 Research Article
Research Article
Analysis and Control of the Inflammatory Immune Response Model
Lakshmi N Sridhar*
Department of Chemical Engineering, University of Puerto Rico, USA
Lakshmi N Sridhar, Department of Chemical Engineering, University of Puerto Rico, Mayaguez, PR 00681, USA
Received Date:October 26, 2025; Published Date:November 10, 2025
Abstract
Inflammation is the body’s way to respond to infection and one needs to develop effective strategies to control the inflammation. In this work, bifurcation analysis and multiobjective nonlinear model predictive control are performed on an inflammatory immune response model. Bifurcation analysis is a powerful mathematical tool used to deal with the nonlinear dynamics of any process. Several factors must be considered, and multiple objectives must be met simultaneously. The MATLAB program MATCONT was used to perform the bifurcation analysis. The MNLMPC calculations were performed using the optimization language PYOMO in conjunction with the state-of-the-art global optimization solvers IPOPT and BARON. The bifurcation analysis revealed the existence of limit and branch points. The MNLMC converged on the Utopia solution (best possible). The limit and branch points (which cause multiple steady-state solutions from a singular point) are very beneficial because they enable the Multiobjective nonlinear model predictive control calculations to converge to the Utopia point (the best possible solution) in the model.
Keywords:Bifurcation; optimization; control; inflammation; immune
Background
Inflammation is one of the most fundamental and ancient biological processes in multicellular organisms, serving as a protective mechanism that the body employs in response to harmful stimuli such as pathogens, damaged cells, or irritants. It represents a complex biological response of vascular tissues to injury or infection, involving immune cells, blood vessels, and molecular mediators that collectively aim to eliminate the initial cause of cell injury, clear out necrotic cells and tissues, and initiate tissue repair. The process of inflammation can be both beneficial and detrimental, depending on its duration, intensity, and regulation. When it is properly controlled and self-limited, inflammation is an essential component of the body’s healing process. However, when dysregulated or chronic, it becomes a pathological condition that underlies many diseases, ranging from arthritis and asthma to atherosclerosis, diabetes, and cancer.
The term inflammation originates from the Latin word inflammatio, meaning “to set on fire,” reflecting the classical signs of redness, heat, swelling, and pain that accompany it. These cardinal features, described centuries ago by Celsus and later expanded upon by Galen and Virchow, remain the clinical hallmarks of inflammation today. The redness and heat result from increased blood flow to the affected area, while swelling is due to the accumulation of fluid and cells in the tissue. Pain arises from the activation of sensory nerve endings by inflammatory mediators, and loss of function often accompanies these symptoms when the local tissue structure is compromised. Inflammation can be broadly categorized into acute and chronic forms. Acute inflammation is the immediate and early response to an injurious agent and is characterized by the exudation of plasma proteins and the emigration of leukocytes, particularly neutrophils. It develops rapidly and is typically shortlived, lasting from a few minutes to a few days. Its main objective is to eradicate the offending agent and set the stage for tissue repair. Chronic inflammation, by contrast, is a prolonged response in which the active inflammation, tissue destruction, and attempts at healing occur simultaneously. It often involves mononuclear cells such as macrophages, lymphocytes, and plasma cells and leads to tissue remodeling or fibrosis if not resolved properly.
The process of acute inflammation begins with the recognition of pathogens or tissue injury by specialized receptors in immune and non-immune cells. Pattern recognition receptors, such as Tolllike receptors, detect conserved microbial structures known as pathogen-associated molecular patterns or endogenous molecules released from damaged cells called danger-associated molecular patterns. Once these receptors are engaged, intracellular signaling cascades are activated, leading to the production of inflammatory mediators such as cytokines, chemokines, and eicosanoids. These molecules act locally to increase vascular permeability, recruit immune cells, and amplify the inflammatory response. Vascular changes are among the earliest events in inflammation. They include vasodilation, which increases blood flow and produces the characteristic redness and heat, and enhanced permeability of the microvasculature, allowing plasma proteins such as antibodies and complement components to enter the tissue. The leakage of protein-rich fluid into the interstitial space results in edema, which contributes to the swelling of inflamed tissue. Endothelial cells lining the blood vessels also express adhesion molecules that facilitate the rolling, adhesion, and transmigration of leukocytes from the bloodstream into the affected tissue.
Once in the tissue, neutrophils are the first responders. They migrate toward the site of infection or injury in response to chemotactic gradients formed by bacterial products, complement fragments, and chemokines. Neutrophils exert their defensive functions through phagocytosis, degranulation, and the generation of reactive oxygen species that kill pathogens. However, the same mechanisms can also damage host tissues if unchecked. As the inflammatory response progresses, monocytes arrive and differentiate into macrophages, which continue the process of phagocytosis, secrete cytokines, and orchestrate the resolution or propagation of inflammation depending on the context. Resolution of inflammation is not a passive process but an active and highly regulated one. It involves the clearance of apoptotic neutrophils by macrophages, the cessation of leukocyte recruitment, and the release of anti-inflammatory mediators such as lipoxins, resolvins, and cytokines like transforming growth factor-beta. These molecules help restore tissue homeostasis, promote repair, and prevent excessive scarring. When resolution fails, the persistent presence of inflammatory stimuli or immune dysregulation can lead to chronic inflammation, characterized by ongoing tissue injury and repair cycles that contribute to disease progression.
Chronic inflammation differs from the acute form not only in duration but also in cellular composition and outcome. Instead of being dominated by neutrophils, it involves macrophages, lymphocytes, and fibroblasts. The continuous activation of macrophages and T cells sustains the production of cytokines and growth factors, which can lead to tissue remodeling and angiogenesis. Over time, fibroblasts proliferate and deposit collagen, resulting in fibrosis that compromises tissue function. This process is seen in conditions like chronic hepatitis, pulmonary fibrosis, and rheumatoid arthritis. In certain cases, chronic inflammation arises not from a persistent infection but from an inappropriate immune response against self-antigens, as seen in autoimmune diseases. In rheumatoid arthritis, for example, the immune system targets synovial tissues, leading to inflammation and joint destruction. Similarly, in inflammatory bowel disease, an abnormal immune response to intestinal microbes causes chronic inflammation of the gut lining. In other cases, environmental factors such as smoking, obesity, or exposure to pollutants can trigger or exacerbate inflammatory pathways, linking lifestyle and chronic disease through systemic inflammation.
At the molecular level, the regulation of inflammation involves a delicate balance between pro-inflammatory and anti-inflammatory signals. Cytokines such as interleukin-1, interleukin-6, and tumor necrosis factor-alpha are potent mediators that amplify the response by recruiting more immune cells and inducing the acute-phase reaction in the liver. Anti-inflammatory cytokines like interleukin-10 counteract these effects, maintaining homeostasis. The transcription factor NF-κB plays a central role in controlling the expression of many inflammatory genes, and its dysregulation has been implicated in numerous chronic inflammatory diseases and cancers. The interplay between inflammation and metabolism is another area of intense study. Adipose tissue, for instance, is not merely a fat store but an active endocrine organ that secretes cytokines known as adipokines. In obesity, the infiltration of macrophages into adipose tissue and the increased production of inflammatory mediators contribute to systemic low-grade inflammation, insulin resistance, and metabolic syndrome. Similarly, inflammation in the vascular endothelium promotes atherosclerosis by facilitating the accumulation of lipids and immune cells within arterial walls, leading to plaque formation and the risk of cardiovascular events.
The nervous system also communicates with the immune system to regulate inflammation. The vagus nerve exerts an antiinflammatory effect through the release of acetylcholine, which inhibits cytokine production in macrophages-a mechanism known as the cholinergic anti-inflammatory pathway. This neuroimmune interaction highlights the integrated nature of bodily systems in controlling inflammation and maintaining balance. While inflammation is indispensable for defense and repair, its chronic or excessive activation has profound pathological consequences. Persistent inflammation generates oxidative stress, DNA damage, and an environment that favors cellular proliferation, angiogenesis, and inhibition of apoptosis, all of which can promote tumor initiation and progression. In the context of cancer, inflammatory cells and mediators contribute to the tumor microenvironment, supporting growth and metastasis. Similarly, neuroinflammation is increasingly recognized as a contributing factor in neurodegenerative diseases such as Alzheimer’s and Parkinson’s, where microglial activation and cytokine release exacerbate neuronal damage.
Therapeutically, controlling inflammation has long been a cornerstone of medical practice. Non-steroidal anti-inflammatory drugs inhibit cyclooxygenase enzymes, reducing the synthesis of prostaglandins that mediate pain and fever. Corticosteroids act at the genomic level to suppress multiple inflammatory pathways. In recent years, targeted biologic therapies have revolutionized the treatment of chronic inflammatory diseases by neutralizing specific cytokines or blocking their receptors. For instance, inhibitors of tumor necrosis factor-alpha, interleukin-6, and Janus kinase pathways have shown efficacy in conditions like rheumatoid arthritis, psoriasis, and inflammatory bowel disease. Lifestyle interventions also play a role in modulating inflammation. Diets rich in fruits, vegetables, and omega-3 fatty acids have anti-inflammatory effects, while sedentary behavior, smoking, and high-fat diets promote inflammation. Regular exercise induces anti-inflammatory cytokines and enhances immune regulation, underscoring the connection between healthy habits and inflammatory control. Moreover, psychological stress and sleep deprivation are known to increase inflammatory mediators, suggesting that mental and physical health are tightly linked through immune pathways.
Research continues to uncover the complexity of inflammation at the systems level, integrating molecular, cellular, and environmental factors. Advances in genomics, proteomics, and metabolomics are providing insights into individual variability in inflammatory responses, paving the way for personalized medicine. Understanding the dual nature of inflammation-as both protector and destroyer-is central to developing strategies that harness its benefits while preventing its harms. Inflammation, therefore, stands at the crossroads of immunity, healing, and disease. It reflects the body’s attempt to preserve integrity against internal and external insults, but when misdirected, it becomes the seed of pathology. The story of inflammation is thus one of balance-between defense and destruction, between repair and fibrosis, between survival and degeneration. The challenge for modern medicine lies in deciphering and controlling this balance, ensuring that the fire of inflammation remains a healing force rather than a source of harm.
Bone [1] researched the systemic inflammatory response syndrome (SIRS) and the multiple organ dysfunction syndrome (MODS). Volk, et al. [2], discussed the Clinical aspects of systemic inflammation and ’immunoparalysis’. Annane, et al. [3] discussed the effect of treatment with low doses of hydrocortisone and fludrocortisone on mortality in patients with septic shock. Cross and Opal [4] developed a new paradigm for the treatment of sepsis considering combination therapy. Kumar, et al. [5] discussed the dynamics of acute inflammation. Chow, et al. [6], investigated the acute inflammatory response in diverse shock states. Day, et al. [7] developed a reduced mathematical model of the acute inflammatory response capturing scenarios of repeated endotoxin administration. Reynolds, et al. [8], developed a reduced mathematical model of the acute inflammatory response. Vodovotz, et al. [9] investigated in silico models of acute inflammation in animals. Daun, et al. [10] discussed an ensemble of models of the acute inflammatory response to bacterial lipopolysaccharide in rats. Florian, et al. [11] developed nonlinear model predictive control strategies for dosing daily anticancer agents using a novel saturating-rate cell-cycle model, Day, et al. [12], used nonlinear model predictive control to find optimal therapeutic strategies to modulate inflammation.
In this work, bifurcation analysis and multiobjective nonlinear model predictive control is performed on the inflammation model described in Day, et al. [12]. The paper is organized as follows. First, the model equations are presented, followed by a discussion of the numerical techniques involving bifurcation analysis and multiobjective nonlinear model predictive control (MNLMPC). The results and discussion are then presented, followed by the conclusions.
Model Equations (Day, et al. (2010)[12]
The variables pv, nv, dv, and cv represent the bacterial pathogen population, the pro-inflammatory cytokines, tissue damage, and the anti-inflammatory mediators. up and ua are the control parameters, which are the pro-inflammatory and the anti-inflammatory therapy of the system.
The model equations are


The base parameter values are
kpm=0.6; kmp=0.01; sm=0.005; kpg=1;pinf=20e+06;
kpn=1.8;knp=0.1;knn=0.01;snr=0.08;
knd=0.35; xdn=0.06; cinf=0.28;sc=0.0125;kcn=0.04;kcnd=48;
up=0; ua=0;
μnr = 0.12;μn = 0.05;μc = 0.1;μd = 0.02;μm = 0.002.
Bifurcation analysis
Bifurcation analysis is performed using the MATLAB software MATCONT which locates branch points limit points and Hopf bifurcation points (Dhooge Govearts, and Kuznetsov [13]; Dhooge Govearts, Kuznetsov, Mestrom and Riet, [14]). Consider a set of ordinary differential equations

x∈Rn with a bifurcation parameter beα . Since the gradient
is orthogonal to the tangent vector,
The tangent z=[z1, z2, z3, z4 ,.... zn+1 ]
must satisfy
Az = 0 (4)
A is given by
A = [∂f / ∂x | ∂f / ∂α ]
where ∂f / ∂x is the Jacobian matrix. For both limit and branch
points, the matrix [∂f / ∂x] must be singular. The n+1 th component
of the tangent vector z n+1 = 0 for a limit point (LP)and for a branch
point (BP) the matrix 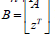 must be singular. At a Hopf bifurcation
point,
must be singular. At a Hopf bifurcation
point,

@ indicates the bialternate product and In is the n-square identity matrix. Hopf bifurcations cause limit cycles and should be eliminated because limit cycles make optimization and control tasks very difficult. More details can be found in Kuznetsov [15,16] and Govaerts [17].
Multiobjective Nonlinear Model Predictive Control (MNLMPC)
The procedure developed by Flores Tlacuahuaz, et al. [18] is
used for performing the MNLMPC calculations Let the objective
function variables
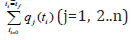 (j=1, 2..n) for a problem involving a set
of ODE
(j=1, 2..n) for a problem involving a set
of ODE

Where tf is the final time value, and n the total number of
objective variables and u the control parameter. First, the single
objective optimal control problem independently and individually
optimizing each of the variables
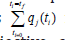 is solved. Leading to the
values q*j
. Then the multiobjective optimal control (MOOC)
optimization problem that will be solved is
is solved. Leading to the
values q*j
. Then the multiobjective optimal control (MOOC)
optimization problem that will be solved is

This will provide the values of u at various times. The first
obtained control value of u is implemented and the rest are
discarded. This procedure is repeated until the implemented and
the first obtained control values are the same or if the Utopia point
where 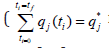 for all j) is obtained.
for all j) is obtained.
Pyomo [19] is used for these calculations. Here, the differential equations are converted to a Nonlinear Program (NLP) using the orthogonal collocation method The NLP is solved using IPOPT [20] and confirmed as a global solution with BARON [21]. Sridhar [22] proved that the MNLMPC calculations to converge to the Utopia solution when the bifurcation analysis revealed the presence of limit and branch points . This was done by imposing the singularity condition on the co-state equation [23]. This makes the constrained problem an unconstrained optimization problem, and the only solution is the Utopia solution. More details can be found in Sridhar [23].
Results
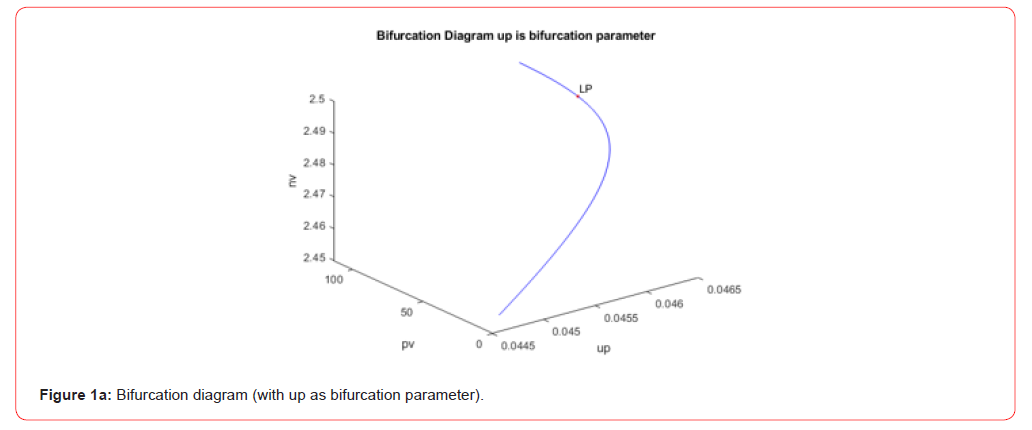
When up was the bifurcation parameter, a limit point was found
at (pv,nv,dv,cv,up) values ((75.123799, 2.493135, 50, 0.524251,
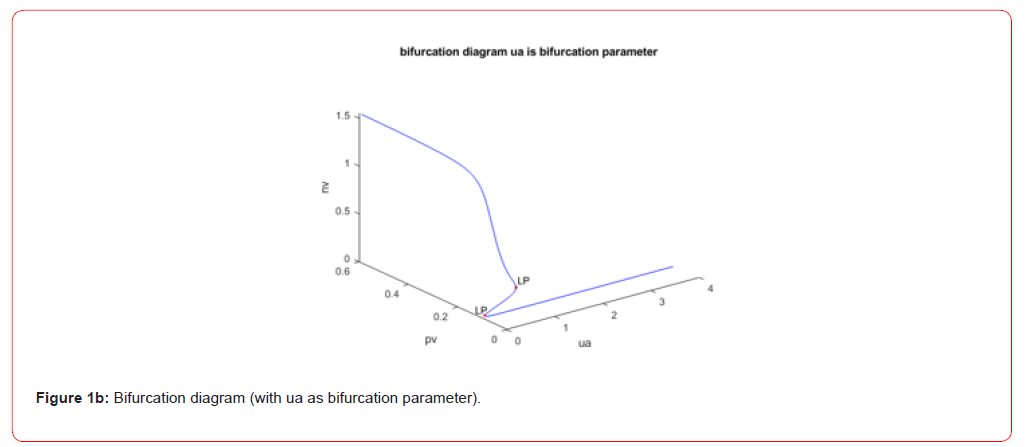
0.046348) (Figure 1a). When ua is bifurcation parameter, two limit
points were found at (pv,nv,dv,cv,ua) values of((0.100172, 0.226022,
40.042712, 7.458674, 0.704163); (0.10125, 0.019855, 0.007619,
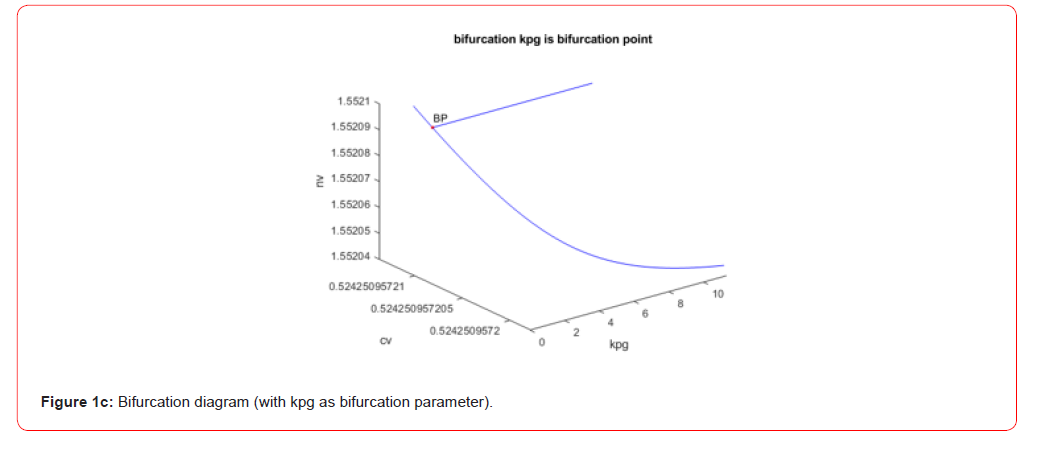
0.772682, 0.063055 ) (Figure 1b). When kpg was the bifurcation
parameter, a branch point was located at (pv,nv,dv,cv,kpg) values
of ( 0.000000, 1.552090, 49.999999, 0.524251, 2.120064 ) (Figure
1c). For the MNLMPC ua and up are the control parameters, and
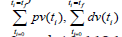 were minimized individually, and led to values of
0 and 10.6686069466. The overall optimal control problem will
involve the minimization of
were minimized individually, and led to values of
0 and 10.6686069466. The overall optimal control problem will
involve the minimization of
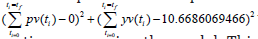 − was
minimized subject to the equations governing the model. This led
to a value of zero (the Utopia point). The MNLMPC values of the
control variables, ua and up were 1.504 and 1.472 . The MNLMPC
profiles are shown in Figure 2a-2c. The control profiles of ua and
up exhibited noise and this was remedied using the Savitzky-Golay
filter to produce the smooth profiles uasg;upsg .
− was
minimized subject to the equations governing the model. This led
to a value of zero (the Utopia point). The MNLMPC values of the
control variables, ua and up were 1.504 and 1.472 . The MNLMPC
profiles are shown in Figure 2a-2c. The control profiles of ua and
up exhibited noise and this was remedied using the Savitzky-Golay
filter to produce the smooth profiles uasg;upsg .
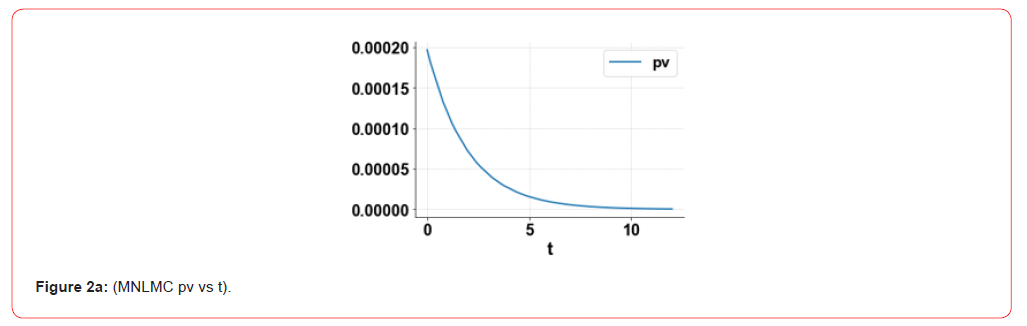
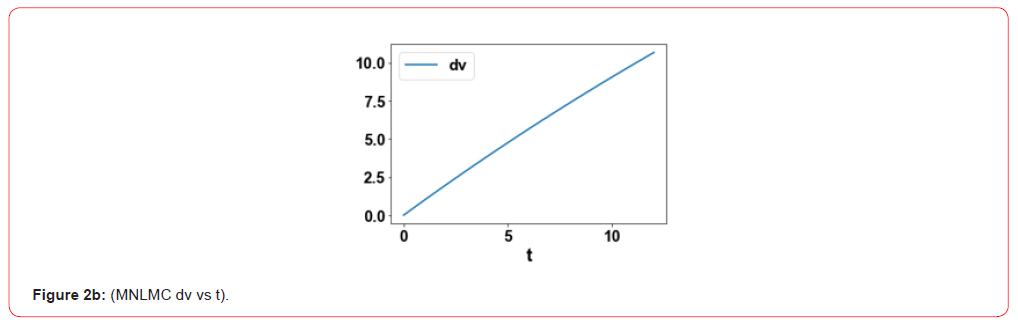
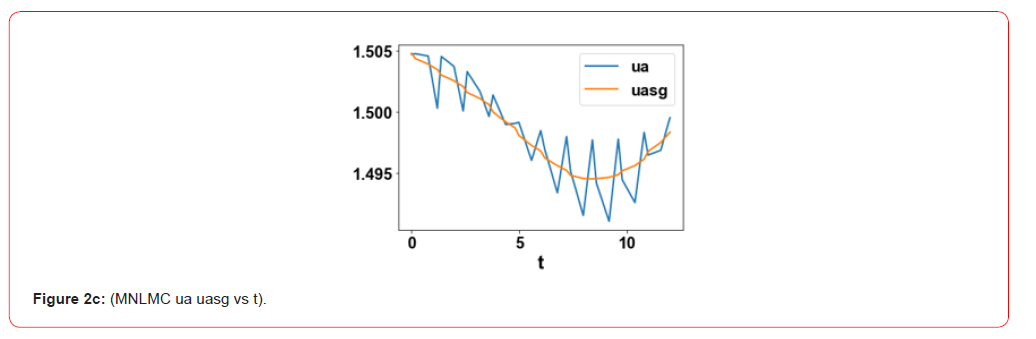
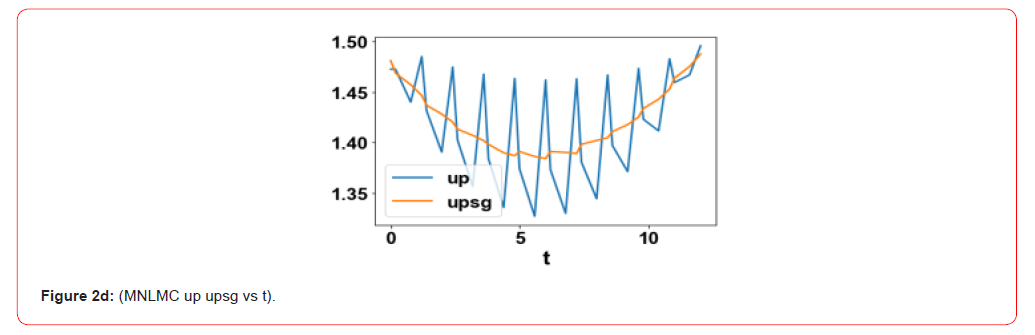
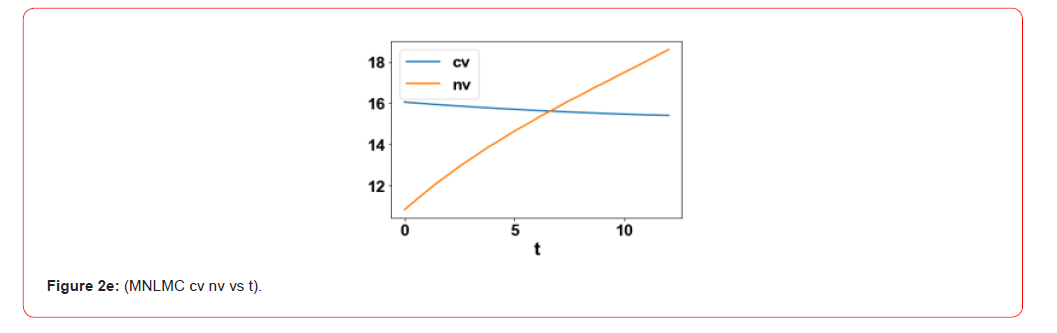
Discussion of Results
Theorem
If one of the functions in a dynamic system is separable into two distinct functions, a branch point singularity will occur in the system.
Proof
Consider a system of equations

The tangent at any point x; z=[z1, z2, z3, z4 ,.... zn+1 ] must satisfy
Az = 0
The matrix 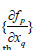 must be singular at both limit and branch
points. The n+1 th component of the tangent vector zn +1 = 0 at a
limit point (LP) and for a branch point (BP) the matrix
must be singular at both limit and branch
points. The n+1 th component of the tangent vector zn +1 = 0 at a
limit point (LP) and for a branch point (BP) the matrix
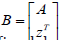 must be singular. Any tangent at a point y that is defined by
z=[z1, z2, z3, z4 ,.... zn+1 ] must satisfy
must be singular. Any tangent at a point y that is defined by
z=[z1, z2, z3, z4 ,.... zn+1 ] must satisfy
Az = 0
For a branch point, there must exist two tangents at the singularity. Let the two tangents be z and w. This implies that
Az = 0
Aw = 0
Consider a vector v that is orthogonal to one of the tangents (say z). v can be expressed as a linear combination of z and w ( v =α z +β w). Since Az = Aw = 0 ; Av = 0 and since z and v are orthogonal,
zT v = 0 . Hence 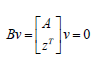 which implies that B is singular
where
which implies that B is singular
where 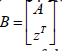
Let any of the functions fi are separable into 2 functions φ1 ,φ2 as
fi =φ1φ2
At steady-state fi( x, α) =0 and this will imply that either φ1 = 0 or φ2 =0 or both φ1 and φ2 must be 0. This implies that two branches φ1 = 0 and φ2 =0 will meet at a point where both φ1 and φ2 are 0.
At this point, the matrix B will be singular as a row in this matrix would be


This implies that every element in the row 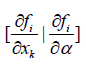 would be 0, and hence the matrix B would be singular. The singularity in B
implies that there exists a branch point.
would be 0, and hence the matrix B would be singular. The singularity in B
implies that there exists a branch point.
The branch point occurred at (pv,nv,dv,cv,kpg) values of ( 0, 1.55209, 49.999999, 0.524251, 2.120064 ) Here, the two distinct functions can be obtained from the first ODE in the model .

Since, pv=0, nv=1.55209. kpg=2.120064, kpm=0.6, kmp=0.01, sm=0.005, μm =0.002, cv=0.524251, cinf=0.28, both distinct equations are satisfied, validating the theorem.
The presence of the limit point and is beneficial because it allows the MNLMPC calculations to attain the Utopia solution, validating the analysis of Sridhar(2024).
Conclusions
Bifurcation analysis and multiobjective nonlinear control (MNLMPC) studies on an inflammatory immune response model. The bifurcation analysis revealed the existence of limit and branch points . The limit and branch points (which cause multiple steadystate solutions from a singular point) are very beneficial because they enable the Multiobjective nonlinear model predictive control calculations to converge to the Utopia point (the best possible solution) in the models. A combination of bifurcation analysis and Multiobjective Nonlinear Model Predictive Control(MNLMPC) for an inflammatory immune response model is the main contribution of this paper.
Data Availability Statement
All data used is presented in the paper
Conflict of Interest
The author, Dr. Lakshmi N Sridhar has no conflict of interest.
Acknowledgement
Dr. Sridhar thanks Dr. Carlos Ramirez and Dr. Suleiman for encouraging him to write single-author papers
References
- Bone RC (1996) Immunologic dissonance: A continuing evolution in our understanding of the systemic inflammatory response syndrome (SIRS) and the multiple organ dysfunction syndrome (MODS). Ann Intern Med 125: 680-687.
- Volk HD, Reinke P, Docke WD (2000) Clinical aspects: From systemic inflammation to ‘immunoparalysis’. Chem Immunol 74: 162-177.
- Annane D, Sebille V, Charpentier C, Bollaert PE, Francois B, et al. (2002) Effect of treatment with low doses of hydrocortisone and fludrocortisone on mortality in patients with septic shock. JAMA 288: 862–871.
- Cross AS, Opal SM (2003) A new paradigm for the treatment of sepsis: Is it time to consider combination therapy. Ann Intern Med 138: 502-505.
- Kumar R, Clermont G, Vodovotz Y, Chow CC (2004) The dynamics of acute inflammation. J Theor Biol 230: 145–155.
- Chow CC, Clermont G, Kumar R, Lagoa C, Tawadrous Z, et al. (2005) The acute inflammatory response in diverse shock states. Shock 24: 74-84.
- Day J, Rubin J, Vodovotz Y, Chow CC, Reynolds A, et al. (2006) A reduced mathematical model of the acute inflammatory response II. Capturing scenarios of repeated endo-toxin administration. J Theor Biol 242: 237-256.
- Reynolds AM, Rubin J, Clermont G, Day J, Vodovotz Y et al. (2006) A reduced mathematical model of the acute inflammatory response: I Derivation of the model and analysis of anti-inflammation. J Theor Biol 242: 220–236.
- Vodovotz Y, Chow CC, Bartels J, Lagoa C, Prince JM, et al. (2006) In silico models of acute inflammation in animals. Shock 26: 235–244.
- Daun S, Rubin J, Vodovotz YV, Roy A, Parker RS, et al. (2008) An ensemble of models of the acute inflammatory response to bacterial lipopolysaccharide in rats: Results from parameter space reduction. J Theor Biol 253: 843-853.
- Florian Jr, JL Eiseman JA, RS Parker (2008) Nonlinear model predictive control for dosing daily anticancer agents using a novel saturating-rate cell-cycle model. Comput Biol Med 38 339–347.
- Day Judy, Jonathan Rubin, Gilles Clermont (2010) Using nonlinear model predictive control to find optimal therapeutic strategies to modulate inflammation. Mathematical Biosciences and Engineering 7(4): 739-763.
- Dhooge A, Govearts W, Kuznetsov AY (2003) MATCONT: A Matlab package for numerical bifurcation analysis of ODEs”. ACM transactions on Mathematical software 29(2): 141-164.
- Dhooge A, Govaerts W, Kuznetsov YA, Mestrom W, Riet AM (2004) Cl_matcont: a continuation toolbox in Matlab. Conferences 161-166.
- Kuznetsov YA (1998) Elements of applied bifurcation theory. Springer,
- Kuznetsov YA (2009) Five lectures on numerical bifurcation analysis, Utrecht University, NL.
- Govaerts WJF (2000) Numerical Methods for Bifurcations of Dynamical Equilibria. SIAM.
- Flores-Tlacuahuac A, Pilar Morales, Martin Riveral Toledo (2012) Multiobjective Nonlinear model predictive control of a class of chemical reactors. I & EC research 5891-5899.
- Hart William E, Carl D Laird, Jean-Paul Watson, David L Woodruff, Gabriel A Hackebeil, et al. Pyomo – Optimization Modeling in Python Second Edition. Vol. 67.
- Wächter A, Biegler L (2006) On the implementation of an interior-point filter line-search algorithm for large-scale nonlinear programming. Math Program 106: 25–57.
- Tawarmalani M, Sahinidis NV (2005) A polyhedral branch-and-cut approach to global optimization. Mathematical Programming 103(2): 225-249.
- Sridhar LN (2024) Coupling Bifurcation Analysis and Multiobjective Nonlinear Model Predictive Control. Austin Chem Eng 10(3): 1107.
- Upreti, Simant Ranjan (2013) Optimal control for chemical engineers. Taylor and Francis.
-
Lakshmi N Sridhar*. Analysis and Control of the Inflammatory Immune Response Model. Arch Biomed Eng & Biotechnol. 6(3): 2025. ABEB.MS.ID.000686.
-
Bifurcation; optimization; control; inflammation; immune
-

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.






