Research Article
Research Article
Maxillary Alveolar Carcinoma: Risk of Recurrence and its Relation with Prognostic Indicators
Shehla Khan and Zubair Durrani*
Oral and Maxillofacial Surgery Department, Rehman Medical Institute, Pakistan
Zubair Durrani, Oral and Maxillofacial Surgery Department, Rehman Medical Institute, Peshawar, Pakistan.
Received Date: September 01, 2020; Published Date: September 17, 2020
Abstract
Background: Maxillary Alveolar carcinoma is a relatively uncommon entity with occurrence reported between 3.5 to 10% of all oral cancers. Recurrence when occurs locally are particularly difficult to treat due to the close proximity of this region to the infratemporal fossa and the base of skull. There is also a debate when to perform elective neck dissection in these patients.
Objectives: The aim of this study was to identify cases with recurrence and correlate its association with various prognostic factors and disease free survival in these patients.
Methodology: Overall, 15 cases were identified with maxillary alveolar carcinomas that were treated between 2011 to 2015. Eight of these cases were Squamous cell carcinomas, 5 Adeno carcinomas and 2 Adenocystic carcinomas. T stages, N stage, grade of tumor, surgical margin involvement by the tumor, presence of perineural and lymphovascular invasion were identified as prognostic indicators. The chi-square and Pearson correlation tests were applied to analyze the association between recurrence and these prognostic factors.
Results: Recurrence developed in 3 SCC (37.5%) and 1 ADC (20%) patient during the three years period following the treatment. Among the SCC patients, 2 recurrences were local and 1 regional. Recurrence was found to have a strong positive correlation with T stage (r=0.330, p=0.271), surgical margin involvement (r=0.575, p=0.566) and three years disease free survival (r=0.959, p=0.000). A weak positive correlation was also established with tumor grade (r=0.155, p=0.0613) and perineural invasion (r=0.149, p =0.725).
Keywords: Oral squamous cell carcinoma; Maxillary alveolus; Hard palate; Prognostic factors; Survival rates; Recurrence
Introduction
Background
Oral carcinoma is one of the ten most frequently occurring cancers around the world and when grouped together with pharyngeal cancer, it is the sixth most common cancer in the world [1]. In the Asian and South east population the prevalence of oral cancer is on the rise. The unique cultural practices in the Asian population such as snuff dipping, smoking, use of alcohol and areca nut are the main predisposing factors for the development of this cancer. The incidence of OSCC restricted to the hard palate and maxillary ridge is very low, with an estimated 3.5 to 10 % of all OSCC [2,3]. Because of the anatomical proximity of the upper alveolar mucosa with the maxillary gingivo‐buccal sulcus, cancer of the maxillary alveolus may spread to the nearby spaces thus making it difficult to locate the actual initiation point of the disease [4].
Various prognostic factors have been suggested that could affect patients overall survival and disease specific survival in oral cancer. Of the known prognostic factors, the TNM stage, histological grade, and tumor thickness are widely recognized [5]. Other prognostic factors like lympho-vascular invasion (LVI) and peri-neural invasion (PNI), extracapsular spread and involvement of surgical margin by tumor cell are also known to play a role in recurrence and overall poor prognosis [6-9].
Objectives
The aim of this study was to identify cases of recurrence and correlate its association with various prognostic factors and disease-free survival in these patients.
Methodology
A retrospective cohort study was planned to identify patients who had undergone surgical resection for maxillary alveolar carcinoma over a period of 4 years from 2011 to 2015. Data was collected through clinical and pathological records of the patient. A total of 26 Maxillectomies were performed during this period. Out of these, 15 cases were maxillary alveolar carcinomas while the rest were either non-malignant lesions or the sarcomas of maxillary alveolus. The 15 carcinomas were then classified according to their type. Gender, age distribution, T stage, N stage, histological grade, perineural and lymphovascular invasion and involvement of surgical margin by the tumor was determined. The cases which had received Adjuvant Radiotherapy and those who had developed recurrence were identified. Disease free survival was measured at three years following completion of treatment.
4 cases with recurrence were then categorized separately. Various prognostic indicators that might have played a role in recurrence were correlated with risk of recurrence and their significance determined using chi-square and Pearson correlation. These prognostic indicators were tumor grade, T stage, N stage, Perineural invasion (PNI), lymphovascular invasion (LVI) and involvement of surgical margin by tumor. Recurrence was then evaluated for its impact upon disease free survival.
Results
The distribution of the 15 carcinomas according to its type was as following: 8 squamous cell carcinomas (SCC), 5 Adenocarcinomas (ADC) and 2 Adeno-cystic carcinomas (ACC). Gender wise, SCC had affected 8 females, ADC 5 males and ACC 1 male and 1 female. SCC had occurred between the ages of 35 to 76, ADC between 35 to 58 while the 2 ACC cases were found in 43 and 53 years old. Mean age for SCC was 51.1 years, median 50 years and modal 45 years with standard deviation of 12.35 years. 7 cases of SCC were well to moderately differentiated while 1 was poorly differentiated. 4 ADC cases were well differentiated and 1 was poorly differentiated while there was 1 each in well and poorly differentiated category for ACC. Only 1 SCC presented at T1 stage, 5 SCC and 2 ADC presented at T2 stage, 1 SCC, 3 ADC and 1 ACC presented at T3 stage while 1 SCC and 1 ACC presented at T4 stage. On histopathological evaluation perineural invasion and lympho-vascular invasion was found in 2 case of SCC only. 5 SCC and 2 ADC had microscopically margin involved on surgical resection specimen. Adjuvant Radiotherapy was given to 5 SCC cases, 1 ADC and 2 ACC cases (Table 1).
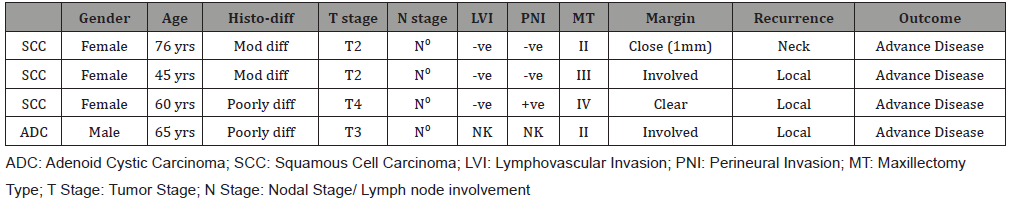
5 SSC (62.5%), 4 ADC (80%) and 2 ACC (100%) patients were disease free at 3 years following treatment. 3 SCC (37.5%) and 1 ADC (20%) patient had developed recurrence. Of the three recurrences in SCC cases, 2 were local and 1 regional. The impact of various prognostic factors on 4 cases of recurrence was then evaluated. Recurrence was found to have strong positive correlation with T stage (r=0.330, p=0.271) and surgical margin involvement (r=0.575, p=0.566) while a weak positive correlation was identified with tumor grade (r=0.155, p=0.0613) and perineural invasion (r=0.149, p=0.725). Since none of the recurrence cases had either clinical or radiological nodal involvement or lymphovascular invasion found in them, it was designated as not having any relationship with recurrence. Recurrence was also found to have a strong positive relation with three years disease free survival (r=0.959, p =0.000) (Table2).
Table 1: Cancer Cases.
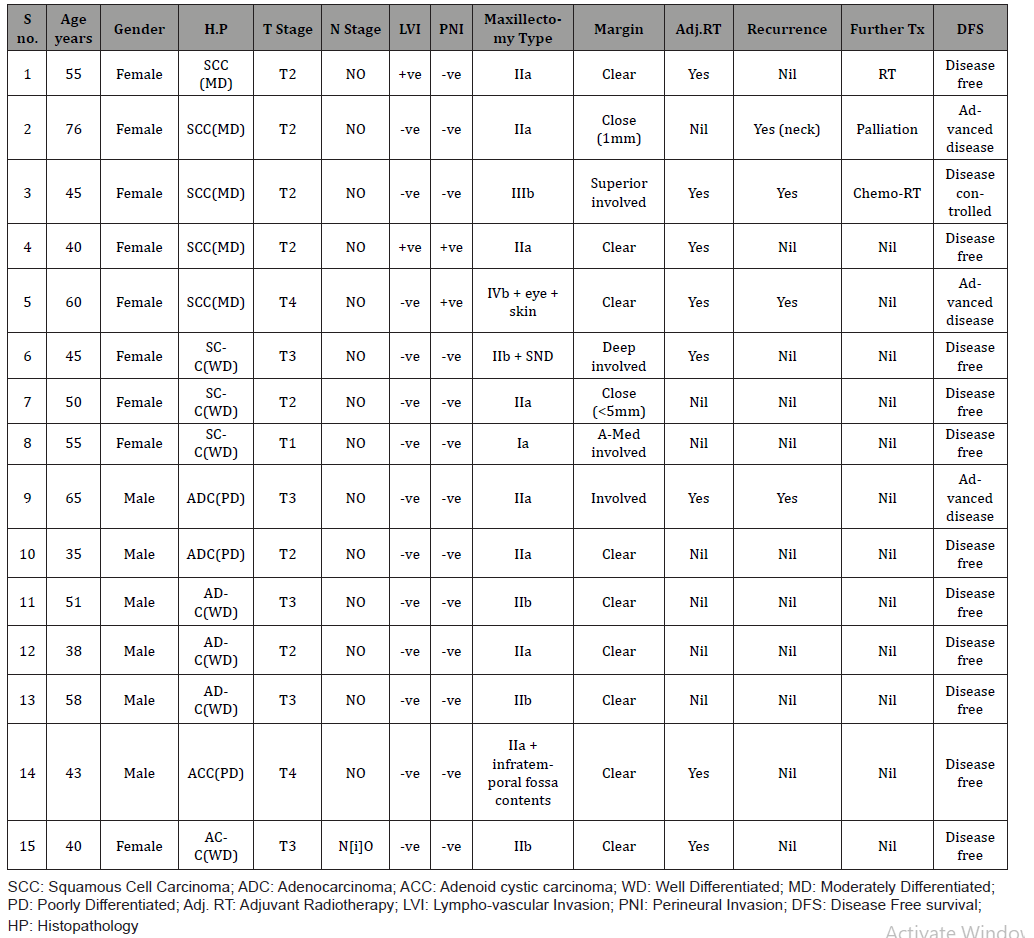
Table 2: Recurrence Cases.
Discussion
The biology of oral cancer varies with site of its presentation. The tongue carcinoma, for example, is a much aggressive entity as compared to lip carcinoma. Similarly maxillary alveolar carcinoma differs from Mandibular alveolar carcinoma in terms of biological behavior. The rarity of maxillary alveolar carcinoma, its location in oral cavity and the limited information about its biology has led to debate about its optimum management.
Lin, et al. [10] in a case series of 725 cases of maxillary alveolar and hard palate carcinoma, found cervical metastasis in 4.1% in T1, 14.9% in T2, 10.3% in T3 and 24.7% in T4 cases. They also found significant effect of T stage and Nodal stage on mean overall survival [10]. Dalal, et al. [11] found 23% cervical metastasis in 25 of the 30 cases of T4 squamous cell carcinoma at initial presentation. Hence recommending Elective neck dissection for all T4 maxillary alveolar carcinomas [11]. Joosten et al reported that in a group of 77 patients out of 95, 14.3% (11/77) had occult metastasis. In T2-T4 patients, this number increased to 19.0% while in T4 alone it was 24.1%. 45.5% of the occult metastasis developed in the contralateral neck. Their salvage rate for recurrence cases was 40%. Based upon these findings, they recommended elective neck dissection for all T2-T4 cases [12]. Similar recommendation was also made by Morris et al, based upon their analysis of 139 patients of maxillary alveolar and hard palate carcinoma. Regional failure in their study was 28.4%, ranging from 18.7% (pT1) to 37.3% (pT4) [13].
The other peculiarity of maxillary alveolar carcinoma is its anatomical location. Anterior alveolus is superiorly related to nasal cavity, middle alveolus is closely associated with maxillary sinus and then to orbital cavity and the posterior alveolus carcinomas tend to infiltrate to the infratemporal fossa. McMahon, et al. [14] in a review of 50 patients reported recurrences in 16, out of which 11 were local. Of these 11, 8 were in superior and posterior direction that included orbit, infratemporal fossa, pterygopalatine fossa, the traversing canals of sphenoidal bone, the gasser an ganglion and the dura of the middle cranial fossa. Hence they concluded that the advance cancer of mid face often equated with disease of the skull base [14]. Wang, et al. [15] in a 10-year review of survival outcome for hard palate and maxillary alveolar carcinoma found involvement of soft palate or infratemporal fossa as poor outcome indicators. Ulcerative tumor features, tumor volume larger than 10 ml and local disease that could not be salvaged had poor survival outcome [15]. Li, et al. [16] in-case series of 155 cases stated that the 5-year disease specific survival rates N+ and N- patients was 21.54 % and 47.36% respectively. They also reported that cervical metastasis was found in as high as 49.03% of all cases with 40% presenting at initial consultation and 9.03 % presenting after therapy [16]. Likhterov, et al. [17] in a study of 75 cases reported 19 out of total of 22 recurrences occurred locally. Additionally they identified recurrence was associated with T4 disease, positive margins and surveillance imaging. Furthermore in cases where reconstructive flap was used to repair the defect, it needed flap mobilization to obtain biopsy. In 13 of the 19 cases salvage surgery was attempted and was successful in 6 (42%) cases [17].
Conclusion
Based upon the literature search, it seems the consensus is developing that Elective neck dissection is indicated for lesions between T2-T4 (SCC). The most important prognostic factors are T stage, N stage and surgical margin involvement. Tumor grade and perineural invasion may be weaker indicators but play a role in recurrence and disease free survival. Recurrence can occur in the form of nodal metastasis but local is more common. Surgical salvage probably offers the best chance to the patient for clearance but the overall survival for these patients remains poor.
Acknowledgement
Dr. Iftikhar Qayum, Director Research, Rehman Medical Institute for his support in preparing research methodology and data analysis.
Conflict of Interest
No conflict of interest.
References
- Warnakulasuriya S (2009) Global epidemiology of oral and oropharyngeal cancer. Oral Oncol 45: 309-316.
- Rao DN, Shroff PD, Chattopadhyay G, Dinshaw KA (1998) Survival analysis of 5595 head and neck cancers – Results of conventional treatment in a high-risk population. Br J Cancer 77: 1514-1518.
- Baishya N, Rahman T, Das AK, Kalita CR, Sharma JD, et al. (2019) Squamous cell carcinoma of upper alveolus: An experience of a tertiary care center of Northeast India. South Asian J Cancer 8: 44-46.
- Pathak KA, Mathur N, Talole S, Deshpande MS, Chaturvedi P, et al. (2007) Squamous cell carcinoma of the superior gingival-buccal complex. Oral Oncol 43: 774-779.
- Carvalho AL, Ikeda MK, Magrin J, Kowalski LP (2004) Trends of oral and oropharyngeal cancer survival over five decades in 3267 patients treated in a single institution. Oral Oncol 40: 71-76.
- Binmadi NO, Basile JR (2011) Perineural invasion in oral squamous cell carcinoma: a discussion of significance and review of the literature. Oral Oncol 47: 1005-1010.
- Sutton DN, Brown JS, Rogers SN, Vaughan ED, Woolgar JA (2003) The prognostic implications of the surgical margin in oral squamous cell carcinoma. Int J Oral Maxillofac Surg 32: 30-34.
- Jones HB, Sykes A, Bayman N, Sloan P, Swindell R, et al. (2009) The impact of lymphovascular invasion on survival in oral carcinoma. Oral Oncol 45: 10-15.
- Shaw RJ, Lowe D, Woolgar JA, et al. (2010) Extracapsular spread in oral squamous cell carcinoma. Head Neck 32: 714-722.
- Lin HW, Bhattacharyya N (2009) Survival impact of nodal disease in hard palate and maxillary alveolus cancer. Laryngoscope 119(2): 312-315.
- Dalal AJ, McLennan AS (2013) Cervical metastases from maxillary squamous cell carcinoma: retrospective analysis and review of the literature. Br J Oral Maxillofac Surg 51(8): 702-706.
- Joosten MHMA, de Bree R, Van Cann EM (2017) Management of the clinically node negative neck in squamous cell carcinoma of the maxilla. Oral Oncol 66: 87-92.
- Morris LG, Patel SG, Shah JP, Ganly I (2011) High rates of regional failure in squamous cell carcinoma of the hard palate and maxillary alveolus. Head Neck 33(6): 824-830.
- McMahon JD, Wong LS, Crowther J, Taylor WM, McManners J, et al. (2013) Patterns of local recurrence after primary resection of cancers that arise in the sinonasal region and the maxillary alveolus. British Journal of Oral and Maxillofacial Surgery 51(5): 389-393.
- Wang TC, Hua CH, Lin CC, Tsou YA, Tseng HC, et al. (2010) Risk factors affects the survival outcome of hard palatal and maxillary alveolus squamous cell carcinoma: 10-year review in a tertiary referral center. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 110(1): 11-17.
- Li Q, Wu D, Liu WW, Li H, Liao WG, et al. (2013) Survival impact of cervical metastasis in squamous cell carcinoma of hard palate. Oral Surgery, Oral Medicine, Oral Pathology and Oral Radiology 116(1): 23-27.
- Likhterov I, Fritz MA, El-Sayed IH, Rahul Seth, Rayess HM, et al. (2017) Locoregional recurrence following maxillectomy: implications for microvascular reconstruction. Laryngoscope 127(11): 2534-2538.
-
Shehla Khan, Zubair Durrani. Maxillary Alveolar Carcinoma: Risk of Recurrence and its Relation with Prognostic Indicators. On Otolaryngol & Rhinol. 3(4): 2020. OJOR.MS.ID.000567.
-
Neck dissection, Vascular invasion, Tumor, Adenocarcinomas, Tongue carcinoma, Sphenoidal bone, Cranial fossa, lymphovascular, Smoking.
-

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.