 Research Article
Research Article
Determination Of the Amount of Peptide Bound Amino Acids in Food and Feed Matrices – Comparability of Hydrolysates (Oxidized or Unoxidized Samples) Between Digestion Using A Heating Oven/Heating Block and Microwave Digestion
Klaus Schwadorf 1*, Daniela Dircks1, Gernot Kopp2
1Core Facility Hohenheim (CFH, 640), Analytical Chemistry, University of Hohenheim, Emil-Wolff-Strasse 12, 70599 Stuttgart, Germany
2MLS Mikrowellen-Labor-Systeme GmbH, MWS Mikrowellensysteme-Vertriebs GmbH, Auenweg 37, 88299 Leutkirch, Germany
Klaus Schwadorf, Core Facility Hohenheim (CFH, 640), Analytical Chemistry, University of Hohenheim, Emil-Wolff-Strasse 12, 70599 Stuttgart, Germany.
Received Date:March 06, 2026; Published Date:March 19, 2026
Abstract
A reliable determination of the levels of relevant amino acids in food and feed matrices generally requires validated analytical methods, such as the official testing method for food [1] and/or for feed [2]. The official methods specify temperature in an oven of 110 °C and time duration of 24 hours for the hydrolysis step and as analytical device for the determination of amino acids from the hydrolysates an Amino Acid Analyzer (AAA) or HPLC equipment with ion exchange column, device for ninhydrin post-column derivatization and photometric detector. Microwave digestion resp. microwave hydrolysis would be an interesting alternative (time savings, economic benefits, individual temperature control of the contents of each sample vessel, reproducibility and improved laboratory safety). But are the results of the amino acid determination using microwave digestion comparable to those of the oven resp. heating block, as described in the official methods? The aim of this work is to determine whether both methods can demonstrate satisfactory agreement or equivalence.
Introduction
Proteins, along with fats and carbohydrates, are among the main or basic nutrients in food and feed. Amino acids are the building blocks of these proteins and peptides and are organic compounds that contain at least one amino group and one carboxyl group as their functional group. Approximately 500 amino acid structures are known, with 20 - 23 amino acids are naturally incorporated into proteins (“proteinogenic AAs”). 20 AAs are encoded by the universal genetic code (“canonical AAs”–DNA, RNA). Each protein is char acterized by a specific amino acid sequence (primary structure).
The importance of proteins in the human and animal body is manifold; for example, they function as enzymes, hormones, structural proteins, antibodies, and transport proteins. It is important to focus on essential and limited amino acids. Essential amino acids cannot be synthesized by human and animal body and have to be supplied through diet. For the human body, for example, 8 (-10) amino acids are essential (Lysine, Methionine and/or Cyst(e)in, Threonine, Valine, Leucine, Iso-Leucine, Phenylalanine and/or Tyrosine and Tryptophan + Histidine for children + Taurine for newborn babies). Depending on the dietary habits of humans or the feeding options of animals, dietary supplements are recommended or added. Some amino acids are present in low proportion and so limit the functioning of the proteins (biological value). To ensure complete protein diets, complementation is recommended (combination of different food proteins and/or supplementation of diets).
A reliable determination of the levels of relevant amino acids in food and feed matrices generally requires validated analytical methods (see Methods). The use of such analytical methods in laboratories includes internal quality controls (e. g. suitable and tested equipment, use of certified chemicals, use of certified reference materials, co-analysis of control samples) as well as external quality controls (laboratory participation in formal intercomparisons), particularly within the framework of accreditation, for example, according to DAkkS DIN EN ISO/IEC 17025. If a laboratory decides to perform the determination of amino acids or the amino acid profile according to one of the aforementioned official methods and cites this method in the results report, then the laboratory is generally obligated to check any deviations in equipment or other aspects from the specifications for their equivalence, to revalidate the method, and to document the validation.
Materials and Methods
Materials
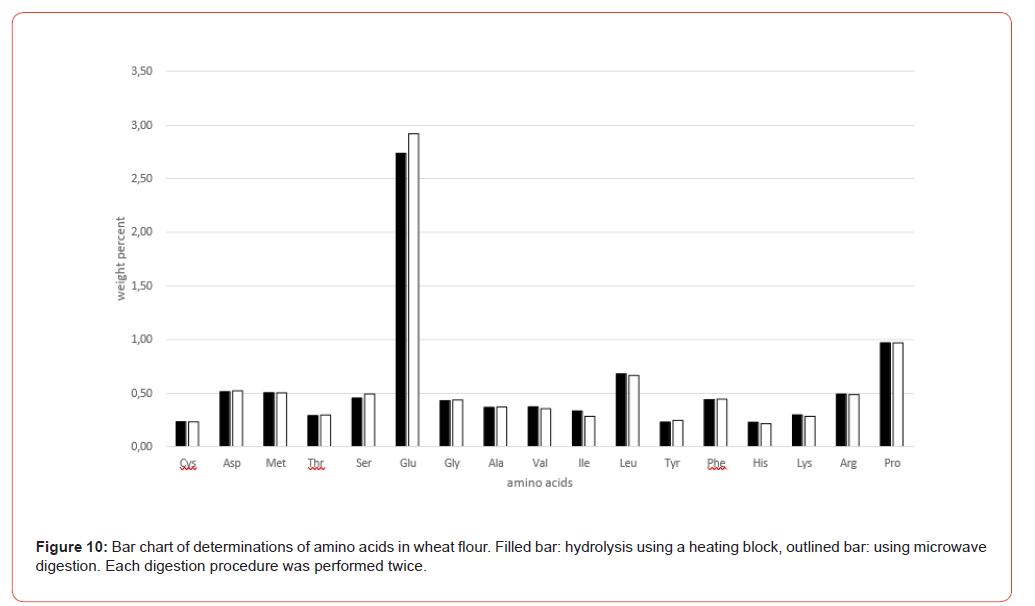
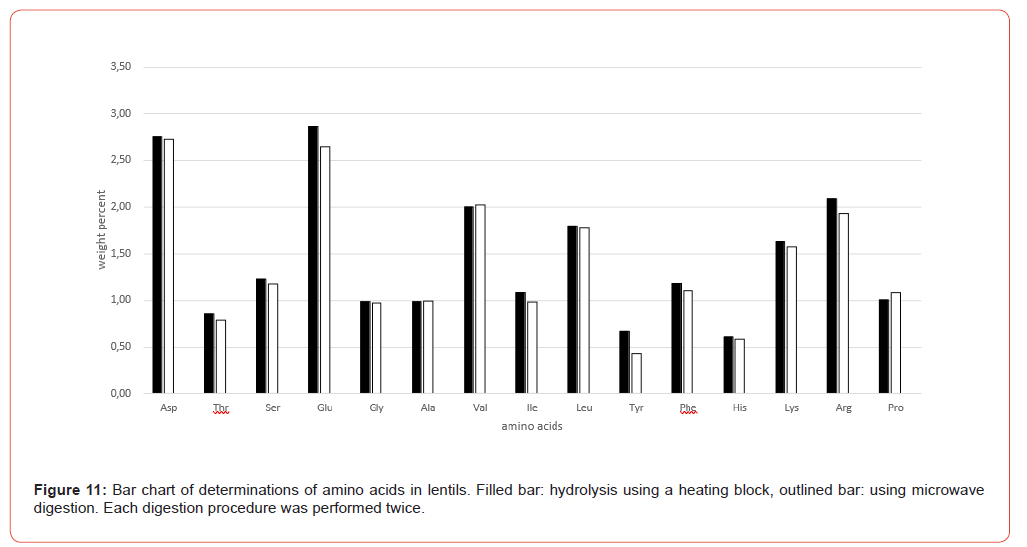
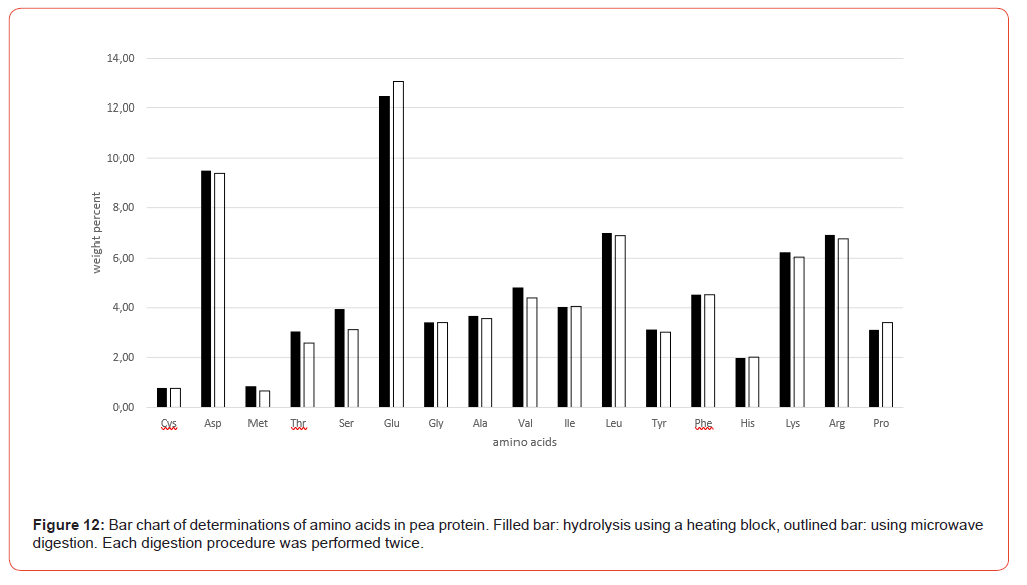
For comparative testing, a selection of different sample matrices was used, largely covering the range of food and feed samples, including 2 proviciency test samples from the annual VDLUFA quality assurance collaborative trial, a formal intercomparison for external quality control [3]: Wheat flour, lentils, pea protein, yeast protein, soy protein, cassava leaves, collagen powder, sausage casing (made from bovine split, gelatine/collagen), turkey fattening feed (VDLUFA ring test sample 2025, no. 505Q) and supplementary feed for dairy cows (VDLUFA ring test sample 2026, no. 512Q).
Sample Devider and Grinding or Crushing Test Samples
Pelletized, granular and powdered samples were divided using a RETSCH PT 100 sample divider with DR 100 (RETSCH GmbH, 42781 Haan, Germany) and ground with rotor high-speed mill FRITSCH Pulverisette 14 (12-rib mill’s rotor) to a sieve size of 1 mm (1 mm sieve ring) (FRITSCH GmbH, 55743 Idar-Oberstein, Germany). All other sample matrices were gently crushed to a particle size of 0.75 - 1 mm using suitable shredding equipment and homogenized.
Heating Block Device
Heating block according to Kjeldahl, Type KT 20s with Controller Type TRs CSB/SMA for 20 samples each run (see Figure 1; C. Gerhardt GmbH & Co. KG; 53639 Königswinter, Germany). The hydrolyses were carried out in digestion tubes for hydroxyproline determination, 250 mL, with GL45 (P/N 12-0316, C. Gerhardt …). To estimate or control the actual temperature inside the digestion tubes, one digestion tube per load is filled with only the hydrolysis solution and tightly sealed with a rubber stopper. A thermometer (Testo 110; Testo SE & Co. KgaA, 79822 Titisee-Neustadt, Germany) is inserted through this rubber stopper.
Microwave Laboratory Device
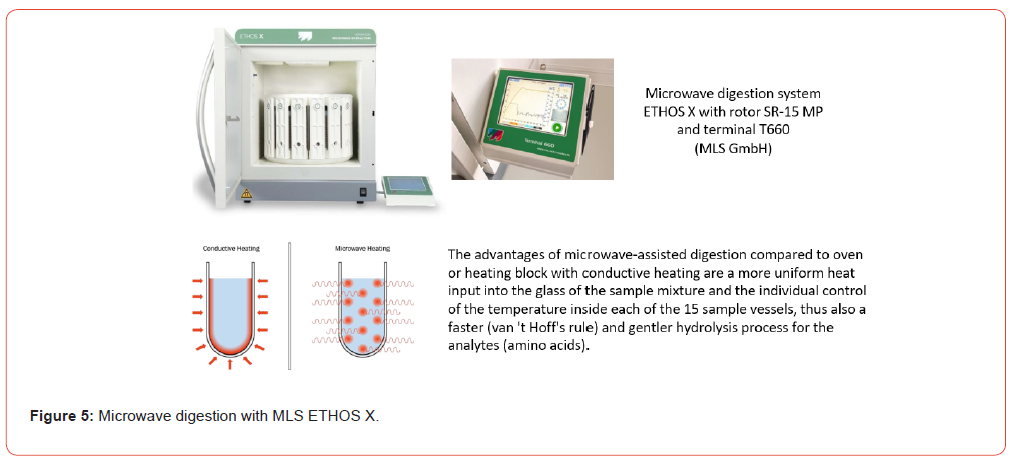
Professional Microwave System ETHOS X (MLS LC01014) equipped with control terminal 660 and easy Control software (MLS TER66050) (MLS GmbH, 88299 Leutkirch, Germany). Rotor turnable SR-15 MP ITC (15 places each load) (MLS LCR15030D) ITC 310 Infrared temperature control (non-contact temperature control of all samples through vessel walls made of PTFE and glass using IR sensor technology) (MLS IRT0300C) Rotor segment SR-12/15 with threaded insert and ITC measurement (MLS LCR0012I) Glas inserts (roll border vials/headspace N 20-50 DIN) (LLG Labware Art- No 7.060 459; 53340 Meckenheim, Germany or Roth Art No LC67.1; Carl Roth GmbH & Co. KG, 76185 Karlsruhe, Germany) PTFE lids Ø1 6x16 mm with slot 2mm, for roll border vials (MLS DD00210) Magnetic stir bar SE, Ø 9 x 15 mm, for glass insert extraction segment (glass container) (MLS MR 90001) PTFE container with IR control, ID=32 mm (MLS HB00036A) Protective jacket with finned cooling (MLS DM00050) PTFE sealing film Ø 40x 0.3 mm (MLS APU6102) + valve cover (MLS DD00195) TFM lid for PTFE container with ID=32 mm (MLS DD00068)
Amio Acid Analyzer Device
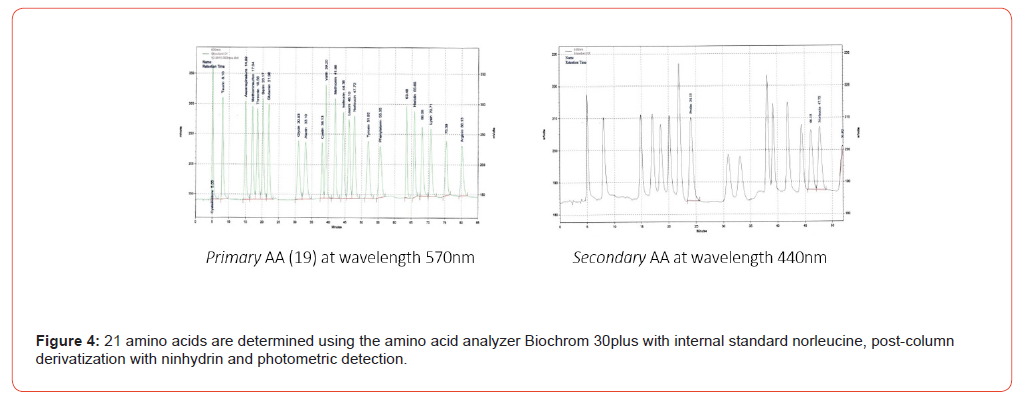
All amino acid analysis were performed using the amino acid analyzer Biochrom 30plus with Lithium buffer system (Biochrom Ltd., Cambridge, UK, CB25 9PE; Laborservice Onken GmbH, 63584 Gründau, Germany). Separation by ion exchange chromatography (oxidized feedstuff column, type cation exchanger resin, 200 x 4.6 mm), post-column reaction with ninhydrin and photometric detection (570 nm for primary AA and 440 nm for secondary AA; see Figure 4).
Internal standard for chromatography: Norleucine standard solution at a concentration of 400 μg/mL (DL-Norleucine p.a., e. g. Sigma-Aldrich N1398-5G). Standard solution for use as a basic calibration standard for the amino acid analyzer: AA calibration standard, feed hydrolysate (oxidized hydrolysate amino acids), Art-No 5.403.154, and AA calibration standard collage-hydrolysate, Art-No 5.403.176 (Laborservice Onken GmbH, 63584 Gründau, Germany). Certified standard solution for cross-checking: NIST Standard Reference Material SRM 2389a, amino acids in 0.1 mol/L hydrochloric acid (National Institute of Standards and Technology, Gaithersburg, MD 20899, USA).
All chemicals used are of analytical grade quality
The buffer solutions for operating the Biochrom 30plus amino acid analyzer are ready-made solutions from Biochrom and were purchased from Laborservice Onken GmbH, 63584 Gründau, Germany.
Methods
The official testing method for amino acids in food or feeding stuffs is according to § 64 of the German Food and Feed Code [1] resp. Commission Regulation (EC) No 152/2009 [2]. In contrast to the analysis of free amino acids or physiological solutions, the analytical determination of individual amino acids or the amino acid profile in food and/or feed matrices (predominantly peptide- bound) requires that they first be released from the protein matrix via a digestion step. Depending on which amino acids are being analyzed, the sample matrices must first be hydrolyzed with hydrochloric acid. Sulfur-containing amino acids (Methionine, Cyst(e)ine) must extra be protected by an additional oxidation step before the actual hydrolysis with hydrochloric acid (see Figure 1). The amino acid tryptophan is destroyed by both hydrolysis and oxidation and must be hydrolyzed with alkaline solution in a separate procedure (see Figure 2). The official analytical procedures describe the hydrolysis step, regardless of any prior oxidation, as follows: “Place the bottle containing the mixture prepared in accordance with … in an oven at 110 °C … for 24 hours …”. Instead of a laboratory warming oven, a metal heating block can also be used, with the temperature of 110 °C and the reaction time of 24 hours remaining unchanged in both cases. It should be noted, however, that the temperature of 110 °C refers to the matrix mixture to be hydrolyzed into the glass vessel, not what the display of the oven or controller shows, which is difficult to control with these devices. Furthermore, the official methods specify the chromatographic determination of the amino acid hydrolysates by using an amino acid analyzer (AAA) or HPLC equipment with ion exchange column, device for ninhydrin post-column derivatization and photometric detector. Unfortunately, it makes no mention of other equipment that is indispensable in modern analytics (see Figure 3). Microwave digestion can represent a viable alternative for various reasons, particularly the time savings in the hydrolysis step (see Figure 5). The aim of this work is to optimize microwave digestion to achieve results comparable to those obtained with oven resp. heating block digestion in amino acid determination.
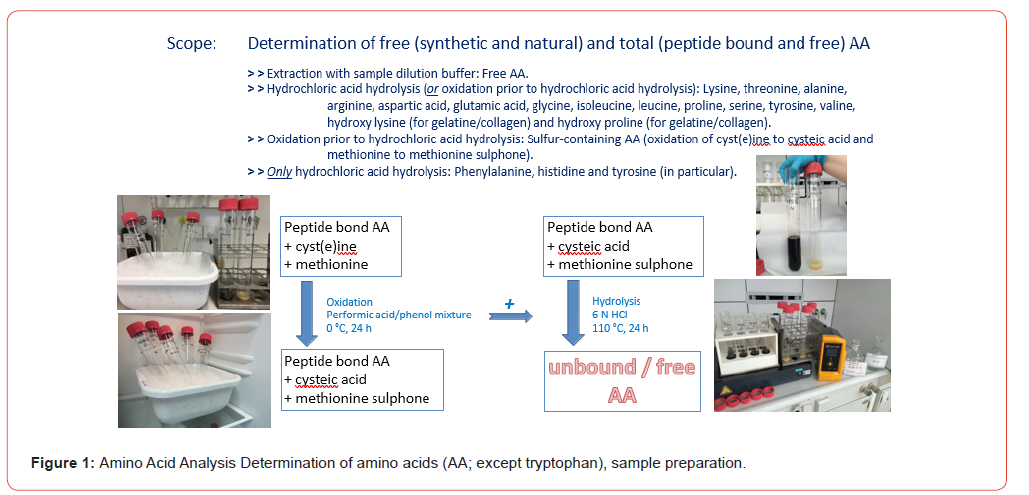
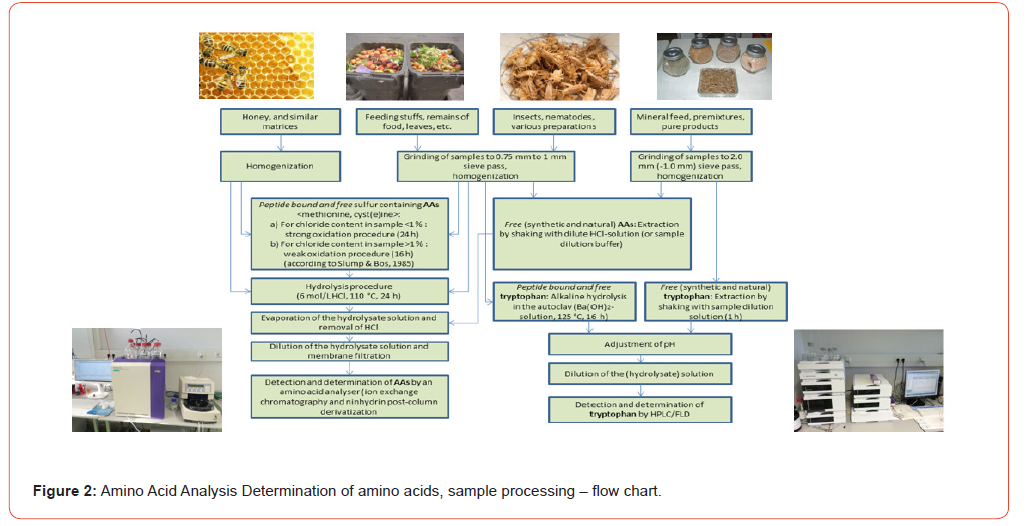

Conducting the tests
Brief Description of the Procedure
The homogenized sample, optionally after prior oxidation with an oxidation solution, is hydrolyzed with hydrochloric acid in a closed digestion tube for the heating block or in a closed microwave digestion vessel (PTFE container with protective sleeve) equipped with a glass insert. An aliquot of the hydrolysate is purified, then concentrated using a rotary evaporator to remove the hydrochloric acid, and the residue is dissolved in a defined volume of sample dilution buffer (pH 2.20; Lithium Loading Buffer, Biochrom 80-2038- 10).
Performing hydrochloric acid hydrolysis (microwave digestion)
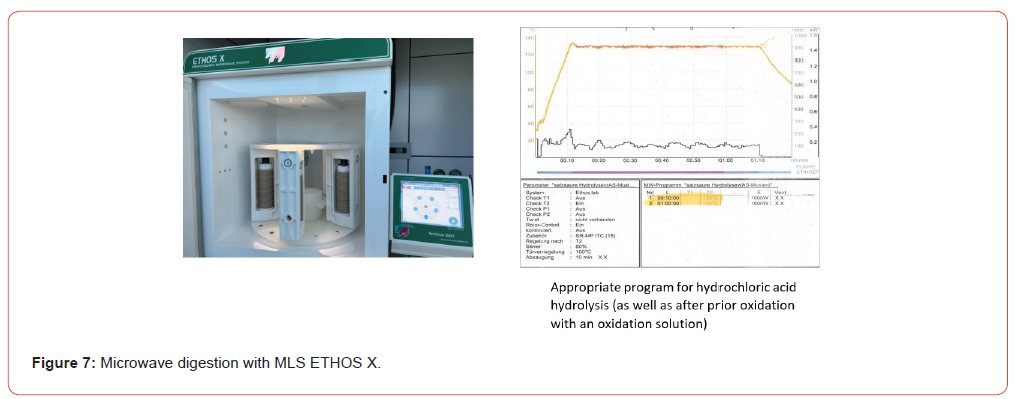
Weigh 0.5 - 1.0 g of the homogenized sample into a glass insert (roll border vial) and attach a magnetic stir bar (Ø 9 x 15 mm). The sample is then mixed with approximately half of the 25 mL of 6 molar hydrochloric acid, the insert covered with a PTFE lid, and stirred on the magnetic stirrer until no lumps remain. Once all lumps have dissolved, add the remaining volume of HCl. The glass insert is now placed in the prepared PTFE container, which contains 4 mL of pure water. This PTFE container is then enclosed in the protective jacket and tightly sealed in the rotor segment with the corresponding TFM lid for container including the PTFE sealing film/valve cover. Place the rotor segments with the digestion vessels in the microwave oven‘s holder and start the appropriate program (Figure 6,7). After sufficient cooling time (approx. 15 min; sample temperature <30 °C) the PTFE container is carefully opened and the hydrolysate is transferred quantitatively to a suitable volumetric flask (100 mL). Fill the volumetric flask to the mark with pure water and shake. The content of the flask is filtered through a folded filter paper (Ø 18.5 cm, e. g. MN 615 ¼; Macherey-Nagel, 52355 Düren, Germany) into a 100 mL Erlenmeyer flask. Take an aliquot of this filtrate, corresponding to the target content or crude protein content, and pipette it into a 100 mL round bottom flask. Add 1 mL of internal standard solution (norleucine solution, 400 μg/mL). Using a rotary evaporator, concentrate the content until dry. The water bath temperature of the rotary evaporator must not exceed 40 °C. The concentration process should be repeated at least twice with pure water to remove the hydrochloric acid completely (Figure 8). Dissolve the content of the round bottom flask with sample dilution buffer (pH 2.20) using an ultrasonic bath. Transfer the mixture carefully and quantitatively into a 20 mL volumetric flask, rinsing with small volumes several times, and fill the flask to the mark. Shake the contents of the 20 mL volumetric flask vigorously. Immediately afterwards, this solution is membrane-filtered (disposable cellulose acetate membrane syringe filter, pore diameter 0.22 μm, filter diameter 25 mm; LLG Labware Art-No 9.055 501; 53340 Meckenheim, Germany) into a suitable autosampler vial for analysis using the Biochrom 30plus amino acid analyzer (Figures 3, 4).


Performing the oxidation prior to the hydrochloric acid hydrolysis (microwave digestion)
Weigh 0.5 - 1.0 g of the homogenized sample into a glass insert (roll border vial) and attach a magnetic stir bar (Ø 9 x 15 mm). Using a 5 mL piston-stroke pipette, 2.5 mL of oxidation solution (1 part by volume of 30% hydrogen peroxide with 9 parts by volume of 88% phenol-containing formic acid) is added and the glass insert covered with a PTFE lid. Stir on the magnetic stirrer until no lumps remain. If this is insufficient, a brief ultrasonic treatment can be performed, followed by the addition of the remaining 2.5 mL of oxidation solution (in total 5 mL). The glass insert is then placed in an ice bath and left therefore 24 hours using a refrigerator. After 24 hours, the ice bath with glass inserts is taken out of the refrigerator. To stop the oxidation, two spatula tips of sodium disulfite (> 98 %, e. g. Carl Roth-Nr. P755.1, 76185 Karlsruhe, Germany) are added into the glass insert. Then 25 mL of hydrolysis solution (1.0 g of phenol is added to 1 L of 6-molar hydrochloric acid) are added, the glass insert is resealed with the lid, and the mixture is stirred again (Figure 6). Now the glass insert is placed in the prepared PTFE container which contains 4 mL of pure water. This PTFE container is then enclosed in the protective jacket and tightly sealed in the rotor segment with the corresponding TFM lid for container, including the sealing film/valve cover. Place the rotor segments with the digestion vessels in the microwave oven’s holder and start the appropriate program (Figure 6, 7).
After sufficient cooling time (approx. 15 min; sample temperature <30 °C) the PTFE container is carefully opened and the hydrolysate is transferred quantitatively to a suitable volumetric flask (100 mL). Fill the volumetric flask to the mark with pure water and shake. The content of the flask is filtered through a folded filter paper (Ø 18.5 cm, e. g. MN 615 ¼; Macherey-Nagel, 52355 Düren, Germany) into a 100 mL Erlenmeyer flask. Take an aliquot of this filtrate, corresponding to the target content or crude protein content, and pipette it into a 100 mL round bottom flask. Add 1 mL of internal standard solution (norleucine solution, 400 μg/mL). Using a rotary evaporator, concentrate the content until dry. The water bath temperature of the rotary evaporator must not exceed 40 °C. The concentration process should be repeated at least twice with pure water to remove the hydrochloric acid completely (Figure 8).
Dissolve the content of the round bottom flask with sample dilution buffer (pH 2.20) using an ultrasonic bath. Transfer the mixture carefully and quantitatively into a 20 mL volumetric flask, rinsing with small volumes several times, and fill the flask to the mark. Shake the contents of the 20 mL volumetric flask vigorously. Immediately afterwards, this solution is membrane-filtered (disposable cellulose acetate membrane syringe filter, pore diameter 0.22 μm, filter diameter 25 mm; LLG Labware Art-No 9.055 501; 53340 Meckenheim, Germany) into a suitable autosampler vial for analysis using the Biochrom 30plus amino acid analyzer (Figures 3, 4).
Calibration and Measurement
The Biochrom 30plus amino acid analyzer is calibrated with the AA calibration standard solutions (Art-No 5.403.154, resp. Art-No 5.403.176) in μg/mL or μmol/mL. For this purpose, 30 μL of the amino acid calibration standard solution are injected repeatedly until the retention time and peak areas are constant.
One sequence consists of two injections of the amino acid calibration standard solution, where the calibration is performed on the 2nd chromatogram, then one injection of the NIST amino acid solution SRM 2389a and again the amino acid calibration standard solution as a test sample, the amino acid calibration standard solution from the last sequence, followed by 8 test samples for analysis.
Results and Discussion
To assess the variations or deviations of the hydrolysis method in the determination of amino acids using oven/heating block and microwave methods, the analytical tolerances (ATs) for amino acids defined by the VDLUFA could be used [4]. The ATs developed and published by the VDLUFA Feed Analysis Working Group are based on interlaboratory comparisons for quality assurance, of which more than 460 have been conducted to date. These comparisons are based on the test results obtained from these VDLUFA interlaboratory comparisons for all participants in the survey (in dry matter), from which – after eliminating any outliers – the standard deviation (relative repeatability standard deviation SR or relative comparison standard deviation VR) is calculated. The specified ATs correspond to a comparison range of ± 2 SR or ± 2 VR, corresponding to a statistical confidence level of P = 95%, meaning that in only 5% of cases is it expected that a test result will deviate from the mean of all results by more than ± 2 SR (or ± 2 VR). When conducting these interlaboratory analyses, sample preparation (grinding and splitting of samples) is the responsibility of the participating laboratories. This ensures that not only the variations of the analytical method are incorporated into the ATs, but also those of the sample preparation in the laboratories. This complies with the requirements for determining measurement uncertainty as formulated in DIN EN ISO/IEC 17025-2018-03, section 7.6.1.
The ATs thus correspond to an „Expanded Measurement Uncertainty“ as required by Annex II, Part C, No. 6 of Regulation (EC) No. 152/2009 for the assessment of undesirable substances. ATs were determined and established for amino acids lysine, cysteine, methionine, threonine (and tryptophan), which may be present in the matrix as additives and/or ingredients. The results and statistical evaluations of the VDLUFA interlaboratory studies led to the development of ATs that necessitate a concentration-dependent scale.
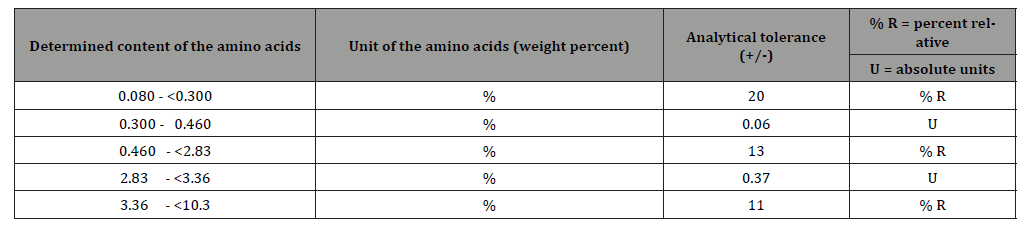
The following scaled 5 concentration ranges were established for the aforementioned amino acids: Note: Since a sliding scale was used for this classification, in addition to the relative percentages, absolute were defined for two ranges (Table 1).
Table 1:Analytical tolerances for amino acids [4].
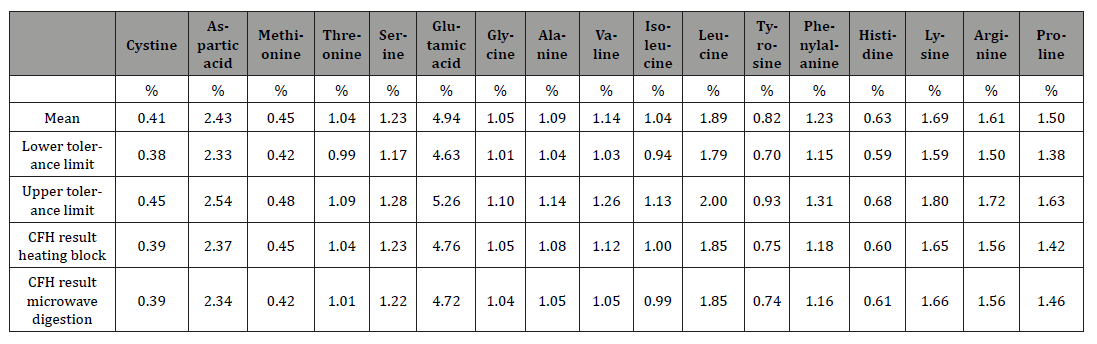
From the perspective of error propagation, the largest analytical error in determining the amino acid content in different matrices by means of hydrolysis is therefore probably to be found there. After developing and subsequently optimizing the microwave method, a comparison between this hydrolysis method with the traditional method using an oven or heating block shows good agreement. The amino acid values from the statistical evaluation of the 2025 ring test sample support this. All results were between the lower and upper tolerance limits. Likewise, no outliers were found (Table 2).
Table 2:VDLUFA ring test sample 2025, no. 505Qa (Turkey fattening feed). Results of the interlaboratory comparison evaluation. Number of participating laboratories
per parameter: between 17 and 21. Results for each individual amino acid in weight percent and based on dry matter. The statistical evaluation of the
VDLUFA feed survey 505 Q was carried out according to DIN ISO 5725-2 using the Program “ProLab Plus, V: 2024.7.30.0” from QuoData, Dresden, Germany.
The evaluation was carried out according to the following algorithm: Calculation of the mean value and tolerance limits, taking into account all submitted data.
<Lower/upper tolerance limit = mean -/+ 2 x comparison standard deviation>.
Using the ring test sample 2026, it was shown how the level of the individual amounts of amino acids and their deviations behave in multiple determinations (entire sample preparation, including hydrolysis and oxidation + hydrolysis) with the traditional method using a heating block compared to the microwave method. The sample was processed 16 times, including 6 hydrolysis using a heating block and 10 hydrolysis using the microwave method. The numerical results are shown in Table 3.
Table 3:VDLUFA ring test sample 2026, no. 512Qb (Supplementary feed for dairy cows). Multiple determinations resp. hydrolysis (6 times using a heating block, 10 times using a microwave method). All analytical values are based on original substance (with a water content of 8.98 %).
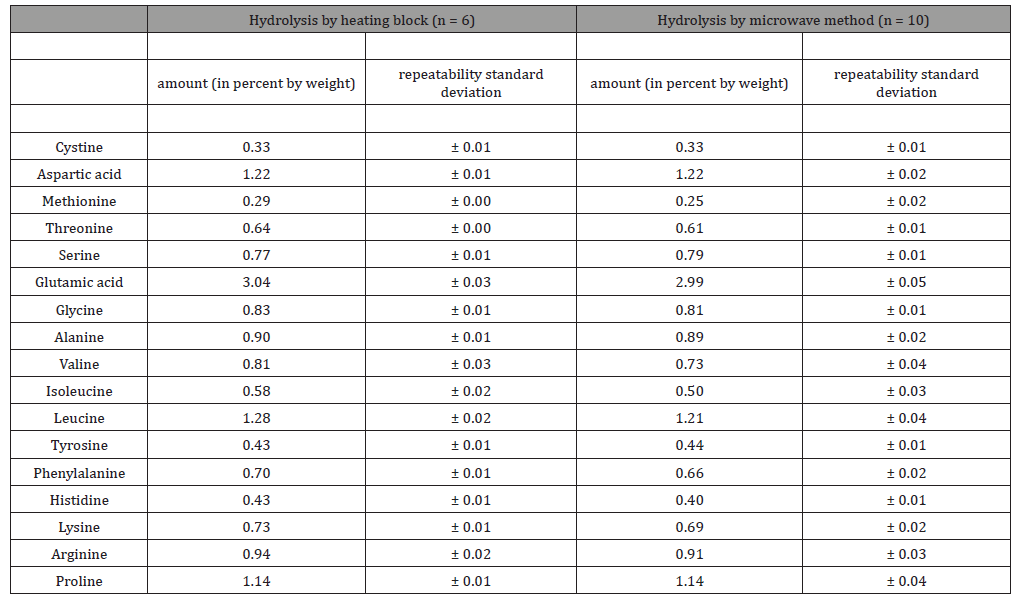
Figure 9 shows the amino acid contents of the individual sample preparations of Table 3 as bar charts, illustrating the variations both in their entirety and separately according to the hydrolysis methods (heating block and microwave methods).
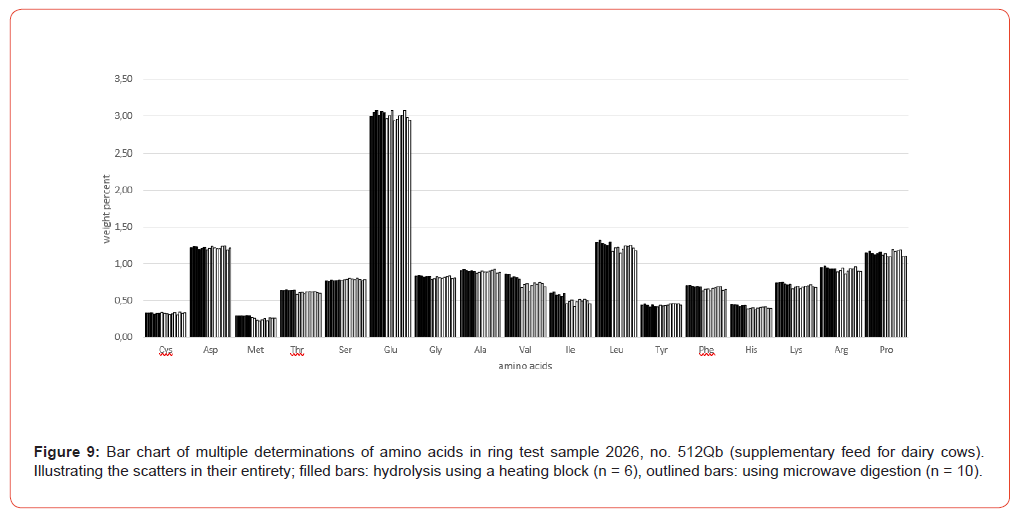
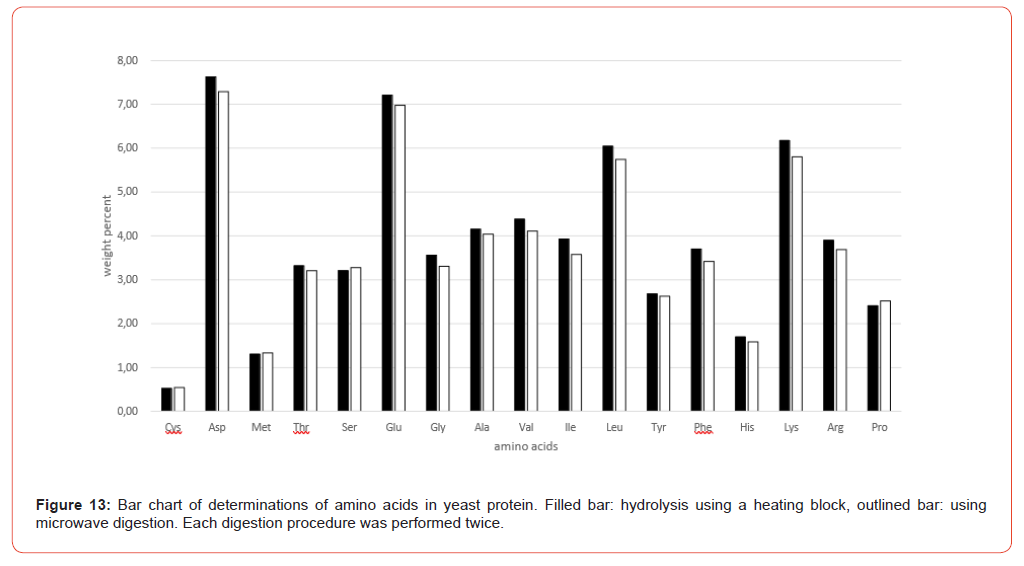
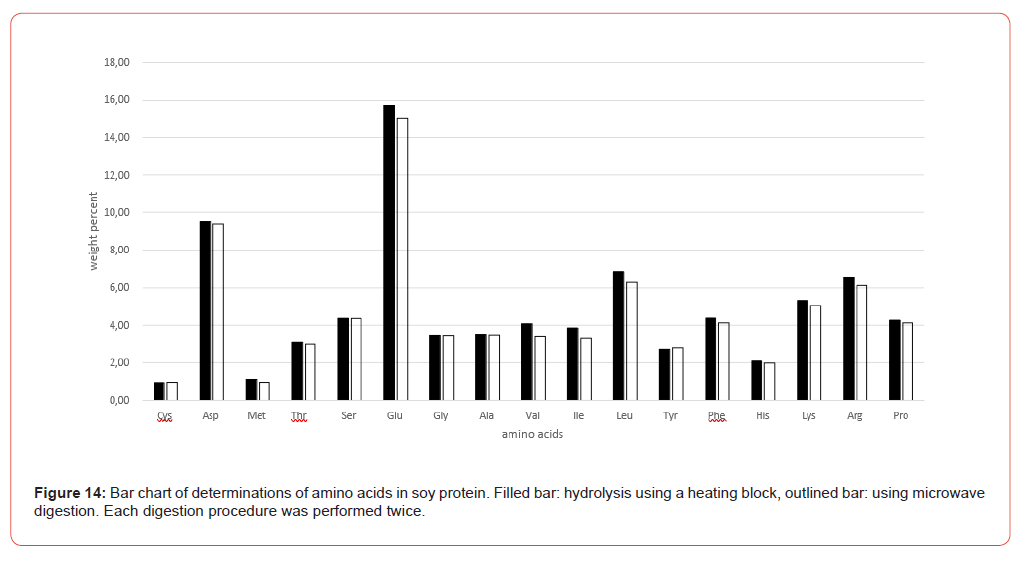
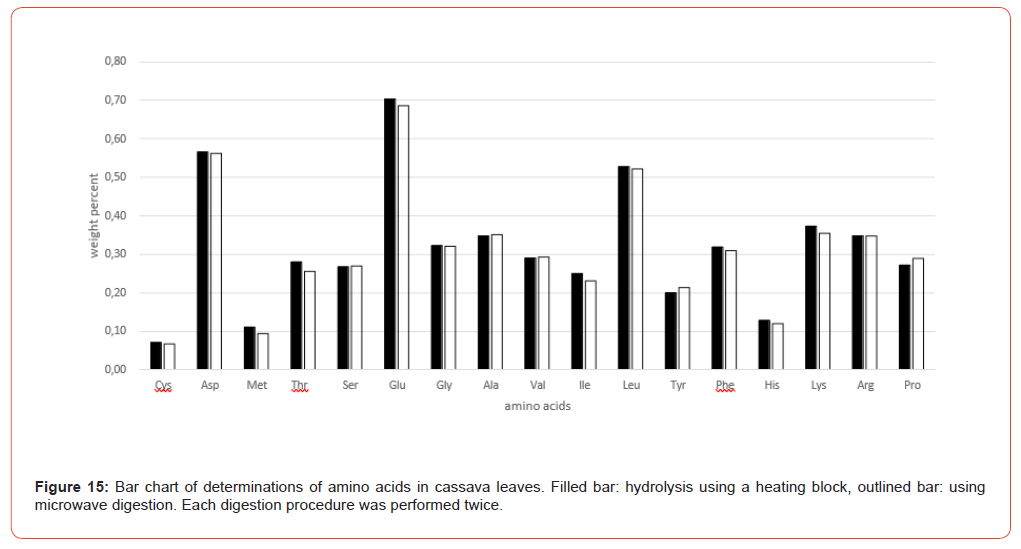
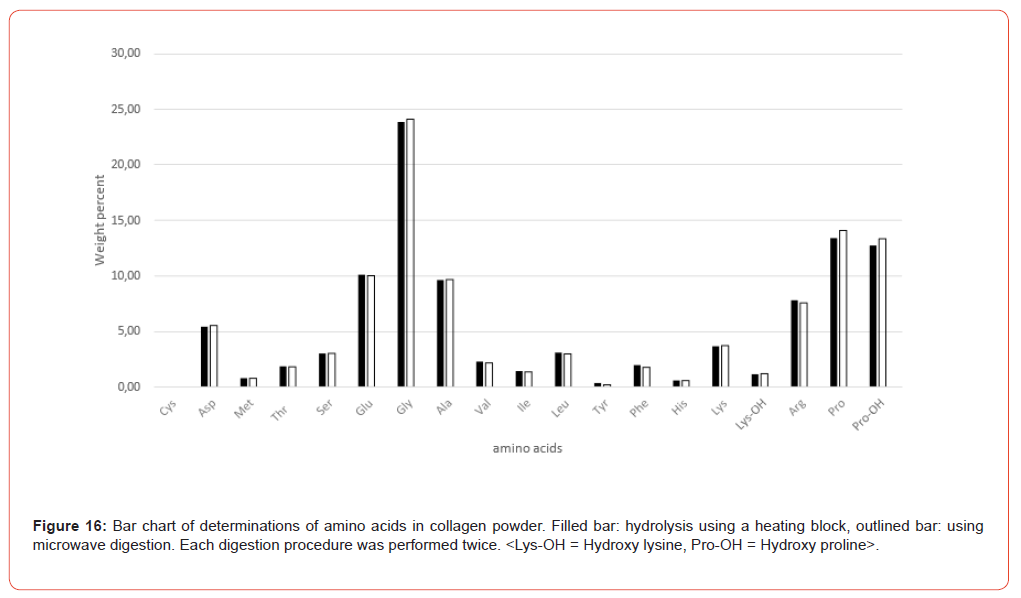
The amino acid content analyzed after the hydrolysis processes both using heating block and microwave methods is in very good agreement in terms of order of magnitude. It is sometimes noticeable that some amino acids like glutamic acid, Valine, Isoleucine and Leucine shows a slightly higher scatter (Figure 9). These scatters are mostly matrix-related and not uncommon in multiple determinations, where the overall error is influenced not only by sample preparation but also by the homogeneity of the sample material and thus a certain degree of inaccuracy in weighing, plus the entire sample preparation procedure up to the preparation of the analytical solution. The final analytical measurement of the sample solution using AAA contributes the least error in this analysis. Taking the VDLUFA ATs into account, these apparent variations are put into perspective.
Conclusion
The microwave digestion method described here, along with the comparative studies, has demonstrated that microwave analysis is a highly efficient tool for amino acid analysis, yielding reproducible and reliable results in multiple assays. The significant reduction in the time required for amino acid hydrolysis from 24 hours (for max. 20 positions in an oven/heating block) to approximately 1.5 hours (for max. 15 positions in microwave) is enormous. In addition to the time-saving aspect, there is also the chemical-saving aspect and thus the environmental benefit: Instead of 100 mL of hydrochloric acid resp. 70 mL of hydrolysis solution (for oxidation + hydrolysis of methionin and cysteine) in the oven/heating block method, only 25 mL resp. 25 mL, are required in the microwave method, per each sample preparation. All of this results in substantial economic benefits. Furthermore, the closed microwave system eliminates the risk of bursting glass vessels, burns from hot hydrochloric acid, etc., thus significantly improving workplace safety.
References
- Amtliche Sammlung von Untersuchungsverfahren (ASU) nach § 64 LFGB < Official Collection of Investigation Procedures > BVL L49.07-01: Untersuchung von Lebensmitteln; Bestimmung der Aminosäuren in Aminosäurengemischen, 1-3 < Examination of foodstuffs;determination of amino acids in amino acid mixtures >BVL L49.07-02: Untersuchung von Lebensmitteln; Bestimmung des Aminosäurengehaltes in diätetischen Lebensmitteln auf der Basis von Proteinhydrolysaten, 1-3< Examination of foodstuffs;determination of the amino acid content in dietary foods based on protein hydrolysates >LFGB = Lebensmittel- und Futtermittelgesetzbuch < German Food and Feed Code >BVL = Bundesamt für Verbraucherschutz und Lebensmittelsicherheit < Federal Office for Consumer Protectionand Food Safety >.
- Commission Regulation (EC) No 152/2009 of 27 January 2009 laying down the methods of sampling and analysis for the official control of feed. Official Journal of the European Union, 52 (2009), Annex III F, Determination of amino acids (except tryptophan), L54/23-L54/32.
- VDLUFA = Verband Deutscher Landwirtschaftlicher Untersuchungs- und Forschungsanstalten Association of German Agricultural Analytic and Research Institutes. Organizer oft the annual quality assurance collaborative trial: VDLUFA Section VI ‚Feedstuff Analysis.
- Analysenspielräume (ASR) (2022) VDLUFA, Fachgruppe Futtermitteluntersuchung, S. 10 analytical tolerances (AT), version 13 (2022);VDLUFA, Feed Analysis Working Group. p. 10.
-
Klaus Schwadorf *, Daniela Dircks, Gernot Kopp. Determination Of the Amount of Peptide-Bound Amino Acids in Food and Feed Matrices – Comparability of Hydrolysates (Oxidized or Unoxidized Samples) Between Digestion Using A Heating Oven/Heating Block and Microwave Digestion. Insi in Chem & Biochem. 3(3): 2026. ICBC. MS.ID.000565.
-
Peptide-Bound Amino Acids; Amino Acid Analyzer
-

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.






