 Editorial
Editorial
The Importance of Statistical Knowledge and Biostatistical Interpretation of Scientific Manuscripts for the Healthcare Team
Marcel Marcano Lozada*
Aggregate Professor Microbiology Department, Medicina Faculty, Universidad Central de Venezuela and Medical Microbiologist at Angios Vascular Center / Wound Clinic, Caracas, Venezuela
Marcel Marcano-Lozada, Aggregate Professor Microbiology Department, “J.M. Vargas” School of Medicine, Universidad Central de Venezuela, Caracas, Venezuela, email: marcelmarcano@gmail.com
Received Date: December 18, 2018; Published Date: December 21, 2018
Editorial
One question comes to my memory when I’m thinking in the importance of know and understand the signification of the statistical analysis when a scientific manuscript is reading, and this question comes from many of my students… “Professor we´re doctors (or nurses, technicians, biomedical engineers, microbiologists, etc.), our focus is the patient, so, why we need to dedicate time to learn about statistical analysis and interpretation of it?, we´re so busy to take another task”.
As a physician and based on the need to get a clear and simply answer to all my colleagues and students, I use practical examples from the real-life setting and clinical and microbiological experience to let them know why statistical knowledge is pretty important for the healthcare personnel. The most reticent group to learn about biostatistics are physicians; yes, the doctors thinks that numbers, proofs, values and measures aren’t necessaries to their daily duties, and the opposite, represents a waste of time; but the opposite is that nothing is so far from the truth. When we take the decision to use a “gold standard technique” despite other alternatives, when we choose a medication dose, when we prefer use an antibiotic over other, we’re applying biostatistical! all the clinical trials to bring us the knowledge about “superiority”, “equivalence! or “nor inferiority” passed by the statistical control to ensure that the results are not related to the random serendipity. Moreover, the clinical trials design involves biostatistics since their conception, for samples size calculation, required participants to validate or deny hypothesis, etc. Is really important than healthcare team understood the precious value of “statistical significance” and have the capacity to use it along with the concept of “clinical significance”, because many members of our healthcare teams only knows the basic concept of “p value <0,05” and understand it as a true value that ensure positive connotation to the concept endorse to it (drug, technique, etc.). I like to use a very simple example to explain the previous point, considering a drug, a clinical outcome measure as blood pressure reduction with its “p value”, the statistical and clinical significances of it, with the final conclusion of physician’s behavior and adoption of the treatment, so please let me use the following table to explain it (Table 1):
Table 1: Drug Pressure Statistical and Clinical Significance.
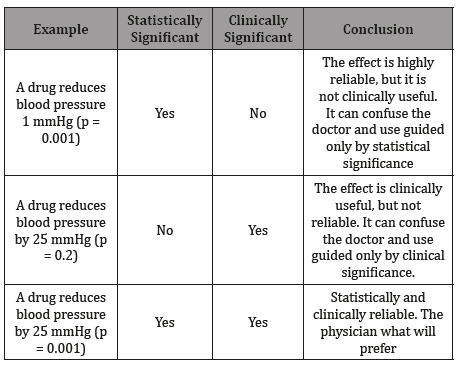
In the first situation (1), the drug reduces in a minimum value the blood pressure measures, but with a great statistical significance, so, if the responsible to analyze this information only looks for a statistical significance value under 0,05, maybe made a wrong selection of the drug, because in this matter, statistical significance doesn’t go paired with the clinical significance, it means that in the real-life, the drug fails to reduce the blood pressure levels in the populations of hypertensive patients. In situation (2), the amount of the reduction of blood pressure levels is important (25 mmHg), and clinically according to the pointed in many treatment guidelines, but, the statistical analysis of it doesn’t support its use in the real life (p > 0,05), because failures in the control of blood pressure figures may occur in many hypertensive patients.
The third situation (3), shows the ideal scenario, when statistical (p < 0,05) & clinical significances (25 mmHg reduction in blood pressure values) goes together, and it means that the results of reduction in blood pressure levels due to the use of the drug could be extrapolated to the real-life setting and the drug achieves the benefit of blood pressure reduction (and its concomitant risks) in the hypertensive patient population. When the healthcare team needs to analyze a scientific information comes from a clinical trial publication (for example, in the committees related to infection control, budget, etc.), many times the unknowledge of the population-based analysis could lead to wrong conclusions, so, I try to explain the importance of understood this particular matter to gain access to a strong conclusion that leads to the greatest benefits for the patients. Is here when 3 letters make a huge importance, the ITT & PPP analysis of the results, so, I want to use some highlights to understand it and their real significance for healthcare decisions. Let me use 2 tables to show it (Table 2):
Table 2: (a)Drug related mortality, (b)IIT and PPP interpretation & conclusion.
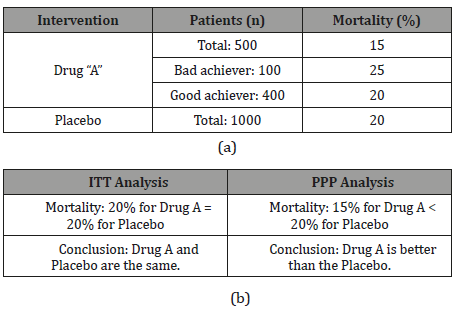
In the example, the selection of the population analysis method for the results leads to different and opposite conclusions. The Intention-To-Treat (ITT) analysis includes all patients from the groups in which they were randomly assigned (met or not the entry criteria, independent of the treatment they actually received, or if they abandoned the treatment or deviated from the protocol). Advantages of ITT analysis are a more realistic estimate of clinical practice, with less bias in the real magnitude of the effect, it admits the existence of non-ideal situations and considers the loss of patients after randomization. This is the type of analysis required in studies that seek to demonstrate superiority.
The analysis of Population-Per-Protocol (PPP) includes the patients that comply with the inclusion criteria and none of the exclusion criteria (they have received a minimum % of the drug in research and have not suffered any deviation greater than the protocol). PPP analysis maximizes the possibility of showing that the new treatment is effective, admits the existence of ideal situations and doesn’t consider the loss of patients after randomization. It is the type of analysis required for clinical trials that seek to demonstrate non-inferiority.
Based on the previous considerations, and applying to the example table, we can understand the importance of selection of the right type of statistical analysis to gain a correct interpretation of the results of the trial and to produce the adequate conclusion. If the ITT analysis was applied, no differences among use of Drug A and Placebo exists, so, no advantages of the Drug over the inactive substance were show, or, Drug A doesn’t represent any benefit for the patient. If PPP analysis is use, a reduction in mortality were observed compared to Placebo, so, Drug A is an option to treat patients and reduce mortality.
The primary end-point of the clinical trial were related with the statistical population analysis type, in the present days, the majority of clinical trials are design to demonstrate non-inferiority, so, the PPP analysis is the ideal for it, but, in some cases, the combine use of ITT & PPP analysis can improve the strength of the conclusions when both results are similar. So, is important in the lecture and interpretation of a scientific manuscript look the relation among end-points and statistical population analysis type to generate a conclusion that really fits with the real-life setting (the wrong selection of the type of statistical analysis of the population, can lead in real-life to the presentation of therapeutic faults of control of the disease, based on a wrong conclusion presented in the scientific publication).
From my humbly and different optics and positions as a clinical physician, laboratory professional (medical microbiologist), professor and researcher, I hope that these 2 examples of the importance of the statistical significance analysis (related to the clinical significance) and the right choose of the statistical analysis of the population, demonstrating the need of the healthcare team members having basic biostatistical knowledge to make its own conclusion based on the review of the published information in the scientific literature, and take the best decisions.
Acknowledgement
None
Conflict of Interest
No conflict of interest.
-
Marcel Marcano Lozada. The Importance of Statistical Knowledge and Biostatistical Interpretation of Scientific Manuscripts for the Healthcare Team. Annal Biostat & Biomed Appli. 1(3): 2018. ABBA.MS.ID.000511.
Statistical knowledge, Biostatistical, Scientific manuscripts, Healthcare team, Microbiology, Biomedical engineers, Microbiologists, Drug.
-

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.






