Research Article
Research Article
Supralabyrinthine Petrous Bone Cholesteatoma as a Cause of a Profund Sensorineural Hearing Loss
Ahd Al-Dhabi1, Christian Henker2, Gunnar Gaffke3, Tobias Breyer4, Dörte Fischer1,5 and Tino Just1*
1 Department of Otorhinolaryngology, Head and Neck Surgery, KMG Klinikum, Güstrow, Germany
2 Center for Orthopedics, Trauma, and Spinal Surgery, KMG Klinikum, Güstrow, Germany
3 Department of Diagnostic and Interventional Radiology, KMG Klinikum, Güstrow, Germany
4 Department of Radiology and Neuroradiology, Mediclin Hospital, Plau am See, Germany
5 Department of Audiology, KMG Klinikum, Güstrow, Germany
Tino Just, M.D., Department of Otorhinolaryngology, Head and Neck Surgery Friedrich-Trendelenburg-Allee 1, D-18273 Güstrow, Germany.
Received Date: October 31, 2025; Published Date:November 11, 2025
Introduction
This paper describes a case of acquired supralabyrinthine cholesteatoma that could be accurately diagnosed only through preoperative imaging and the correct interpretation of the findings. The patient was referred to the hospital with an inquiry about hearing improvement. He denied any recurrent infections of the middle ear or inflammations of the outer ear canal. The presented case of supralabyrinthine cholesteatoma was not diagnosed with a detailed otologic history or otomicroscopy. Instead, the high-resolution CT and MR imaging were used for diagnosis. Supralabyrinthine cholesteatoma is one of the five subtypes of petrous bone cholesteatoma (PBC) [1, 2], centered around the geniculate ganglion, with limited medial extension beyond the otic capsule [3, 4].
According to Eggink et al., the classification of cholesteatoma is useful only if it has practical implications for treatment management strategies [5]. Current cholesteatoma classifications, such as JOS [6], EAONO-JOS [6], ChOLE [7], and STAMCO [8], have limited prognostic value in predicting recurrent and residual cholesteatoma, and adverse events. In cholesteatoma surgery, the primary aim is to achieve a disease-free ear and to reduce the risk of disease recidivism [9]. Recidivism encompasses both the sum of residual and recurrent cholesteatoma after surgery. Several factors significantly influence disease recidivism, including surgery type, follow-up, and age [5].
Materials and Methods
The preoperative neuro-otological evaluation included clinical examination, pure tone audiometry, the Freiburger monosyllabic word recognition test at 65 dB (Fb65), speech intelligibility tests, caloric tests, and the Head Impulse Test.
All audiological assessments were conducted in an audiometric sound-attenuated room, using calibrated signals and equipment according to accepted ISO standards. For patient assignment, pure tone audiometry was performed to measure air conduction (AC) and bone conduction (BC) thresholds. The PTA4 was calculated across the frequencies of 0.5, 1, 2, and 4 kHz. Freiburger monosyllable word tests were performed through headphones at 60 dB, 80 dB, and 100 dB. Speech intelligibility tests were conducted in the free field using the Freiburger monosyllable word test in quiet and in noise. The loudspeakers were positioned 1 m away from the participant’s head. In quiet, i.e., under S0 condition, speech intelligibility was measured at 65 dB SPL. In noise, i.e., under S0N0 condition, speech intelligibility was measured with a fixed noise level at 60 dB SPL and speech level at 65 dB SPL, resulting in a signal-to-noise ratio (SNR) of 5 dB.
The taste test was performed on both sides of the tongue using four different concentrations of four taste solutions (sweet: 0.03, 0.1, 0.4, 2 g/mL sucrose solution; sour: 0.01, 0.05, 0.1, 0.15 mL citric acid; salty: 0.025, 0.075, 0.15, 0.36 mL sodium chloride solution; bitter: 0.0002, 0.0005, 0.001, 0.01 mL quinine hydrochloride) [10].
Tympanometry was performed using the Madsen® Zodiac (type 1096; Otometrics) at 226 Hz from -400 to +200 daPa. The resonance frequency of the admittance (Fr [Hz]) and the peak admittance value (P [*10-2 mmho]) were measured using MedWave®. Detailed information is presented elsewhere [11, 12].
Tympanometry was performed using the Madsen® Zodiac (type 1096; Otometrics) at 226 Hz from -400 to +200 daPa. The resonance frequency of the admittance (Fr [Hz]) and the peak admittance value (P [*10-2 mmho]) were measured using MedWave®. Detailed information is presented elsewhere [11, 12].
Case
The 72-year-old patient was referred to the clinic due to persistent hearing loss in his left ear, having been treated for sudden hearing loss 12 years earlier. Pure tone threshold audiometry revealed high-frequency inner ear hearing loss in the right ear and severe sensorineural hearing loss in the left ear. Additionally, he described having experienced dizziness during rapid changes in position and unsteadiness while walking, but without lateropulsion. He did not report tinnitus, middle ear infections, ear discharge, taste disorders, or otalgia; nor had he ever experienced any problems with his ears, besides progressive hearing loss in his left ear.
The otoscopic examination revealed medium-range ear canals on both sides. The right eardrum appeared dull, gray, and unremarkable. The left eardrum was intact but not translucent, with no mesotympanic or epitympanic retraction and without perforation with a mass causing slight bulging of the anterosuperior quadrant of the retrotympanic membrane (Figure 1).
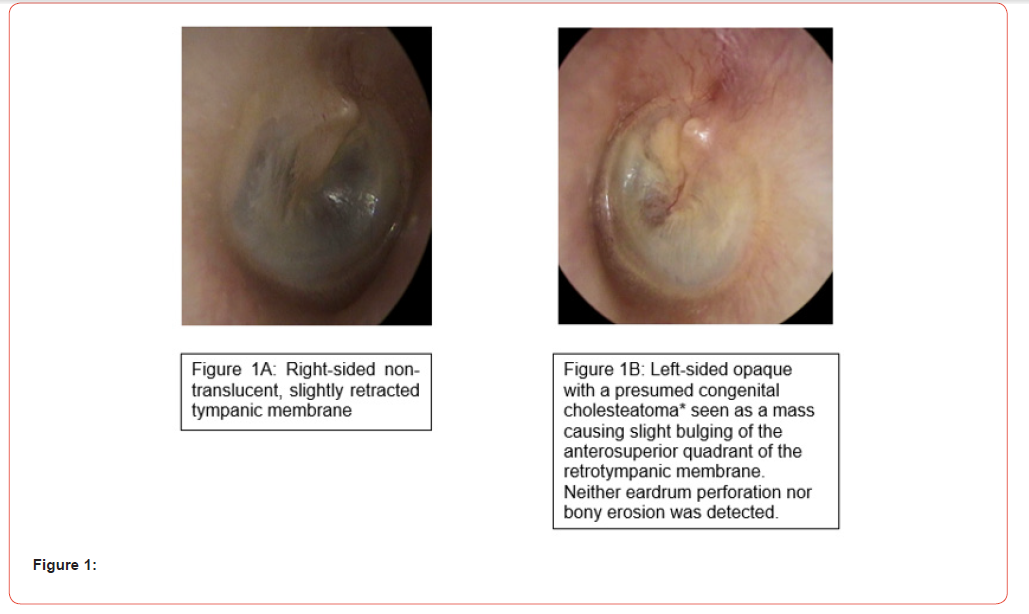
In Weber’s tuning fork test (a1, c3), the tuning fork tone was specified on the right ear. Rinne was positive on the right and negative on the left.
Using single-frequency 226-Hz tympanometry, a tympanogram type A was found in the right ear, and a flat curve (tympanogram type B) on the left side. Acoustic immittance measurements through the outer ear canal revealed a resonance frequency of the admittance of 452.2 Hz for the right ear and 527.6 Hz for the left ear. Video-nystagmography revealed no spontaneous nystagmus. The fistula test was conducted by increasing the external auditory canal pressure on the left ear, indicating a negative result, demonstrating the absence of pressure-induced nystagmus. The positioning test provoked nystagmus when turning the body to the left and right and when shaking the head. The clinical head impulse test indicated a corrective saccade, testing the lateral and anterior canals. However, the video head impulse test showed reduced VOR gains and catchup saccades in both lateral canals and the left posterior canal. The taste test revealed hypogeusia for all tested taste qualities on the left side tongue.
Table 1:
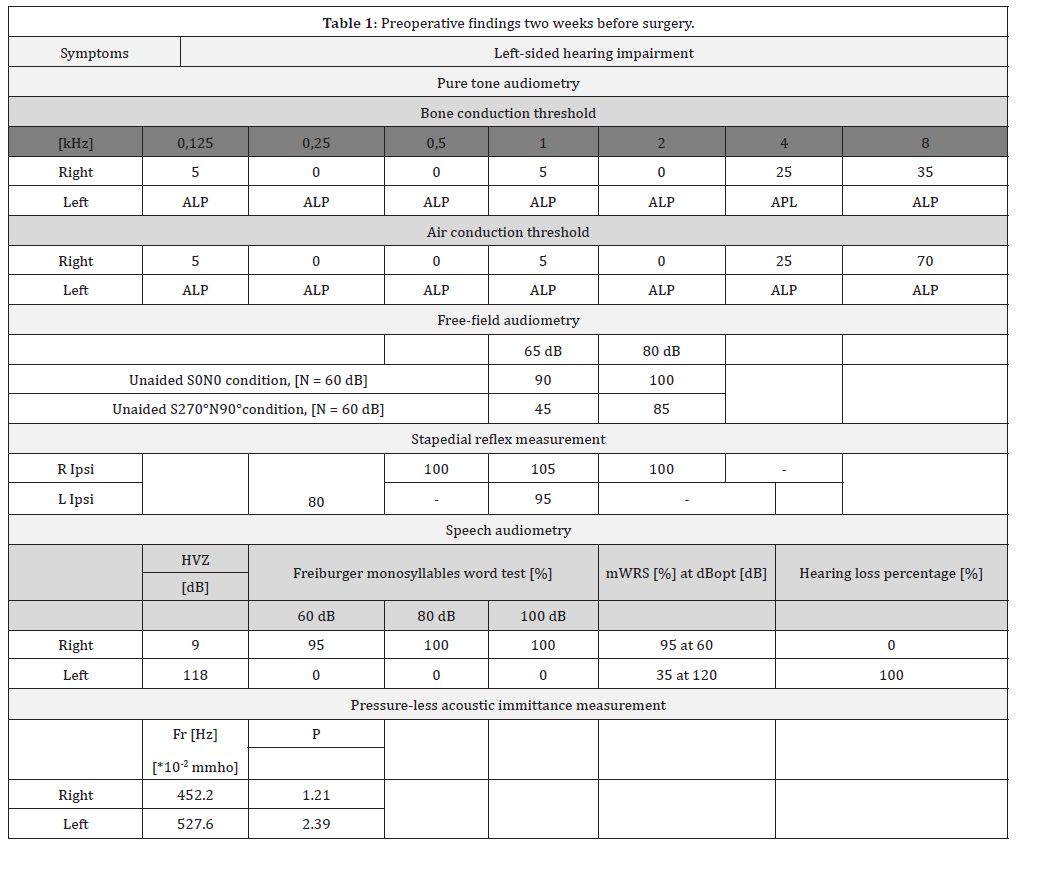
Unaided maximum word recognition score (mWRS); audiometer power limit (APL) resonance frequency of the admittance (Fr [Hz]) and the peak admittance value (P [*10-2 mmho])
The patient underwent a preoperative high-resolution CT scan of the temporal bone (Figure 2A and B) and an MRT scan 1.5 Tesla (Figure 3). The preoperative CT scan of the head revealed a mass within the petrous bone on the left side (see Figure 2). This mass was found to be destroying the arcuate eminence and the underlying anterior semicircular canal, opening the middle fossa. From the most superior part, the tumor invaded medially into the fundus of the internal acoustic meatus and into the middle turn of the cochlea.
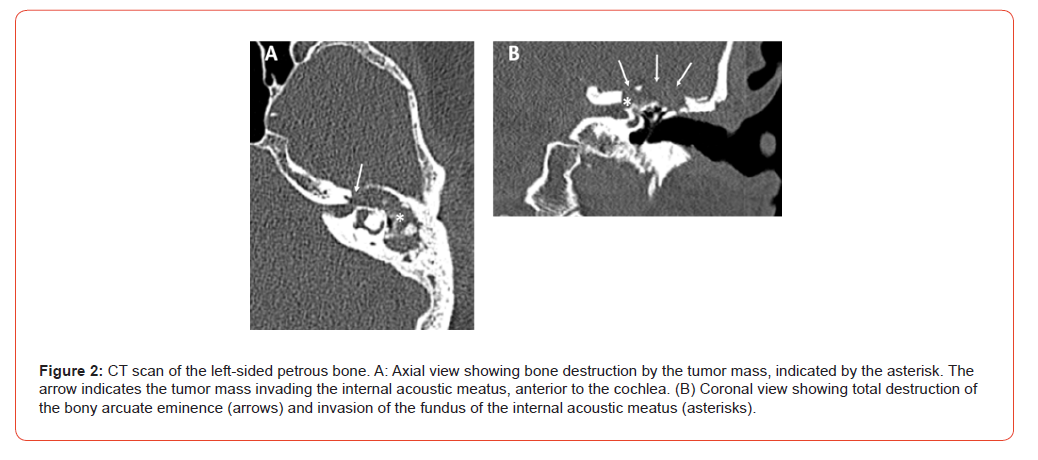
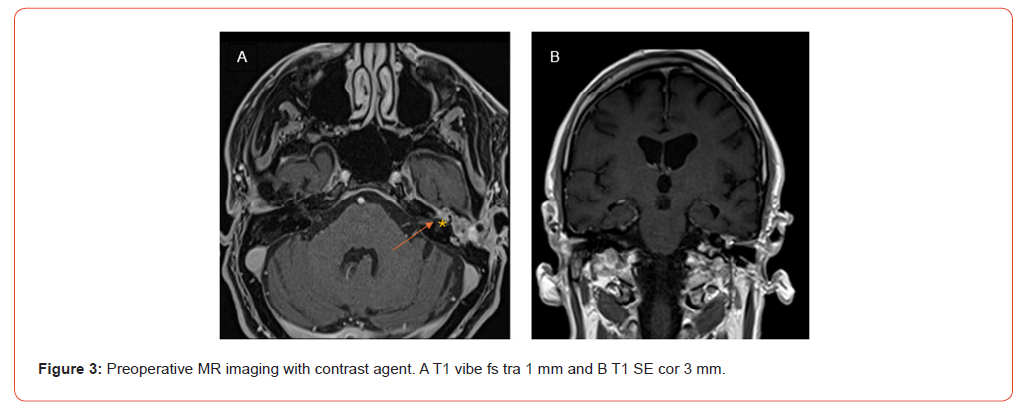
The cholesteatoma invades the cochlea (arrow) and internal acoustic meatus (asterik). Due to suspected supralabyrinthine cholesteatoma, we indicated cholesteatoma surgery with facial neuromonitoring through a modified translabyrinthine approach.
Results
During the modified translabyrinthine approach, an extended cholesteatoma was found, destroying the superior semicircular canal and the middle turn of the cochlea, opening the middle fossa. The circumscribed mass behind the anterosuperior quadrant of the tympanic membrane was a tympanosclerotic plaque. From the most superior part, the cholesteatoma had invaded medially into the fundus of the internal acoustic meatus and into the middle turn of the cochlea, resulting in cerebrospinal fluid leakage. The facial nerve was identified in the middle ear, preserved, and exposed up to the geniculate ganglion. An open mastoid cavity was created, and the head of the malleus and incus were removed, with the chorda tympani nerve dissected. Despite the affected ear having experienced long-term inner ear hearing loss, while the contralateral ear showed nearly normal hearing, the cholesteatoma matrix in the mid-turn of the cochlea and the anterior semicircular canal was completely removed without leaving the matrix intact over the fistula to avoid recurrence. The mid-turn of the cochlea was covered with cartilage and fascia. The large tegmental erosion was repaired using cavum conchae cartilage to prevent meningoencephalic herniation. As the cholesteatoma had invaded the internal auditory channel, it was gently removed. The fundus of the internal acoustic meatus was repaired with two layers of cartilage, temporal fascia, and fibrin glue. Bone dust and cartilage were used to reduce the size of the mastoid cavity.
The patient presented with facial nerve paralysis (House- Brackmann grade VI (HB-VI)) and transient vertigo postoperatively. He exhibited vertical spontaneous nystagmus. High-resolution CT and MRI scans obtained 2-4 days after surgery revealed no intracranial complications and no postoperative cerebrospinal fluid leakage. Intravenous Ceftriaxone (2 g per day) was administered for four days. The patient received vertigo training and a facial exercise program and was discharged five days after surgery. The sutures were removed on the sixth postoperative day, and the tamponade three weeks after surgery. Figure 4 shows the postoperative otoscopic situs.

The residual hearing in the left ear that was present had disappeared postoperatively, and spatial hearing deteriorated (Table 2). According to the patient, the BiCROS hearing aid resulted in a slight improvement in spatial hearing. The dizziness symptoms have completely disappeared four months after the operation. Regarding the facial nerve function, the patient showed a slight improvement four months after surgery (HB VI to IV). A control MRI taken four months after surgery showed no cholesteatoma recurrence, no intracranial complications, and a fluid-filled inner ear.
Table 2:
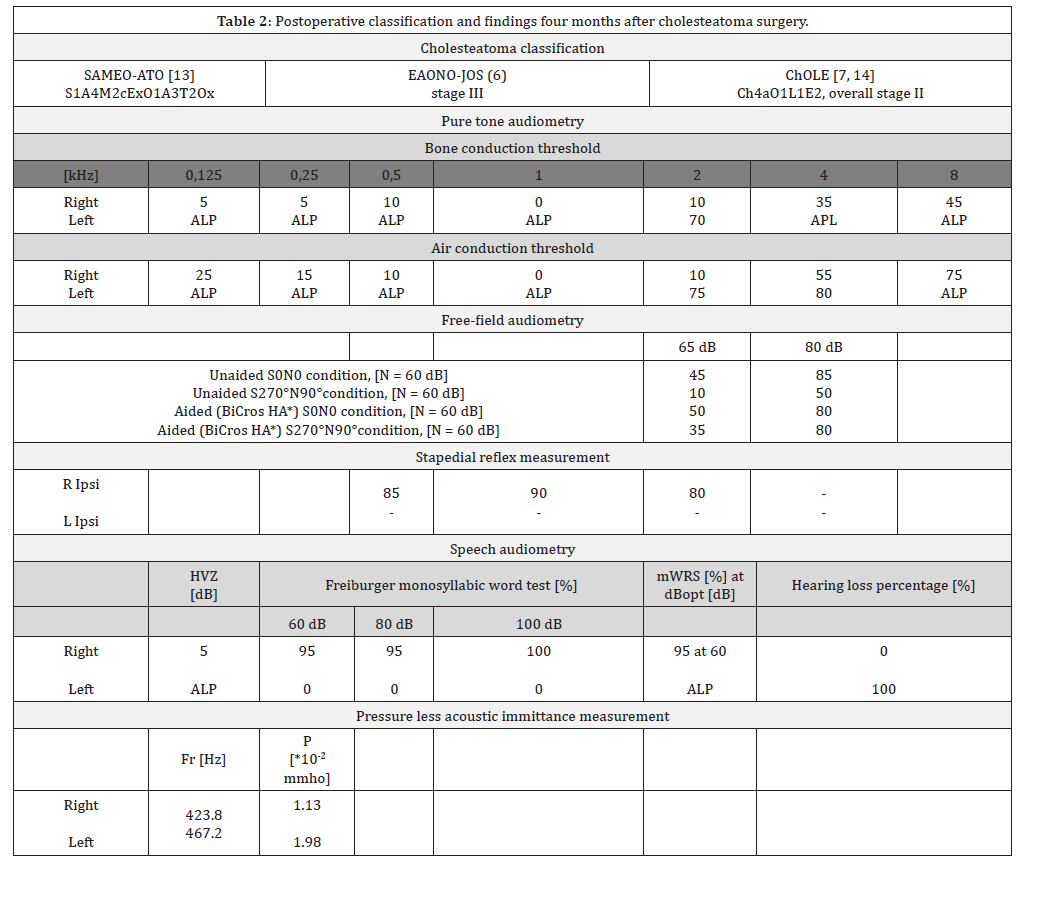
Unaided maximum word recognition score (mWRS); audiometer power limit (APL) resonance frequency of the admittance (Fr [Hz]) and the peak admittance value (P [*10-2 mmho]); HA hearing aid.
Discussion
Despite the absence of a retraction pocket, tympanic membrane retractions, perforation, inflammation, especially otitis media with effusion, or a history of trauma or ear surgery, the presented PBC was presumably either acquired or unclassifiable. Congenital cholesteatoma is believed to be derived from entrapped ectodermal cellular debris during embryonic development. When it involves the middle ear, it appears as a whitish retrotympanic mass localized either anterior or posterior to the malleus. In rare cases, cholesteatoma may develop in some part of the temporal bone away from the tympanic membrane. In such instances, the tympanic membrane may appear intact, or the only indication may be the presence of middle ear effusion. In these cases, the lesion may be recognized in radiological examinations performed after neurological symptoms appear or only by chance. In our case, the slight bulging of the tympanic membrane anterior to the malleus was caused by a calcified plaque. The tympanic cavity was aerated, with no inflammation. In petrous bone cholesteatoma, otoscopy appears irrelevant. A high degree of suspicion is required when a patient presents neurological symptoms that could be explained by various temporal bone lesions. A PBC should be considered, and in this situation, MRI is necessary for diagnosis. MRI provides information for differential diagnosis with cholesterol granuloma, mucocele, and tumors [15].
Generally, a CT scan is not required to diagnose cholesteatoma. However, in the presence of severe sensorineural hearing loss, sudden deafness, headache, vertigo, or facial nerve paralysis, a highresolution CT scan of the temporal bone is essential for accurate diagnosis and evaluation of disease extension.
PBCs are slow-growing epidermoid, non-neoplastic lesions in the petrous portion of the temporal bone. This entity grows locally aggressively and causes bone erosions with extension to the deep bony labyrinth through superior, inferior, and posterior cell tracts, with or without further extensions to nearby structures [16]. If these cholesteatomas are not treated, they may extend beyond the confines of the petrous bone and involve structures such as the nasopharynx, clivus, sphenoid sinus, infratemporal fossa, parapharyngeal space, and intradural space [3, 17, 18].
The incidence of all petrous pyramid lesions is less than 10% [19]. PBCs can be congenital, acquired, or iatrogenic. Due to their slow growth, they often remain asymptomatic [4]. However, the most common presenting symptoms in PBCs are hearing loss (84%), tinnitus (48%), and facial nerve paralysis (45%) [19]. Mixed hearing loss (41%) is the commonest audiometric finding, followed by conductive hearing loss (26%) and profound sensorineural hearing loss (4%). Approximately 29% had deafness at presentation. On preoperative facial nerve function examination, 45% of patients exhibited various degrees of paresis and complete paralysis, while 55% had normal HB-I function. The most common degree of paresis noted was HB-III (18%), followed by HB-VI (5%) (19). In our patient, hearing loss was the only leading symptom, and he exhibited normal HB-I function. Taste disturbances can rarely be the sole symptom of chronic otitis media with cholesteatoma [20]. Preoperative diagnosis of taste disturbance on the ipsilateral side of the tongue was not observed by the patient.
Since PBC affects the otic capsule, it often invades the membranous labyrinth, primarily of the cochlea and the vestibular structures [21]. The main complications of PBC surgery are facial nerve palsy and severe hearing loss [22]. Regarding hearing loss, several authors have claimed that hearing preservation cannot be achieved in most supralabyrinthine PBCs due to the high risk of recurrence [17, 23]. A 2009 study reported good hearing preservation after PBC surgery [24].
Due to the existing severe hearing loss, the treatment of this case focused on preserving the facial nerve. In cases with nonserviceable hearing, a transotic approach is preferred [25]. According to the adapted classification of adverse events by Clavien et al. [5, 26], we observed transient postoperative vertigo (grade I), intraoperative CSF leak requiring reconstruction (grade II), and persistent facial nerve paralysis (grade III) with slight improvement four months after surgery (HB VI to IV). Radical surgery for PBCs includes various techniques aimed at the complete removal of the cholesteatoma matrix while preserving nearby structures, preventing facial nerve dysfunction, postoperative CFS leakage, and meningitis, and preserving hearing function when feasible [3]. The surgical approaches have evolved from radical petromastoid exenteration and marsupialization [16] to obliterative techniques following complete removal of the cholesteatoma [17].
As recommended, preoperative facial nerve function should always be assessed [19]. If it is compromised due to nerve compression, decompression through removal of the cholesteatoma matrix is often sufficient [19]. Conversely, if the nerve is interrupted intraoperatively, continuity can be restored through either rerouting and direct end-to-end anastomosis or cable grafting [3, 4]. In our case, damage to the facial nerve at the level of the geniculate ganglion was suspected, which went unnoticed by the surgeon. However, if facial paresis or paralysis persists for more than two years, re-anastomosis with masseter-to-facial anastomosis is recommended [3, 4].
Conclusion
This case demonstrates the necessity of imaging in patients with severe hearing loss that cannot be explained by either the medical history or otoscopic findings.
Acknowledgement
None.
Conflict of Interest
The authors declare that there is no conflict of interest.
References
- Olszewska E, Rutkowska J, Ozgirgin N (2015) Consensus-Based Recommendations on the Definition and Classification of Cholesteatoma. J Int Adv Otol 11(1): 81-87.
- Sanna M, Pandya Y, Mancini F, Sequino G, Piccirillo E (2011) Petrous bone cholesteatoma: classification, management and review of the literature. Audiol Neurootol 16(2): 124-136.
- Sanna M, Zini C, Gamoletti R, Frau N, Taibah AK, Russo A, et al. (1993) Petrous bone cholesteatoma. Skull Base Surg 3(4): 201-13.
- Omran A, De Denato G, Piccirillo E, Leone O, Sanna M (2006) Petrous bone cholesteatoma: management and outcomes. Laryngoscope 116(4): 619-626.
- Eggink MC, de Wolf MJF, Ebbens FA, Dikkers FG, van Spronsen E (2021) Evaluation of Long-term Cholesteatoma Recidivism: Using the JOS, EAONO-JOS, and STAMCO Cholesteatoma Staging Systems. Otol Neurotol 42(7): 1031-1038.
- Tono T, Sakagami M, Kojima H, Yamamoto Y, Matsuda K, Komori M, et al. (2017) Staging and classification criteria for middle ear cholesteatoma proposed by the Japan Otological Society. Auris Nasus Larynx 44(2): 135-140.
- Linder TE, Shah S, Martha AS, Roosli C, Emmett SD (2019) Introducing the "ChOLE" Classification and Its Comparison to the EAONO/JOS Consensus Classification for Cholesteatoma Staging. Otol Neurotol 40(1): 63-72.
- Merkus P, Ten Tije FA, Stam M, Tan FML, Pauw RJ (2017) Implementation of the "EAONO/JOS Definitions and Classification of Middle Ear Cholesteatoma" - from STAM to STAMCO. J Int Adv Otol 13(2): 272-275.
- Nadol JB Jr (1985) Causes of failure of mastoidectomy for chronic otitis media. Laryngoscope 95(4): 410-413.
- Pingel J, Ostwald J, Pau HW, Hummel T, Just T (2010) Normative data for a solution-based taste test. Eur Arch Otorhinolaryngol 267(12): 1911-1917.
- Bassi F, Accardo A, editors. Estimation of middle ear characteristics by an innovative pressure-less acoustic immittance (PLAI™) device. 9th European Medical and Biological Engineering Conference; 2024; Portoroz, Slovenia: EMBEC 2024, IFMBE Proceedings 113.
- PLAI® methodology used in med wave® 2024 [Available from: neuranix.com.]
- Yung M, James A, Merkus P, Philips J, Black B, et al. (2018) International Otology Outcome Group and the International Consensus on the Categorization of Tympanomastoid Surgery. J Int Adv Otol 14(2): 216-226.
- Eggink MC, de Wolf MJF, Ebbens FA, Dikkers FG, van Spronsen E (2022) Assessing the Prognostic Value of the ChOLE Classification in Predicting the Severity of Acquired Cholesteatoma. Otol Neurotol 43(4): 472-480.
- Mohan S, Hoeffner E, Bigelow DC, Loevner LA (2012) Applications of magnetic resonance imaging in adult temporal bone disorders. Magn Reson Imaging Clin N Am 20(3): 545-572.
- Yanagihara N, Nakamura K, Hatakeyama T (1992) Surgical management of petrous apex cholesteatoma: a therapeutic scheme. Skull Base Surg 2(1): 22-27.
- Prasad SC, Piras G, Piccirillo E, Taibah A, Russo A, et al. (2016) Surgical Strategy and Facial Nerve Outcomes in Petrous Bone Cholesteatoma. Audiol Neurootol 21(5): 275-285.
- Pandya Y, Piccirillo E, Mancini F, Sanna M (2010) Management of complex cases of petrous bone cholesteatoma. Ann Otol Rhinol Laryngol 119(8): 514-525.
- Yilala MH, Fancello G, Sanna M (2024) Management of Petrous Bone Cholesteatoma: The Gruppo Otologico Experience. Eur Arch Otorhinolaryngol 281(10): 5211-5224.
- Guder E, Böttcher A, Just T (2012) Isolated taste disorder as primary symptom of a chronic inflammatory middle ear disease. Laryngorhinootologie 91(1): 34-35.
- Milroy CM, Michaels L (1990) Pathology of the otic capsule. J Laryngol Otol 104(2): 83-90.
- Magliulo G (2007) Petrous bone cholesteatoma: clinical longitudinal study. Eur Arch Otorhinolaryngol 264(2): 115-120.
- Al Zaabi K, Hassannia F, Bergin MJ, Rutka JA (2020) Management of invasive intralabyrinthine cholesteatoma: Can one realistically preserve hearing when disease is medial to the otic capsule? Am J Otolaryngol 41(3): 102407.
- Hong SJ, Lee JH, Jung SH, Park CH, Hong SM (2009) Can cochlear function be preserved after a modified translabyrinthine approach to eradicate a huge cholesteatoma extending to the petrous apex? Eur Arch Otorhinolaryngol 266(8): 1191-1197.
- Pace A, Visconti IC, Iannella G, Milani A, Rossetti V, et al. (2024) Petrous Bone Cholesteatoma: Facial and Hearing Preservation. Ear Nose Throat J 103(6): NP374-NP81.
- Clavien PA, Sanabria JR, Mentha G, Borst F, Buhler L, et al. (1992) Recent results of elective open cholecystectomy in a North American and a European center. Comparison of complications and risk factors. Ann Surg 216(6): 618-626.
-
Ahd Al-Dhabi, Christian Henker, Gunnar Gaffke, Tobias Breyer, Dörte Fischer and Tino Just*. Supralabyrinthine Petrous Bone Cholesteatoma as a Cause of a Profund Sensorineural Hearing Loss. On J Otolaryngol & Rhinol. 8(2): 2025. OJOR.MS.ID.000681.
-
Hearing loss, Outer ear canal, Middle ear, Cholesteatoma surgery, Ear discharge, Auditory canal, Mastoid cavity, Facial nerve, Dizziness symptoms
-

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.