1/3 [solution] - 150 mL/hour; PCA [Patient Controlled Analgesia]: Morphine x 48 hours. Dose 1-2mg, lockout 5 minutes, 4-hour limit 30mg; Tylenol # 3, as required.
Post-operative care
On POD #1, at 06:00 hours, over a period of 18 hours, the Foley catheter bag contained 450mL of urine (approximate rate of 25-30 mL/hour), and a bolus of 750mL of 2/3:1/3 solution was ordered and given.
At 13:00 hours, the visual analogue scale score (VAS) was 0-2 and the Patient Controlled Analgesia (PCA) pump was discontinued after 45 mg of total use of morphine. According to her visiting family, in the late afternoon/early evening, the patient seemed drowsy and at 21:05 hrs, she had a cardiac arrest. Following cardiopulmonary resuscitation (CPR), she was transferred to the intensive care unit (ICU) where she never regained consciousness and at noon of POD #4, she was declared dead and was taken off all life support systems. The hematology indices in the ICU are listed in Table 2&3.
Table 1: Immediate postoperative orders.

Table 2: Hematology indices throughout the patient’s stay in the hospital.

According to the Coroner’s Report, there were no significant findings and the cause of death was reported as severe brain edema due to hyponatremia. The pathology of the uterus was reported as: specimen weighing 330g; - Endometrium: proliferative endometrium; - Myometrium: leiomyomas; - Ovaries/tubes: no pathologic diagnosis.
Discussion
This case raises several important issues and questions and provides several learning opportunities including the following.
Potential causes of hyponatremia
Inappropriate Antidiuretic Hormone (ADH) release: A nephrologist in his Consultation Note stated: “With respect to her hyponatremia, given the acute neurological change, sodium of 119 and cerebral edema, she likely had ADH release postoperatively secondary to pain plus/minus hypovolemia”. However, according to the record, at 13:00 hours of POD #1, the VAS was 0-2 and the PCA was discontinued after 45 mg of total use of morphine. This indicates that the pain had been very well controlled and postoperative pain was never an issue.
Dilutional hyponatremia: The cause of hyponatremia was most likely due to infusion of excessive and/or inappropriate fluid. According to the anesthesia record, the patient had at least 1L of Ringers lactate solution pre-/intra-operatively. In addition, the patient received 750mL bolus (2/3:1/3) in the morning of POD #1, and possibly 150mL/hour x 30 hours (4500 mL 2/3:1/3 –hyponatremic solution), in addition to drinking fluids at liberty. Therefore, the patient received approximately 5000mL of hyponatremic fluid, 2/3 of which (1.5-2 L) was free water. Therefore, of 5000mL of 2/3:1/3 solution, 1/3 (1700 mL) was free water.
Studies from fluid absorption during hysteroscopic/ resectoscopic surgery have indicated that for every 100 mL of free water absorbed, the serum Na decreases by 1 mmol/L. [5] upon review, it appears that this woman received over 2000 mL of free water which could potentially account for a decrease in serum Na in the low 120s.
Why did this woman die?
In 1986, Arieff reported on 15 healthy women, aged 22 to 66- year, with severe hyponatremia following uncomplicated elective surgery including 5 TAH, 1 D&C and 11 general surgical procedures. Postoperatively, all patients presented with seizures and coma within 72 hours and 4 died without regaining consciousness while the rest sustained various degrees of significant permanent brain impairment. Although the mechanism of this syndrome, at that time, could not be determined, it was hypothesized that the Na/KATPase system in the brain is less efficient at extruding K in women than in Men; likely being inhibited by some female sex hormones such as progesterone and/or its derivatives [6].
In a subsequent review of 4 publications, Ayus et al., found that 36 of 38 patients with postoperative hyponatremic encephalopathy were women. All but one woman either died or experienced permanent brain damage, and both men recovered without experiencing neurologic sequelae. In addition, the authors sought to determine potential factors associated with the development of encephalopathy and with its clinical course in patients with postoperative hyponatremia. The authors concluded that women and men are equally likely to develop hyponatremia and hyponatremic encephalopathy after surgery. However, when hyponatremic encephalopathy develops, menstruant women are about 25 times more likely to die or have permanent brain damage compared with either men or postmenopausal women [7].
The available evidence indicates that postmenopausal women and men (TURP syndrome) tolerate/overcome hyponatremia better than women [7,8].
Role of GnRH agonists and estrogen: In 1998, Taskin et al., reported on 17 women undergoing radiofrequency hysteroscopic endometrial ablation randomized to 9 receiving a GnRH agonist versus 8 receiving saline. The authors found that the Na/K-ATPase pump activity was significantly increased in the GnRH-a group compared with the saline group and correlated with decreased estradiol levels (0.4+/- 0.08 vs 0.26+/-0.06 micromol/min/ml) [9].
Results
Endometrial pathology was detected in 69 out of 612 patients who underwent biopsy for accepted clinical indications, polyps in 36, simple hyperplasia in 25, complex hyperplasia in 13, and endometrial cancer in [5].
Comparison of endometrial thickness and morphology with biopsy results revealed the following: Simple hyperplasia has increased thickness in 9 out of 15 cases and abnormal appearance in 6 (non-uniform echogenicity, lack of 3-layer pattern); in cases of complex hyperplasia and cancer, endometrial thickness was abnormal in 15 out of 18 cases (83%) and morphology in all cases (heterogeneous, asymmetrical or cystic).
Conclusion
This patient received excessive hyponatremic fluid (2/3:1/3 solution) infusion which resulted in severe hyponatremia (Na<120). Although the patient received a GnRH agonist which is known to create a hypoestrogenic state and also reduces the adverse effects of the ATP-ase pump which may be protective against iatrogenic hyponatremia, the addback therapy negated the protective effects of the GnRH-a. The Estradot patch contains 0.585mg estradiol- 17β and it delivers 37.5μg daily. This, likely, increased the serum estradiol level sufficiently to estrogenize the endometrium since the pathology report indicated a proliferative endometrium.
Acknowledgement
None.
Conflict of Interest
No conflict of interest.
References
- Merrill RM (2008) Hysterectomy surveillance in the United States-1997 through 2005. Med Sci Monit 14(1): CR24–CR31.
- Jacobson GJ, Shaber RE, Armstrong MA, Hung YY (2006) Hysterectomy rates for benign indications. Obstet Gynecol 107(6):1278-1283.
- Wiser A, Holcroft CA, Tulandi T, Abenhaim HA (2013) Abdominal versus laparoscopic hysterectomies for benign diseases: evaluation of morbidity and mortality among 465,798 cases. Gynecol Surg 10(2): 117- 122.
- Kelly EC, Winick-Ng J, McClure JA, Peart T, Chou Q, et al. (2019) Hysterectomy in Ontario: A Population-Based Study of Outcomes and Complications in Minimally Invasive Compared With Abdominal Approaches. J Obstet Gynaecol Can: S1701-S2163.
- Istre O, Skajaa K, Schjoencby A, Forman A (1992) Changes in serum electrolytes after transcervical resection of endometrium and submmucous fibroids with glycine 1.5% for uterine irrigation. Obstet Gynecol 80(2): 218-222.
- Arieff AI (1986) Hyponatremia, convulsions, respiratory arrest, and permanent brain damage after elective surgery in healthy women. N Engl J Med 314(24): 1529-1535.
- Ayus JC, Wheeler JM, Arieff AI (1992) Postoperative hyponatremic encephalopathy in menstruant women. Ann Intern Med 117(11): 891- 897.
- Fraser CL, Kucharczyk J, Arieff AI, Rollin C, Sarnacki P, et al. (1989) Sex differences result in increased morbidity from hyponatremia in female rats. Am J Physiol 256(4 Pt 2): R880-R885.
- Taskin O, Buhur A, Birincioglu M (1998) Endometrial Na+, K+ -ATPase pump function and vasopressin levels during hysteroscopic surgery in patients pretreated with GnRH agonists. J Am Assoc Gynecol Laparosc 5: 119-124.
-
George A Vilos, Angelos G Vilos , Basim Abu Rafea. Death from Acute Hyponatremia Following Uneventful Abdominal Hysterectomy: Lessons from a Case Report. W J Gynecol Women’s Health. 2(4): 2019 WJGWH.MS.ID.000541.
Hysterectomy, Hyponatremia, Death, GnRH agonist, Addback therapy
-

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.






