Research Article
Research Article
Pressure-less Acoustic Immittance Measurements in Children with Otitis Media with Effusion
Munar Beishenova1, Shirin Zhumabaeva1, Blanda Kamin2, Moayad Anbari2, Dörte Fischer2,3, Shayirbek Sulaimanov1 and Tino Just2*
1 Department of Otorhinolaryngology, The National Center of Maternity and Childhood Care, Bishkek, Kyrgyzstan
2 Department of Otorhinolaryngology, Head and Neck Surgery, KMG Klinikum, Güstrow, Germany
2 Department of Audiology, KMG Klinikum, Güstrow, Germany
Dr. Munar Beishenova (ORCID ID: 0009-0000-8375-2563)
Dr. Shirin Zhumabaeva (ORCID ID: 0000-0002-9073-1127)
Blanda Kamin (ORCID ID: 0009-0006-6072-6630)
Moayad Anbari (ORCID ID: 0009-0003-5565-6437)
Dr. Dörte Fischer (ORCID ID: 0000-0003-3736-4441)
Professor Shayirbek Sulaimanov (ORCID ID: 0000-0002-0980-0501)
Professor Tino Just (ORCID ID: 0000-0003-2036-8569)
Tino Just, M.D., Department of Otorhinolaryngology, Head and Neck Surgery Friedrich-Trendelenburg-Allee 1, D-18273 Güstrow, Germany.
Received Date: June 02, 2025; Published Date: July 09, 2025
Abstract
Objective: This two-center study aims to determine whether pressure-less acoustic immittance (PLAI) measurements can be used to diagnose
otitis media with effusion (OME) in children.
Methods: A total of 112 children (224 ears), aged three months to 17 years, were recruited from two Departments of Otorhinolaryngology in
Kyrgyzstan and Germany. The mean age of the participants was 8.29 ± 0.45 years (65 boys and 47 girls). The middle ear pathology group included 63
patients: 22 with unilateral and 41 with bilateral conditions. Healthy children with normal bilateral ears (N = 49) were also included. Three different
age groups were considered: 0-2 years (Nears = 26), > 2–6 years (Nears = 64), and > 6-18 years (Nears = 134).
Ear, nose, and throat examinations were performed by experienced ENT surgeons in both departments. Only children with a non-inflamed
eardrum showing clear signs of effusion were categorized as having “OME.” All children underwent video otoscopy, PLAI measurements using
MedWave® (Neuranix, Italy), and conventional single-frequency tympanometry.
Results: Among the 120 normal-appearing ears assessed via otoscopy, the average resonance frequency (RF) of the admittance was 503.3 Hz
± 140.6 Hz (ranging from 285.3 to 1119 Hz), and the average peak admittance was 1.50*10-2 mmho (ranging from 0.49 to 3.75*10-2 mmho). In the
58 ears with OME, the average RF was 631.1 Hz ± 115.5 Hz (ranging from 247.6 to 958.2 Hz), and the average peak admittance was 1.68*10-2 ± 0.70
mmho (ranging from 0.53 to 4.0*10-2 mmho). Across all age groups, significantly higher RF values were found in OME cases compared to healthy
subjects (all p-values < 0.01). The sensitivity and specificity of PLAI for diagnosing middle ear effusion in children (58 ears) were 60% and 90%,
respectively, with positive and negative predictive values of 74% and 82%, respectively.
Conclusion: PLAI provides reliable data for healthy children of all ages and for children with OME. The average RF and peak admittance values
in children with OME were higher than those observed in healthy controls. Compared to traditional tympanometry, PLAI demonstrated higher
specificity, and a positive predictive value was achieved for diagnosing OME. Clinicians can use PLAI alongside tympanometry for children with
suspected OME when the diagnosis remains uncertain after performing or attempting otoscopy and pneumatic otoscopy.
Introduction
Otitis media with effusion (OME) is a prevalent diagnosis in early childhood. Approximately 90% of children experience OME before school age, with an average of four episodes per ear [1]. Terms such as ear fluid, serous, secretory, or nonsuppurative otitis media are synonymous with OME [2].
In children with OME, fluid in the middle ear typically presents without signs and symptoms of acute middle ear infection [3]. OME often leads to hearing loss in children [4], which can be associated with cognitive development delays [5]. However, many children remain asymptomatic, and many episodes go undetected, including those affecting hearing or school performance [6]. While most episodes of OME resolve spontaneously within three months, 30- 40% of children experience recurrent OME episodes. In 5-10% of children with OME, episodes last ≥ 1 year [1, 7]. Recurrent or persistent fluid in the middle ear due to OME leads to decreased mobility of the tympanic membrane, creating a barrier to sound conduction. (8) About 25% of OME persists ≥ 3 months [9]. Besides hearing loss, OME may be associated with vestibular disorders, behavioral problems, poor academic performance, recurrent acute otitis media, or reduced quality of life [10].
Causes of OME are upper respiratory infections, eustachian tube dysfunction, or inflammatory responses following acute otitis media, primarily between the ages of six months and four years [11]. More than 50% of children experience OME, with the percentage increasing to over 60% by age two [12]. Screening tests in children aged five to six in primary schools in Western Sicily revealed that approximately one in eight had fluid in the middle ear in one or both ears [13].
Pneumatic otoscopy is the gold standard method to diagnose OME, as recommended by clinical practice guidelines [6]. However, this tool has varying degrees of validity and reliability. Consequently, [6] current clinical guidelines suggest a three-month follow-up period to avoid unnecessary surgeries. For observation, otoscopy or tympanotomy is recommended. Tympanometry measures the amount of sound energy reflected back when a small probe is placed in the outer ear canal [14]. The results are displayed through a tympanogram, a graph of energy admitted to the tympanic membrane and middle ear in response to air pressure applied to the outer ear canal. Acoustic energy is transmitted to the ear canal, and an internal microphone measures the reflected sound during pressure changes. Conventional single-frequency 226-Hz tympanometry is used to evaluate the middle ear sound conduction system; however, this method fails to detect subtle changes in the middle ear mechanics [15]. The sensitivity of tympanometry for detecting OME is similar to that of otoscopy (90- 94%). The specificity ranges from 50% to 75% (80% for otoscopy). Studies have shown that tympanometry based on a 226-Hz probe tone is insensitive to OME in infants under six months of age [11, 16, 17]. A comparative study using computed tomography and auditory brainstem response testing revealed that tympanometry with a higher frequency, such as 1000 Hz, is more sensitive to OME in infants younger than six months [18, 19]. Therefore, a higherfrequency probe tone (1000 Hz) is recommended for diagnosing OME in infants [20].
In 2023, a method for pressure-less acoustic immittance (PLAITM) measurement was introduced [21]. This device was developed as a non-invasive tool for assessing middle ear function without pressurization. The technology is based on measuring air velocity in the outer ear canal when an acoustic stimulus is applied at different frequencies to the tympanic membrane. Among various parameters, the admittance curve allows for the calculation of the maximum value of admittance (peak of admittance) and the corresponding average resonance frequency (RF) of admittance [22]. In a preliminary study, PLAI measurements were performed in healthy subjects and patients with OME [23]. The authors found a statistically significant relationship between the RF of the admittance and two tympanometry parameters (equivalent volume and compliance). In a subsequent study, reference values and agespecific ranges were provided for healthy subjects, demonstrating [22] clear age dependence, with a logarithmic fit yielding a robust model for parameter values as a function of age. Considering the reference values, PLAI provides results via dashboards that give a diagnosis of perforation, health, and OME.
Using the PLAI dashboard, we demonstrated normative values of the RF and the peak value of the admittance in healthy adults and patients with various middle ear diseases [24-27]. and compared the results with those of conventional single-frequency tympanometry. PLAI measurements exhibited an opposite pattern compared to tympanometric measurements. Significant underpressure induced by avoiding the opening of the Eustachian tube resulted in a continuous increase in the RF of the admittance [27].
Therefore, this study aims to determine whether PLAI measurements can be used to diagnose OME in children.
Materials and Methods
Ethical considerations
The study design was approved by the Ethics Committee of the General Medical Council of Mecklenburg-West Pomerania (A2024- 0183). This prospective study was conducted in strict adherence to the revised version of the Helsinki Declaration. The study is registered with the German Register of Clinical Studies (DRKS 00035288).
Participants
Only patients with middle ear diseases in one or both ears, aged 0-18 years were considered for the present study. To constitute a control group, healthy subjects in the same age group as the patients were considered.
Overall, 112 children (224 ears), aged three months to 17 years, from two departments of otorhinolaryngology in Kyrgyzstan and in Germany, with a mean age of 8.29 ± 0.45 years (65 boys and 47 girls) and healthy children with normal bilateral ears (N = 49) were included. Three different age groups were considered: 0-2 years (Nears = 26), > 2-6 years (Nears = 64), and > 6-18 years (Nears = 134).
Children with no history of inflammation or disease that has impacted the middle ear, no recent hearing disability or aural symptoms, and a normal tympanic membrane with no atrophy, scarring, retraction, or perforation were defined as normal.
Of the 112 children (224 ears), 49 subjects had bilateral normal ears (98 normal ears), and 63 children had either unilateral (22 patients, 22 ears with pathologies, and 22 normal ears) or bilateral pathologies (41 patients, 82 ears with disease) included. In summary, 120 normal ears (0-2 years (Nears = 15), > 2-6 years (Nears = 30), and > 6-18 years (Nears = 75) and 104 pathological ears (59 OME: (0-2 years (Nears = 9), > 2-6 years (Nears = 28), and > 6-18 years (Nears = 22) were considered.
All 120 normal ears of 71 subjects and 63 patients out of 112 children with various middle ear diseases were included for further analyses (58 ears with OME, 17 ears with tube ventilation disorders [11 ears with mesotympanic retraction, 3 ears with epitympanic retraction, 2 ears with adhesive processes, and 1 atrophic scar], 9 ears with chronic otitis media (COM) with perforation, 3 ears with open mastoid cavities, and 17 other ears [2 cholesteatomas, 1 ear with myringitis, 5 ears with myringosclerosis, 4 ears after cartilage tympanoplasty, and 5 ears with grommets]).
All parents and their children, if appropriate, were informed about the aims of the study and provided their written consent.
Procedure
Medical evaluation
Ear, nose, and throat examinations were performed by experienced ENT surgeons in both departments. Only children with a non-inflamed eardrum showing clear signs of effusion were categorized as having “OME.” All children underwent video otoscopy, and images and videos were stored. Two ENT surgeons later analyzed the data and categorized each ear based on the condition of the outer ear canal and tympanic membrane.
In all healthy children, acute or chronic otitis and other pathologies were excluded. Any pathology of the tympanic membrane (mesotympanic and/or epitympanic retraction, perforation, cholesteatoma, or myringitis) was noted according to the condition of the tympanic membrane in otoscopic findings.
All children older than six years obtained in both centers pure tone audiometry, PLAI using MedWave® (Neuranix, Italy), 226-Hz conventional tympanometry, and finally, video otoscopy. For children younger than six months, 1000-Hz conventional tympanometry was used.
PLAI (Pressure-Less Acoustic Immittance)
The end of a MedWave® probe was inserted into the lateral portion of the external ear canal. This test system applied a signal ranging from 100 to 3000 Hz. Evaluating the responses resulted in the calculation of the complex acoustic admittance. MedWave® provides objective numerical values: the frequency at which the maximum value of the admittance curve is reached (Fr [Hz]) and the peak admittance value (P [*10-2 mmho]). Detailed information is presented elsewhere [23, 28].
Conventional tympanometry
Tympanometry was performed in Güstrow (Germany) using the Madsen® Zodiac (type 1096; Otometrics) at 226 Hz or 1000 Hz from -400 to +200 daPa and in Bishkek (Kyrgyz-stan) using an Inventis middle ear analyzer (Clarinet; Inventis S.R.L.) at 226 Hz or 1000 Hz from -400 to +200 da Pa (maximum range -600 to +400 daPa). The following measures were used for calculation: tympanometric peak pressure (TPP), tympanic width (TW), equivalent ear canal volume (ECV), static peak compliance (SC), and type of tympanogram [Jerger types: A (peak within normal limits for both pressure and height: a normal admittance/compliance reading of 0.3-1.6 mL, a normal middle ear pressure reading of +50 to -50 daPa, and a normal ear canal volume reading of 0.5-2.5 mL), Ad (peak between +100 and -100 daPa, a compliance reading under 0.3 mL, ECV of up to 1.6 mL), As (peak between +100 and -100 daPa, a compliance reading over 1.5 mL, ECV of up to 0.4 mL), B (flat with no significant peak), and C (the height of the peak is or is not within normal limits and is more negative than normal)].
Statistical Analyses
SPSS Version 30.0 (SPSS Inc., Chicago, IL, USA) was used for statistical analyses. Data were evaluated using descriptive statistical methods and comparative tests. The numerical tests were expressed as mean ± standard deviation, and the categorical variables were presented as percentages. Statistical tests (Kolmogorov-test and Shapiro-Wilk test) indicated a non-normal distribution for all parameters (all p-values < 0.05). Nonparametric tests were performed for group comparisons whenever appropriate. Pearson statistics were used for correlational analyses. P value of < 0.05 was accepted as statistically significant in all analyses.
Results
Overall, 120 ears (49 healthy children with bilateral normal ears (N = 98) and the contralateral ear of 22 patients with unilateral ear pathology) were classified as normal using otoscopy and puretone audiometry.
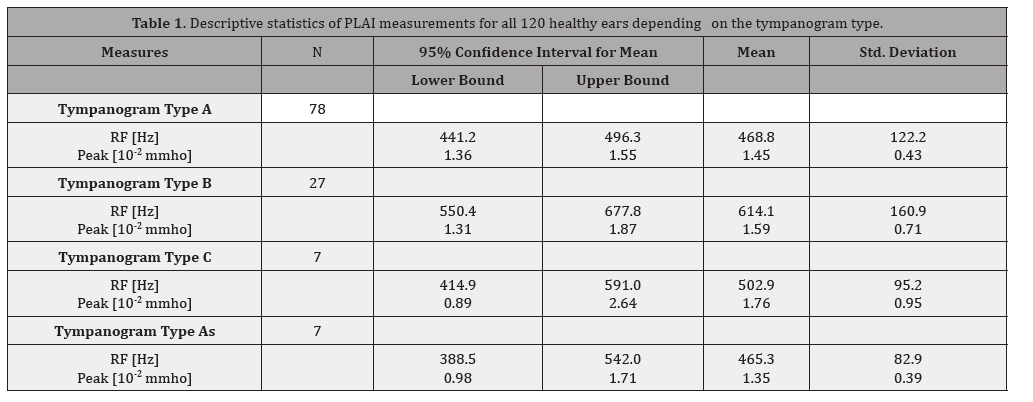
Out of 120 normal ears, 78 had a tympanogram Type A (65%), 27 ears (22.5%) had a tympanogram Type B, while 7 ears (5.8%) showed a tympanogram Type C or as (Table 1). Tympanogram Type Ad was found in less than 1% of the normal-appearing ears assessed via otoscopy.
For all 120 normal-appearing ears assessed via otoscopy, the average RF of the admittance was 503.3 Hz ± 140.6 Hz (ranging from 285.3 to 1119 Hz), and the average peak admittance was 1.50*10-2 mmho (ranging from 0.49 to 3.75*10-2 mmho). Considering age, there was a trend of higher RF of the admittance in younger children compared to those aged >6-18 years (all p-values < 0.05). Figure 1 shows the RFs of the admittance for children under the age of two years, ranging from 453 Hz to 1119 Hz (677.8 Hz ± 218.1 Hz); children > 2-6 years, ranging from 333.8 Hz to 759.1 Hz (522.5 Hz ± 108.8 Hz); and children older than 6-18 years, ranging from 285.3 Hz to 1104 Hz (460.7 Hz ± 100.1 Hz). For all age groups, significantly higher RF values were found in OME compared to healthy subjects (all p-values < 0.01).
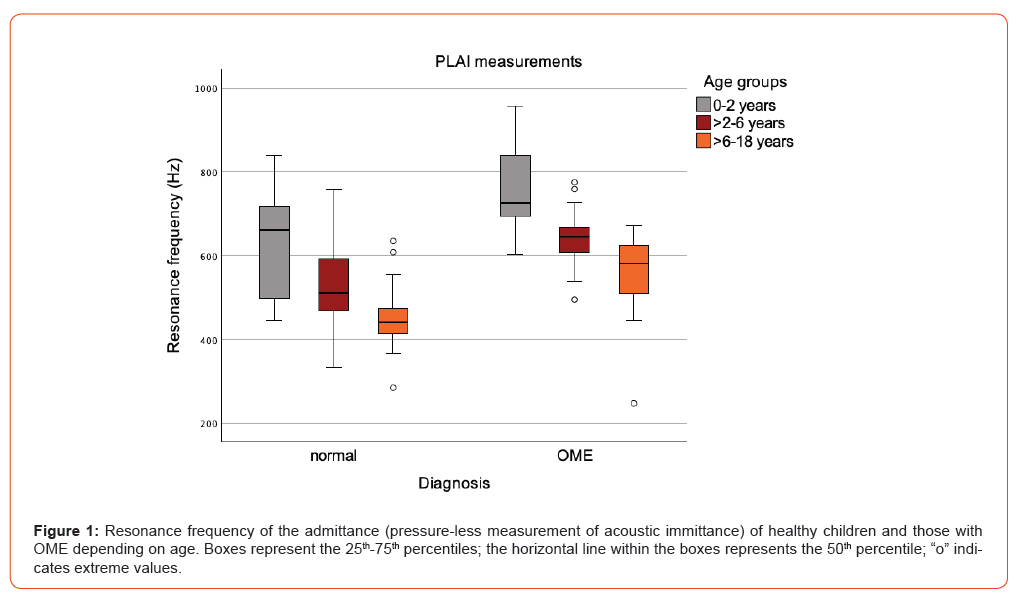

Figure 2 shows the peak admittance values for children under the age of two years, ranging from 0.58*10-2 mmho to 3.67*10-2 mmho (1.45 *10-2 mmho ± 0.81*10-2 mmho); children aged > 2-6 years, ranging from 0.52*10-2 mmho to 2.78*10-2 mmho (1.41*10-2 mmho ± 0.47*10-2 mmho); and children older than 6-18 years, ranging from 0.49*10-2 mmho to 3.75*10-2 mmho (1.55*10-2 mmho ± 0.51*10-2 mmho). No significant differences in peak admittance values were found between the different age groups of the healthy group (all p-values > 0.05). For all age groups, no significant differences were found between the peak measures of OME and healthy subjects (all p-values > 0.05).
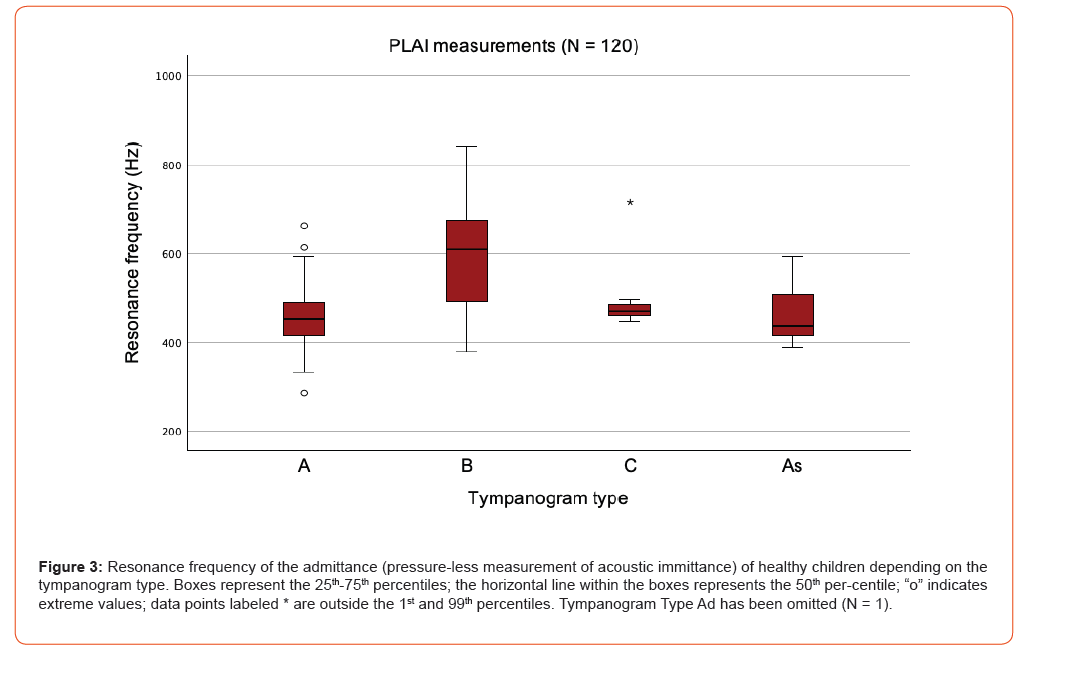
Table 1 shows PLAI measurements depending on the tympanogram type (see also Figures 3 and 4).
Table 1:
Tympanogram type Ad has been omitted (N = 1).
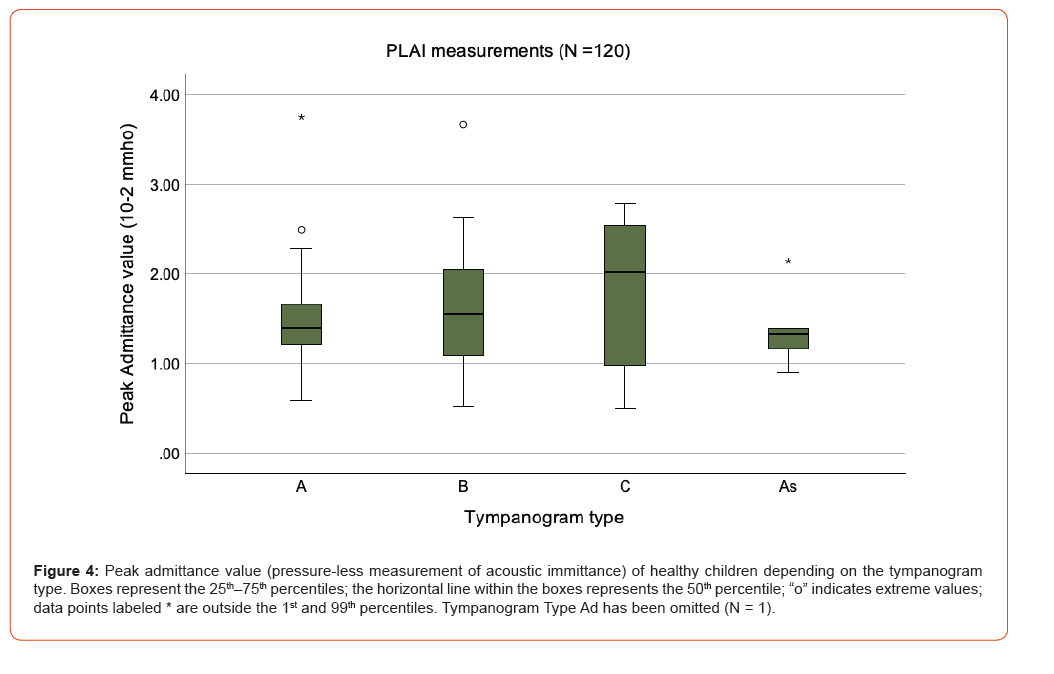
Overall, 90% of all normal-appearing ears received a PLAI diagnosis of “normal” (Table 2), while less than 10% were diagnosed as “soft/perforation” or “rigid/OME.”
Considering tympanometry, 78 out of 120 ears with a normalappearing tympanic membrane had a tympanogram Type A and a PLAI diagnosis of “normal” (65%) (Table 3).
Table 2:
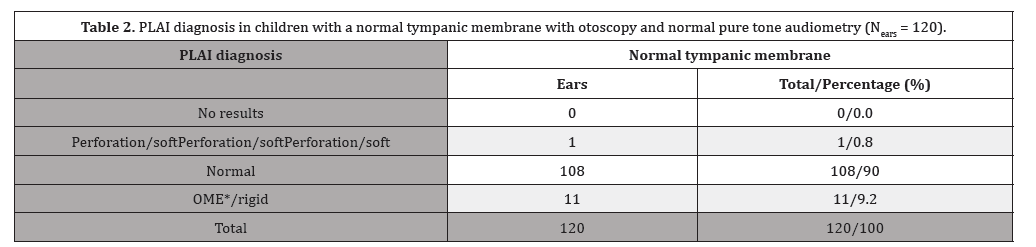
*OME: otitis media with effusion
Table 3:
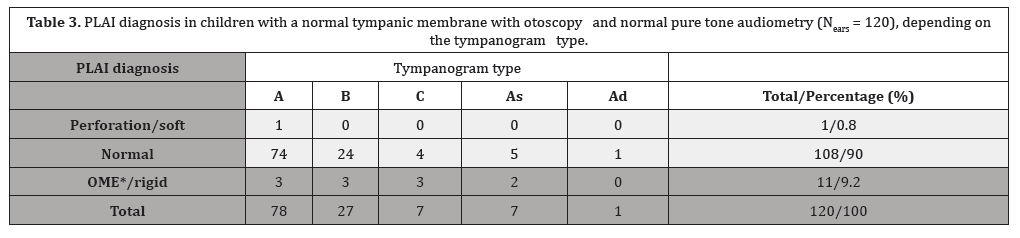
*OME otitis media with effusion
Table 4:
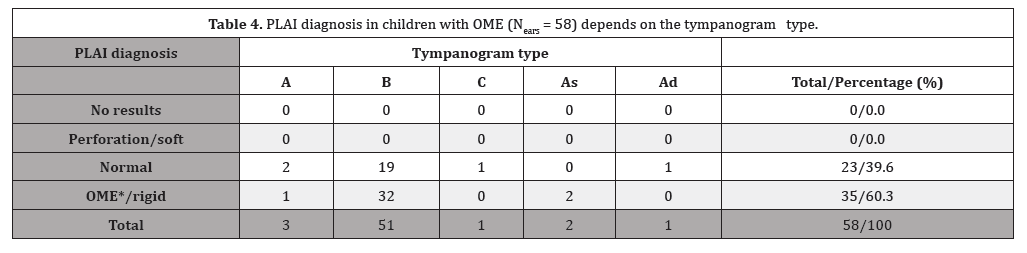
*OME otitis media with effusion
Regarding the relationship between the RF of the admittance and peak admittance values with PLAI, Figure 5 shows only a weak correlation between both parameters in healthy subjects (r120 = 0.26, p < 0.01).
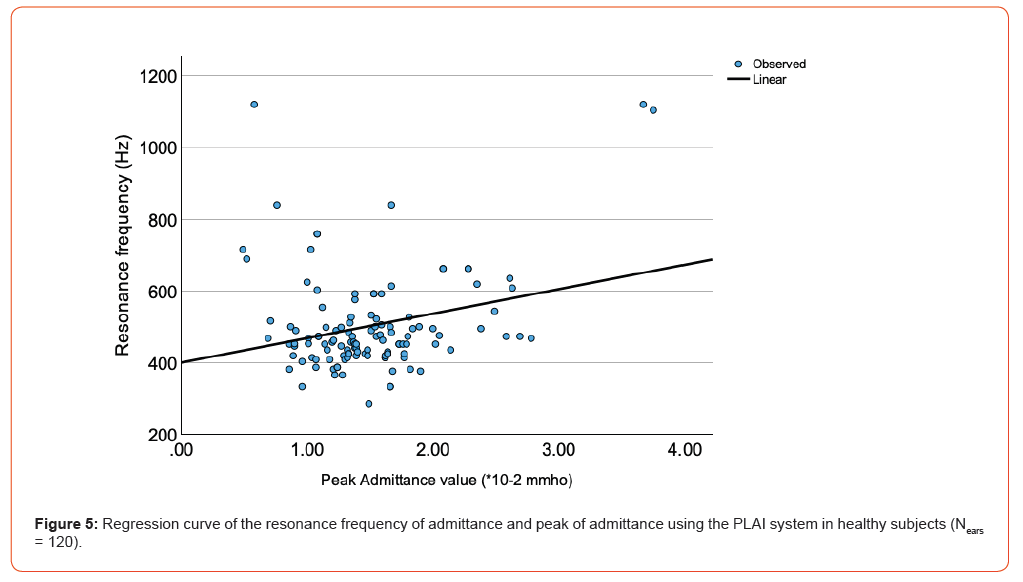
For all 58 ears with OME, the average RF of the admittance was 631.1 Hz ± 115.5 Hz (ranging from 247.6 to 958.2 Hz), and the average peak admittance was 1.68*10-2 ± 0.70 mmho (ranging from 0.53 to 4.0*10-2 mmho). Table 4 shows PLAI measurements depending on all pathologies (see also Figures 3 and 4).
Regarding the relationship between the RF of the admittance and peak admittance values with PLAI, Figure 6 shows only a weak, non-significant correlation between both parameters in patients with OME (r58 = -0.18, p > 0.05).
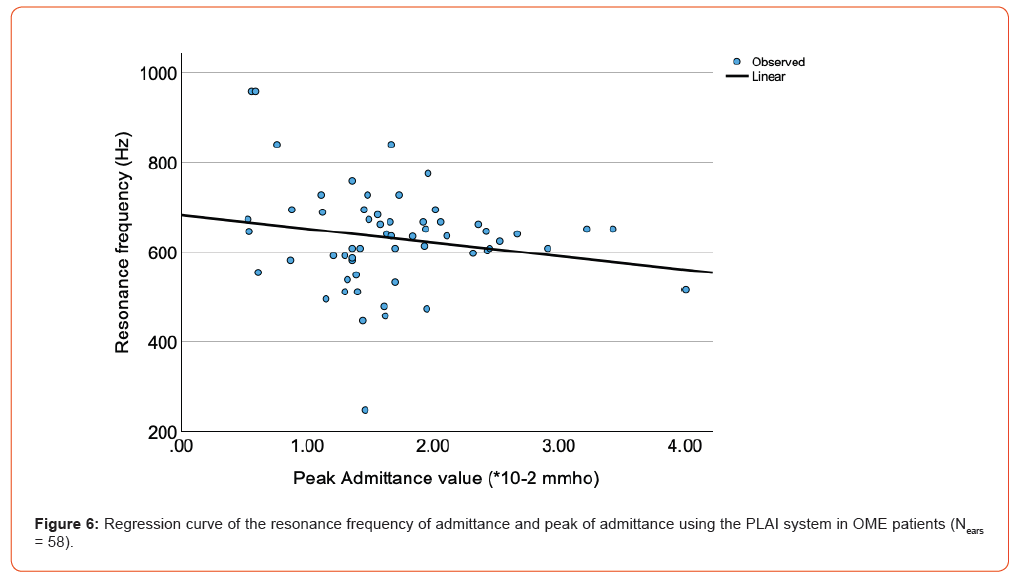
The sensitivity and specificity of PLAI for diagnosing middle ear effusion in children (58 ears) were 60% and 90%, respectively. The positive and negative predictive values (PPV and NPV) were 74% and 82%, respectively. In comparison, tympanometry revealed a sensitivity of 95%, a specificity of 65%, and PPV and NPV of 57% and 96%, respectively.
Discussion
Since OME cannot be diagnosed accurately without myringotomy, reliable findings are essential to guide clinicians in the diagnosis and follow-up of OME [6, 29, 30]. Otoscopy is crucial for the diagnosis of middle ear effusion; however, children sometimes resist this examination. Although otoscopy has good sensitivity, its specificity for diagnosing OME is low [31, 32]. Recent papers and guidelines have recommended pneumatic otoscopy as the primary diagnostic tool for OME [6, 32, 33]. For various reasons, many clinicians rely on information from otoscopy and overlook pneumatic otoscopy [34]. Although pneumatic otoscopy provides high sensitivity and specificity for diagnosing OME, its use in daily practice remains controversial. Both tympanometry and pneumatic otoscopy fail to reliably detect effusion [35]. Wideband acoustic immittance (WAI) measurements encompass a variety of parameters (power reflectance, power absorbance, impedance, and others) derived from outer ear canal recordings in response to the stimulus [36]. WAI evaluates the acoustic response properties of the middle ear across a wide frequency range (226-8000 Hz) and can be obtained at ambient ear canal pressure or during dynamic pressure sweeps [37]. WAI testing performed in the presence of pressure sweeps is referred to as wideband tympanometry (WBT). WAI data displayed as absorbance as a function of frequency are termed wideband absorbance (WBA). Often, WBA data are extracted from a WBT test at ambient ear canal pressure (or at 0 daPa) and at TPP. WBT is described as a powerful and sensitive indicator for detecting middle ear effusion volume in children with OME [35]. Studies have examined the relationship between tympanic membrane changes determined by otoscopy and pneumatic otoscopy over time and WBA. While 226 Hz tympanometry could not determine otoscopic changes between the initial and first-month control, WBA at 2520 Hz and 3175 Hz was moderately correlated with otoscopic findings. Ellison et al. found that 1500-3000 Hz at ambient pressure was the most sensitive frequency range for differentiating between OME and the control group [29]. The density of fluid within the middle ear does not seem to affect WBA measurements at ambient pressure. Aithal et al. found no differences between low and high fluid density [38]. At peak pressure, they reported differences between the two groups.
Several studies have shown that higher reflectance and lower absorbance are found in OME-affected ears [38-40]. Similar results with more sensitive absorbance measurements at peak pressure have been demonstrated. As a result of these studies, absorbance measurements at peak pressure are recommended [17, 41, 42].
In our study, we measured the RF of the admittance and the peak of admittance in children. The PLAI results were compared with tympanometric measures. Otoscopy was used to categorize the disease. In all children diagnosed with OME, honey-colored fluid behind the intact, gray-appearing tympanic membrane was observed. However, it cannot be completely excluded that some ears categorized as healthy may have undetected middle ear fluid. In all children with a normal-appearing tympanic membrane, there was no indication of myringotomy. In only 65% of the normally categorized ears, a tympanogram Type A was noted. In 22.5%, a tympanogram Type B was found, and in 5.8%, tympanogram Types C and as were observed. These tympanometric findings in 42 ears cannot be explained by middle ear fluid behind the tympanic membrane. This finding highlights the limitations of tympanometry in subjects with varying middle ear and mastoid volumes. Previous studies demonstrated an RF of about 400 Hz and a peak of admittance of about 1.65*10-2 mmho in healthy adults [24, 25]. Healthy children exhibited higher RF values and lower peak admittance values than adults. With increasing age, children present decreasing RF values, averaging 460 Hz in the age group older than 6–18 years. RF values were significantly higher in all age groups with OME compared to the healthy control group. These findings can be attributed to increased stiffness caused by middle ear effusion. Similar results were found in patients with severe retraction pockets with effusion in adults, in adhesive processes, and in otosclerosis [24-26]. However, an increased RF of the admittance also indicates heightened stiffness of the tympanic membrane and middle ear and is not solely related to OME. Therefore, the sensitivity and specificity values for diagnosing OME should be interpreted cautiously. A slight to moderate conductive hearing loss in children with a normal-appearing tympanic membrane may also be caused by juvenile otosclerosis or congenital stapes fixation [43, 44]. PLAI measurements improved specificity and the PPV compared to tympanometry. However, sensitivity and NPV were lower compared to tympanometry. Medical reports noting a progression of hearing loss, family history, and otoscopy remain indispensable prerequisites for obtaining an accurate diagnosis in children.
Conclusion
PLAI provides reliable data for healthy children of all ages and for children with OME. RF of the admittance and peak admittance were higher than those in healthy controls. Compared to tympanometry, PLAI achieved higher sensitivity and positive predictive values for diagnosing OME. Clinicians can use PLAI in combination with tympanometry for children with suspected OME when the diagnosis remains uncertain after performing or attempting otoscopy and pneumatic otoscopy.
Funding
This research received no external funding.
Conflict of Interest
The authors declare that there are no conflicts of interest.
Acknowledgment
The authors thank Axel Monien, Anna Bäumer, Tino Bittner, and Daniel Wacker for their assistance with data collection.
References
- Tos M (1984) Epidemiology and natural history of secretory otitis. Am J Otol 5(6): 459-462.
- Mandel EM, Doyle WJ, Winther B, Alper CM (2008) The incidence, prevalence and burden of OM in unselected children aged 1-8 years followed by weekly otoscopy through the "common cold" season. Int J Pediatr Otorhinolaryngol 72(4): 491-499.
- Berkman ND, Wallace IF, Steiner MJ, Harrison M, Greenblatt AM, et al. (2013) Otitis Media with Effusion: Comparative Effectiveness of Treatments. AHRQ Comparative Effectiveness Reviews. Rockville (MD).
- Vanneste P, Page C (2019) Otitis media with effusion in children: Pathophysiology, diagnosis, and treatment. A review. J Otol 14(2): 33-39.
- Stevenson J, McCann D, Watkin P, Worsfold S, Kennedy C (2010) Hearing Outcomes Study T. The relationship between language development and behaviour problems in children with hearing loss. J Child Psychol Psychiatry 51(1): 77-83.
- Rosenfeld RM, Shin JJ, Schwartz SR, Coggins R, Gagnon L, Hackell JM, et al. (2016) Clinical Practice Guideline: Otitis Media with Effusion Executive Summary (Update). Otolaryngol Head Neck Surg 154(2): 201-214.
- Williamson IG, Dunleavey J, Bain J, Robinson D (1994) The natural history of otitis media with effusion--a three-year study of the incidence and prevalence of abnormal tympanograms in four South West Hampshire infant and first schools. J Laryngol Otol 108(11): 930-934.
- Williamson I (2002) Otitis media with effusion. Clin Evid 2002(7): 469-476.
- Rosenfeld RM, Kay D (2003) Natural history of untreated otitis media. Laryngoscope 113(10): 1645-1657.
- Rosenfeld RM, Schwartz SR, Pynnonen MA, Tunkel DE, Hussey HM, et al. (2013) Clinical practice guideline: Tympanostomy tubes in children. Otolaryngol Head Neck Surg 149(1 Suppl): S1-35.
- Paradise JL, Rockette HE, Colborn DK, Bernard BS, Smith CG, et al. (1997) Otitis media in 2253 Pittsburgh-area infants: prevalence and risk factors during the first two years of life. Pediatrics 99(3): 318-333.
- Casselbrant ML, Mandel EM. Epidemiology. In: Rosenfeld RM, Bluestone CD. Evidence-based otitis media. (2nd), Canada: BC Decker Inc; 2003. pp. 147-162.
- Martines F, Bentivegna D, Di Piazza F, Martinciglio G, Sciacca V, Martines E (2010) The point prevalence of otitis media with effusion among primary school children in Western Sicily. Eur Arch Otorhinolaryngol 267(5): 709-714.
- Onusko E (2004) Tympanometry. Am Fam Physician 70(9): 1713-1720.
- Margolis RH, Hunter LL, Giebink GS (1994) Tympanometric evaluation of middle ear function in children with otitis media. Ann Otol Rhinol Laryngol Suppl 163: 34-38.
- Marchant CD, McMillan PM, Shurin PA, Johnson CE, Turczyk VA, et al. (1986) Objective diagnosis of otitis media in early infancy by tympanometry and ipsilateral acoustic reflex thresholds. J Pediatr 109(4): 590-595.
- Hunter LL, Prieve BA, Kei J, Sanford CA (2013) Pediatric applications of wideband acoustic immittance measures. Ear Hear 34 Suppl 1: 36S-42S.
- Baldwin M (2006) Choice of probe tone and classification of trace patterns in tympanometry undertaken in early infancy. Int J Audiol 45(7): 417-427.
- Zhiqi L, Kun Y, Zhiwu H (2010) Tympanometry in infants with middle ear effusion having been identified using spiral computerized tomography. Am J Otolaryngol 31(2): 96-103.
- American Academy of Pediatrics JCoIH (2007) Year 2007 position statement: Principles and guidelines for early hearing detection and intervention programs. Pediatrics 120(4): 898-921.
- Graffigna CE, Stanzial D (2021) Apparatus and method for measuring clinical-audiometric parameters. Italy 2021 European patent 4 125 577 B1.
- Bassi F, Miladinovic A, Accardo A (2025) Relations between age, tympanometric parameters and pressure less measurements. 9th National Bioengineering Group (GNB) Congress; 2025. Palermo, Italy.
- Bassi F, Accardo A (2024) Estimation of middle ear characteristics by an innovative pressure-less acoustic immittance (PLAI™) device. 9th European Medical and Biological Engineering Conference; 2024; Portoroz, Slovenia: EMBEC 2024, IFMBE Proceedings 113.
- Anbari M, Zhumabaeva S, Beishenova M, Fischer D, Sulaimanov S, Just T (2024) Comparison of conventional single-frequency tympanometry with pressure-less acoustic immittance measurements of pathological and normal middle ears. On J Otolaryngol & Rhinol 7(2): 1-11.
- Anbari M, Zhumabaeva S, Beishenova M, Fischer D, Sulaimanov S, Just T (2025) Pressure-less acoustic immittance measurements of patients with persistent ventilation disorders. On J Otolaryngol & Rhinol 7(3): 1-11.
- Kamin B, Anbari M, Fischer D, Just T (2025) Pressure-less acoustic immittance measurements in diagnosis of otosclerosis. On J Otolaryngol & Rhinol 7(4): 1-9.
- Just T, Anbari M, Monien A, Fischer D (2025) Changes of the resonance frequency of the middle ear without opening of the Eustachian tube. Br J Cancer Res 8(1):742-6.
- PLAI® methodology used in med wave®: Neuranix; 2024.
- Ellison JC, Gorga M, Cohn E, Fitzpatrick D, Sanford CA, (2012) Wideband acoustic transfer functions predict middle-ear effusion. Laryngoscope 122(4): 887-894.
- Aithal S, Aithal V, Kei J (2024) Wideband Absorbance Predicts the Severity of Conductive Hearing Loss in Children With Otitis Media with Effusion. Ear Hear 45(3): 636-647.
- Nozza RJ, Bluestone CD, Kardatzke D, Bachman R (1994) Identification of middle ear effusion by aural acoustic admittance and otoscopy. Ear Hear 15(4): 310-323.
- Cho YS, Lee DK, Lee CK, Ko MH, Lee HS (2009) Video pneumatic otoscopy for the diagnosis of otitis media with effusion: a quantitative approach. Eur Arch Otorhinolaryngol 266(7): 967-973.
- Terzi S, Ozgur A, Erdivanli OC, Coskun ZO, Ogurlu M, et al. (2015) Diagnostic value of the wideband acoustic absorbance test in middle-ear effusion. J Laryngol Otol 129(11): 1078-1084.
- Engel J, Anteunis L, Chenault M, Marres E (2000) Otoscopic findings in relation to tympanometry during infancy. Eur Arch Otorhinolaryngol 257(7): 366-371.
- Keskin Yildiz M, Atay G, Mergen EK, Aksoy S, Boke B (2025) A novel approach to the follow up of children with otitis media with effusion: wideband absorbance findings. Eur Arch Otorhinolaryngol.
- Rosowski JJ, Stenfelt S, Lilly D (2013) An overview of wideband immittance measurements techniques and terminology: you say absorbance, I say reflectance. Ear Hear 34 Suppl 1(0 1): 9S-16S.
- Sanford CA, Hunter LL, Feeney MP, Nakajima HH (2013) Wideband acoustic immittance: tympanometric measures. Ear Hear 34 Suppl 1: 65S-71S.
- Aithal V, Aithal S, Kei J, Anderson S, Wright D (2020) Predictive Accuracy of Wideband Absorbance at Ambient and Tympanometric Peak Pressure Conditions in Identifying Children with Surgically Confirmed Otitis Media with Effusion. J Am Acad Audiol 31(7): 471-484.
- Stuppert L, Nospes S, Bohnert A, Lassig AK, Limberger A, et al. (2019) Clinical benefit of wideband-tympanometry: a pediatric audiology clinical study. Eur Arch Otorhinolaryngol 276(9): 2433-2439.
- Hunter LL, Tubaugh L, Jackson A, Propes S (2008) Wideband middle ear power measurement in infants and children. J Am Acad Audiol 19(4): 309-324.
- Shahnaz N, Feeney MP, Schairer KS (2013) Wideband acoustic immittance normative data: ethnicity, gender, aging, and instrumentation. Ear Hear 34 Suppl 1: 27S-35S.
- Keefe DH, Archer KL, Schmid KK, Fitzpatrick DF, Feeney MP, et al. (2017) Identifying Otosclerosis with Aural Acoustical Tests of Absorbance, Group Delay, Acoustic Reflex Threshold, and Otoacoustic Emissions. J Am Acad Audiol 28(9): 838-860.
- Bachor E, Just T, Wright CG, Pau HW, Karmody CS (2005) Fixation of the stapes footplate in children: a clinical and temporal bone histopathologic study. Otol Neurotol 26(5): 866-873.
- Vick U, Just T, Terpe H, Graumüller S, Pau HW (2004) Stapes fixation in children. HNO 52(12): 1076-1082.
-
Munar Beishenova, Shirin Zhumabaeva, Blanda Kamin, Moayad Anbari, Dörte Fischer, Shayirbek Sulaimanov and Tino Just*. Pressure-less Acoustic Immittance Measurements in Children with Otitis Media with Effusion. On J Otolaryngol & Rhinol. 7(5): 2025. OJOR.MS.ID.000672
-
Otitis media, Middle ear, Otoscopy, Tympanometry, Hearing loss, Vestibular disorders, ENT surgeons, Otosclerosis, Audiometry, Nonparametric tests
-

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.