 Research Article
Research Article
Prevalence of Dyslipidemia among Women with Polycystic Ovary Syndrome Based on Body Mass Index
Amal AL Mulla1*, Ahmed EL Sokkary1, Sherif Ekladious1, Amar Hassan Khamis2
1Dubai Health Authority, United Arab Emirates, UAE
2Hamdan Bin Mohammed College of Dental Medicine, Mohammed Bin Rashid University of Medicine and Health Sciences, United Arab Emirates, UAE
Amal AL Mulla, Consultant OBSGYN & Reproductive Medicine, Dubai Health Authority. Street 5B, Al Mamzar, PO Box 88551, Dubai, United Arab Emirates, UAE.
Received Date: June 05, 2020; Published Date: June 12, 2020
Abstract
Objective: The purpose of this study was to detect the prevalence of dyslipidemia among polycystic ovary syndrome (PCOS) women with different BMIs.
Study design: We conducted a cross-sectional study whereby one hundred and fifteen women diagnosed with PCOS were recruited from a fertility clinic in a 6-month period. All participants had their weight and height measured and body mass index (BMI) calculated.
Thereafter, they were divided into two groups: normal BMI (BMI ≤25 kg/m2) and high BMI (BMI >25 kg/m2). Fasting blood samples were obtained and total cholesterol, triglycerides, LDL-C, HDL-C, glucose, and insulin levels were measured in both groups. Insulin resistance was calculated using the homeostasis model assessment of insulin resistance (HOMA-IR) equation. Differences in these variables were assessed using the Chi-square test and Fisher’s exact test.
Results: No statistically significant differences were found between both groups of women in terms of total cholesterol, triglycerides, LDL-C, and HDL-C levels. Insulin resistance was significantly more prevalent in the high BMI (78.3%) than in the normal BMI group (59.1%) (P < 0.05).
Conclusions: Dyslipidemia in women with PCOS is not related to their BMI. Early detection and treatment of dyslipidemia in this group of women will protect them from cardiovascular diseases.
Keywords: BMI; Dyslipidemia; Polycystic ovary syndrome; Cardiovascular risk; Atherosclerosis; Dubai
Introduction
Polycystic ovary syndrome (PCOS) is an endocrinological disturbance characterized by menstrual irregularities, androgenic manifestations, and a polycystic morphological appearance of one or both ovaries. It affects 5 to 20% of women of reproductive age [1].
Furthermore, PCOS encompasses a variety of cardiovascular risk factors such as hypertension, dyslipidemia, and hyperglycemia, all of which are characteristics of the so- called “metabolic syndrome.”
The cause of PCOS in not yet clear. However, insulin resistance seems to play a major role in its pathogenesis. Insulin is an essential orchestrator of fat, carbohydrate, and protein metabolism; reduced sensitivity to its effects results in major metabolic disturbances such as defective intracellular glucose transport, lipotoxicity, and ultimately, impaired energy production [2]. In this setting, adiposities retain their sensitivity to insulin action; therefore, lipid deposition continues and results in increased weight gain, which in turn worsens insulin resistance [3].
Impairments in the antioxidant system and accumulation of free radicals could be the spark that triggers inflammation, which in turn result in a plethora of pathogenic phenomena, such as PCOS and dyslipidemia [4].
Dyslipidemia in women with PCOS is a source of concern given its negative effects on cardiovascular health. In the general population, most epidemiological studies show a steady increase in cardiovascular disease when cholesterol levels exceed 5mmol/L [5]. The pathogenesis of dyslipidemia in PCOS has not been fully elucidated; over production of free radicals, defects in lipolysis, glucose receptor abnormalities, insulin resistance, and obesity have all been proposed as possible mechanisms [6].
In the current study, we aimed to evaluate cholesterol and lipoprotein levels among women with PCOS and assessed their relation to body mass index (BMI).
Materials and Methods
Subjects
Women included in this cross-sectional study were randomly selected during their visits to the fertility clinic in Saudi German Hospital in Dubai between August 2018 and February 2019. Participants were included if they had PCOS diagnosed according to Rotterdam criteria [7] and an age between 19 and 42 years. Women with concurrent hypothyroidism, hyperprolactinemia, or hyperandrogenism due to causes other than PCOS were excluded from the study.
The weight and height of each participant were measured (in kilograms and centimeters, respectively), and the participants were categorized into two groups according to their BMI: normal BMI (BMI ≤ 25 kg/m2) and high BMI (BMI > 25 kg/m2).
Blood samples were collected after a 12-hour fast. Total cholesterol, LDL-C, triglycerides, HDL-C, glucose, and insulin were measured.
Cardiovascular risk factors such as hypertension, diabetes mellitus, family history of dyslipidemia, and smoking status were assessed using a questionnaire filled out by the participants.
Laboratory assays
Venous blood samples were centrifuged, and sera were used to assess total cholesterol, LDL-C, and triglycerides using Adevia 1650 biochemical analyzer (Erba Lachema reagents). HDL-C was determined via direct immune-inhibition method. Insulin level was detected using electro-Chemiluminescence immunoassay (ECLIA) (Cobas E, Roche Diagnostics, and Mannheim, Germany). Hexokinase method (Cobas C, Roche Diagnostics GmbH, and Mannheim, Germany) was used to calculate the glucose level. Insulin resistance was calculated using the homeostasis model assessment of insulin resistance (HOMA-IR) equation.
Variable classification
Cholesterol and lipoprotein reference values were based on the American College of Cardiology and American Heart Association recommendations [8]. Abnormal blood levels were considered as follows: total cholesterol > 6.2mmol/L, HDL-C < 1.3mmol/L, LDL-C > 4.1mmol/L, and triglycerides > 2.2mmol/L. Insulin resistance was defined if HOMA-IR exceeded 1.9.
Data analysis
All data were analyzed using IBM-SPSS for Windows version 23.0 (SPSS Inc., Chicago, IL). Categorical variables were crosstabulated to examine independence between variables; for such variables, the Chi-squared test or Fisher’s exact test was used. Frequency tables bar and pie graphs, measures of percentage, tendency, and dispersion were used as descriptive tools. The Mann- Whitney test was used to compare the ages between both groups. A P-value of less than 0.05 was considered significant in all statistical analysis.
Ethical approval
Written informed consent was obtained from all the participants, and the study was approved by the Research Ethics Review Committee of Dubai Health Authority (approval number: DSREC-09/2018_12). The study was carried out in accordance with the code of ethics of the Declaration of Helsinki.
Results
A total of 115 women were included. Thirty-two of them were Emirati (27.8%), and eighty-three (72.2%) were of other nationalities. Forty-five women were grouped as normal BMI, and seventy as high BMI (Figure 1). The average BMI was 27.4kg/m2 (±5.7).
No age difference was observed between both groups.
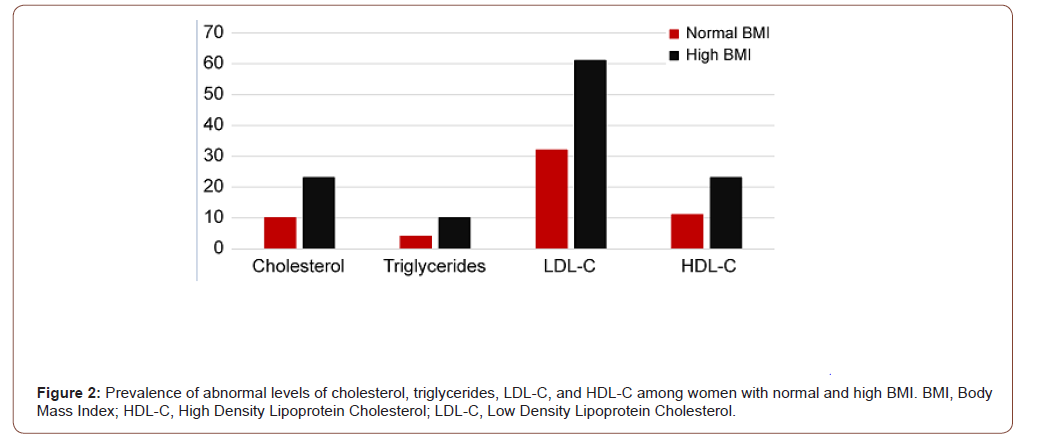
No statistically significant differences were observed between women of normal and high BMI groups concerning elevated values of total cholesterol, triglycerides, and LDL-C, or low values of HDL-C (Figure 2). On the contrary, insulin resistance and diabetes mellitus were significantly more frequent among women in the high BMI group. Both groups showed a similar prevalence of hypertension and family history of dyslipidemia (Table 1). All participants denied current or past smoking.
Table 1: Cardiovascular risk factor distribution among participants.
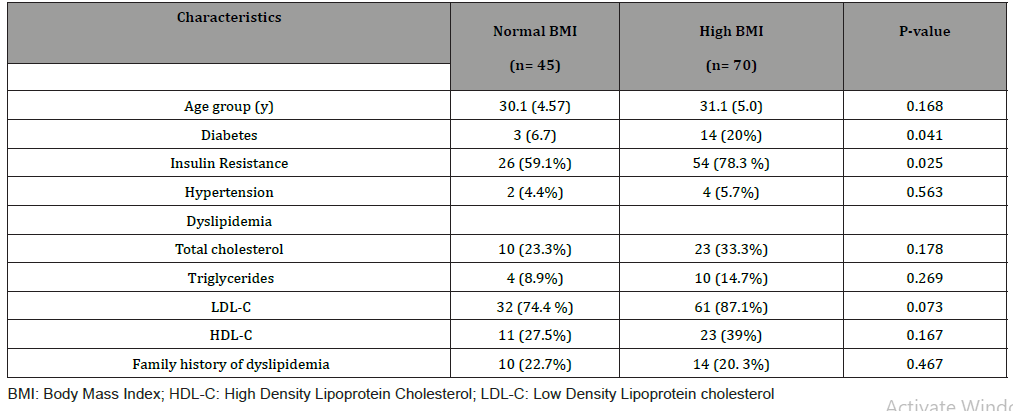
In the normal BMI group, elevated LDL-C levels were more prevalent in women with insulin resistance than in those without insulin resistance (83.8% vs. 52%; p < .05). This relationship was absent in the high BMI group.
Discussion
In the present study, we investigated the prevalence of dyslipidemia in women with PCOS and its relation to BMI. Abnormalities in lipid profiles were not limited to women with an elevated BMI, as they were also present in women with a normal BMI.
Similar findings were found in a study performed by Kim, et al. which reported dyslipidemia among 35.7% of young and lean Korean women with PCOS [9]. Baldani, et al. attributed the metabolic disorders in PCOS to intrinsic factors, as these were also noticed in lean patients [10]. Sam, et al. reported that PCOS was the most frequent cause of dyslipidemia in women of reproductive age [11].
Therefore, women with PCOS were at notable risk for subclinical atherosclerosis [12], irrespective of their BMI. Moreover, cardiovascular risk was reported to be elevated even in women with normal lipid profiles [13]. This relates to an altered composition of LDL-C particles that renders them more atherogenic regardless of its quantitative value [14]. These qualitative abnormalities further aggravate the risk of coronary artery disease [15]; indeed, coronary artery calcification is more common in women with PCOS than in controls without PCOS, irrespective of age or BMI [16].
Another important and often overlooked point is that the toxic effect of dyslipidemia leads to oocytes development arrest [17]. In this regard, Li, et al. reported a statistically significant negative impact of dyslipidemia on the number of oocytes retrieved during assisted reproductive techniques and on clinical pregnancy rate [18].
The main limitations of our study include the small sample size and the bias inherent to its cross-sectional design. Selection bias was a concern, given that all patients were enrolled from only one institution. The results cannot be generalized and need to be confirmed by larger studies.
Conclusions
Our findings confirm that, irrespective of BMI, all women with PCOS are at risk for dyslipidemia. In accordance with the recommendations of the American College of Obstetricians and Gynecologists [19], we consider that dyslipidemia screening should be offered to every woman with PCOS, given that early diagnosis and management can prevent atherosclerosis and its serious consequences. We can think of PCOS in young women as a quiescent bomb that can explode later in the course of their lives, causing serious complications. Dyslipidemia in this group of patients should be thoroughly evaluated irrespective of BMI, in order to prevent cardiovascular disease, minimize squeal, and thus allow maximum productivity in the society.
Acknowledgement
None.
Conflict of Interest
Authors declare no conflict of interest.
References
- Azziz R, Carmina E, Chen Z, Dunaif A, Laven JSE, et al. (2016) Polycystic ovary syndrome. Nat Rev Dis Primers 2: 16057.
- Dali-Youcef N, Mecili M, Ricci R, Andrès E (2013) Metabolic inflammation: connecting obesity and insulin resistance. Ann Med 45(3): 242-253.
- Isganaitis E, Lustig RH (2005) Fast food, central nervous system insulin resistance, and obesity. Arterioscler Thromb Vasc Biol 25(12): 2451-2462.
- Panti AA, Shehu CS, Saidu Y, Tunau KA, Nwobodo EI, et al. (2018) Oxidative stress and outcome of antioxidant supplementation in patients with polycystic ovarian syndrome (PCOS). Int J Reprod Contracept Obstet Gynecol 7: 1667-1672.
- Vikulova DN, Grubisic M, Zhao Y, Lynch K, Humphries KH, et al. (2019) Premature Atherosclerotic Cardiovascular Disease: Trends in Incidence, Risk Factors, and Sex‐Related Differences, 2000 to 2016. J Am Heart Assoc 8(14): e012178.
- Carmina E (2009) Cardiovascular risk and events in polycystic ovary syndrome. Climacteric 12 Suppl 1: 22-25.
- Rotterdam ESHRE/ASRM-Sponsored PCOS Consensus Workshop Group (2004) Revised 2003 consensus on diagnostic criteria and long-term health risks related to polycystic ovary syndrome. Hum Reprod 19(1): 41-47.
- Grundy SM, Stone NJ, Bailey AL, Beam C, Birtcher KK, et al. (2018) AHA/ACC/AACVPR/AAPA/ABC/ ACPM/ADA/AGS/APhA/ASPC/NLA/PCNA Guideline on the Management of Blood Cholesterol: Executive Summary: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Circulation 139(25): e1046-e1081.
- Kim JJ, Choi YM (2013) Dyslipidemia in women with polycystic ovary syndrome. Obstet Gynecol Sci 56(3): 137-142.
- Baldani DP, Skrgatic L, Ougouag R (2015) Polycystic Ovary Syndrome: Important Underrecognised Cardiometabolic Risk Factor in Reproductive-Age Women. Int J Endocrinol 2015: 786362.
- Sam S, Dunaif A (2003) Polycystic ovary syndrome: syndrome XX? Trends Endocrinol Metabol 14(8): 365-370.
- Orio Jr F, Palomba S, Cascella T, De Simone B, Di Biase S, et al. (2004) Early impairment of endothelial structure and function in young normal-weight women with polycystic ovary syndrome. J Clin Endocrinol Metab 89(9): 4588-4593.
- Berneis K, Rizzo M, Hersberger M, Rini GB, Pepe I, et al. (2009) Atherogenic forms of dyslipidaemia in women with polycystic ovary syndrome. Int J Clin Pract 63(1): 56-62.
- Fernandez MO, Sharma S, Kim S, Rickert E, Hsueh K, et al. (2017) Obese neuronal PPARγ knockout mice are leptin sensitive but show impaired glucose tolerance and fertility. Endocrinology 158(1): 121-133.
- Scicchitano P, Dentamaro I, Carbonara R, Bulzis G, Dachille A, et al. (2012) Cardiovascular risk in women with PCOS. Int J Endocrinol Metab 10(4): 611-618.
- Shroff R, Kerchner A, Maifeld M, van Beek EJ, Jagasia D, et al. (2007) Young obese women with polycystic ovary syndrome have evidence of early coronary atherosclerosis. J Clin Endocrinol Metab 92(12): 4609-4614.
- Alves JPM, Bertolini M, Bertolini LR, Silva CMG, Rondina D (2015) Lipotoxicity: Impact on oocyte quality and reproductive efficiency in mammals. Anim Reprod 12(2): 291-297.
- Li X, Ding W, Liu JY, Mao YD, Huang J, et al. (2018) Effects of dyslipidemia on IVF/ICSI pregnancy outcome in patients with polycystic ovary syndrome. Zhonghua Fu Chan Ke Za Zhi 53(6): 402-408.
- (2018) ACOG Practice Bulletin Summary No.194: Polycystic Ovary Syndrome. Obstet Gynecol 131(6): 1174-1176.
-
Amal AL Mulla, Ahmed EL Sokkary, Sherif Ekladious, Amar Hassan Khamis. Prevalence of Dyslipidemia among Women with Polycystic Ovary Syndrome Based on Body Mass Index. W J Gynecol Women’s Health. 4(1): 2020. WJGWH.MS.ID.000580.
BMI, Dyslipidemia, Polycystic ovary syndrome, Cardiovascular risk, Atherosclerosis, Hyperglycemia, Metabolic syndrome, Phenomena, Obesity
-

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.






