 Case Report
Case Report
Congenital CMV Infection and Brain Pseudocysts
Geethanath Ruppa Mohanram1* and George Aaron2
1Neonatal Department, Sunderland Royal Hospital, Sunderland, United Kingdom
2Neonatal Department, Sunderland Royal Hospital, Sunderland, United Kingdom
Geethanath Ruppa Mohanram, Neonatal Department, Sunderland Royal Hospital, Sunderland, United Kingdom.
Received Date: August 08, 2019; Published Date: August 13, 2019
Abstract
Cytomegalovirus is the commonest cause of congenital infection worldwide causing serious disease in newborns resulting in lifelong neurological impairment. It is the leading infectious cause of hearing loss in children. It causes brain injury due to direct cytopathic effects causing neuronal lysis and indirectly due to immune mediated damage and hypoxic brain damage secondary to placental insufficiency. It affects all cell types, but it shows neurotropism and has a predilection for neural stem cells and progenitor cells. It is estimated to affect 1000 babies every year in UK and has enormous economic impact. It is estimated to cost the United Kingdom 750 million pounds every year. CMV can also cause miscarriage, still birth, preterm birth and it is implicated in sensorineural hearing loss, cerebral palsy, epilepsy, autism spectrum disorder etc.
Key words: Congenital CMV infection; Brain injury; Pseudocyst
Abbreviations:CMV: Cytomegalo-Virus; SEPC: Subependymal Pseudocyst; DNA: Deoxyribonucleic Acid; PCR: Polymerase Chain Reaction; IgM: Immunoglobulin M Introduction
Introduction
Cytomegalovirus is the most prevalent infection causing neurological dysfunction in the developing brain. It is the most common infection known to be transmitted in utero. In the United Kingdom, CMV has been implicated in 12% of sensorineural hearing loss and 8% of cerebral palsy in children [1]. Primary CMV infection in pregnancy has a 40% risk of congenital CMV infection [2]. Cranial ultrasound findings in infants with congenital CMV infection include ventricular dilatation, periventricular pseudocysts, lenticulostriate vasculopathy, periventricular calcification and cerebellar hypoplasia [3]. Intraventricular haemorrhage is the commonest cause for subependymal pseudocysts (SEPC). SEPC of nonhaemorrhagic origin should prompt to search for neurotropic congenital viral infections, particularly cytomegalovirus infections. A 5-week-old baby who had bilateral SEPC on cranial ultrasound had congenital CMV infection.
Case Presentation
A female baby was delivered by an emergency caesarean section for suboptimal CTG at 37 weeks gestation to a primigravida mother with an uneventful pregnancy. The birth weight was 2.15kg and head circumference was 31.0cms both of which were below 0.4th centile. There were no dysmorphic features at birth and the baby was otherwise clinically well. She was observed for 24 hours and sent home after establishing feeds. There were no concerns regarding blood sugar during the stay. In view of growth retardation, placenta was sent for histopathological examination. The placenta showed evidence of multifocal chronic villitis and cytomegalic inclusion bearing cells were noted which was confirmed by immunohistochemistry. This was suggestive of chronic CMV villitis. Following this report, baby was reviewed at the age of 4 weeks. She was still jaundiced at 4 weeks of age without any petechial rash. Liver and spleen were enlarged. An ophthalmology examination revealed normal media with no active retinitis but there were 2 small retinal scars in right eye and one small retinal scar on the left eye. Baby was investigated with blood and urine tests for CMV infection which were positive for CMV DNA by PCR with high viral load. A cranial ultrasound examination showed bilateral subependymal pseudocysts with a typical honeycomb pattern. She had a normal hearing screen and was confirmed to have congenital CMV infection as the blood test from the mother was positive for CMV IgM in the stored blood which was taken at 9 weeks gestation during booking appointment. She was commenced on Valganciclovir which she tolerated very well and thriving without any side effects. A follow up examination at 5 months of age showed normal neurodevelopment without any sequelae with the viral load falling nicely (Figure 1&2).
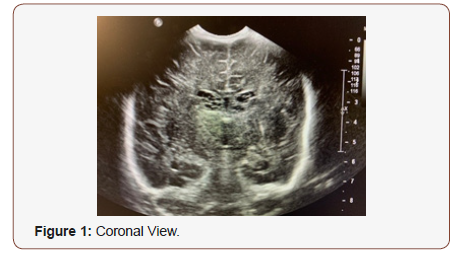

Discussion
Cytomegalovirus infects all cell types, but it shows a preferential tropism for neural stem/progenitor cells4. The germinal matrix is the area in the developing brain which contains neural stem cell population that is situated adjacent to the lateral ventricles as a subependymal layer. SEPC of nonhaemorrhagic origin should prompt to search for neurotropic congenital viral infections, particularly congenital cytomegalovirus and rubella infections. The brain damage due to congenital CMV infection is due to the direct effect of viral replication and indirect effects due to immune mediated brain damage and hypoxic ischemic brain damage secondary to placental insufficiency [5].
Acknowledgement
We acknowledge the parents of this baby who consented for publishing the case report and associated pictures.
Conflict of Interest
No conflicts of interest
References
- Navti O, Hughes BL, Tang JW, Konje J (2016) Comprehensive review and update of cytomegalovirus infection in pregnancy. The Obstetrician& Gynaecologist 18(4): 301-307.
- To M, Kidd M, Maxwell D (2009) Prenatal diagnosis and management of fetal infections. The Obstetrician & Gynaecologist 11(2): 108-116.
- Rennie JM, Hagmann CF, Robertson NJ (2008) Neonatal Cerebral Investigation. (2nd Edn), Cambridge University Press, Cambridge, United Kingdom, p271.
- Gabrielli L, Bonasoni MP, Santini D, Piccirilli G, Chiereghin A, et al. (2012) Congenital cytomegalovirus infection: patterns of fetal brain damage. Clin Microbiol Infect 18(10): E419-E427.
- Teissier N, Fallet-Bianco C, Delezoide AL, Laquerrière A, Marcorelles P et al. (2014) Cytomegalovirus-induced Brain Malformations in Fetuses. J Neuropathol Exp Neurol 73(2): 143-158.
-
Geethanath Ruppa Mohanram, George Aaron. Congenital CMV Infection and Brain Pseudocysts. Glob J of Ped & Neonatol Car. 1(2): 2019. GJPNC.MS.ID.000510.
Congenital CMV Infection, Brain Injury, Pseudocyst, SEPC, CMV infection, viral infections, pseudocysts
-

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.






