 Short Communication
Short Communication
Importance of Dose Metrics for Understanding Sublethal Pentachlorophenol Toxicity in Aquatic Systems
Olli Pekka Penttinen1* and Jussi VK Kukkonen2
1University lecturer, Ecosystems and Environment Research Programme, University of Helsinki, Finland
2Professor, Department of Environmental and Biological Sciences, University of Eastern Finland, Finland
Olli-Pekka Penttinen, Ecosystems and Environment Research Programme, University of Helsinki, Finland.
Received Date:October 13, 2023; Published Date:October 30, 2023
Abstract
Whole animal metabolic rate as mechanism-specific response metrics related to respiratory uncoupling represents sensitive body residue approach that bridges ecotoxicology with ecophysiology. In this context more information is required for understanding the coupling of metabolic rate affecting factors, their directions and magnitude as a functional component of the organism’s metabolic machinery. Here environmental temperature was relevant source of variability to be explored and form the focus of this study. The direct calorimetry was used to measure metabolic rate of oligochaete worm Lumbriculus variegates, and effect of a known uncoupling agent pentachlorophenol (PCP) in acute chemical exposures at low or low and high temperatures (2 and 20 ºC for L. variegatus) was monitored.
The data showed many important accumulation patterns of PCP for aquatic worm. As a result, the highest BCF value of ca. 1300 was found in the worms exposed to PCP concentration at 20ºC. The test organisms responded to treatments of PCP with a significant dose dependent increase in heat output by a maximum factor of three. We conclude that the body residue approach makes a Transition from Toxicology to Ecotoxicology by forming a linkage to abiotic and biotic factors that modify bioavailability, bioaccumulation, and toxic response.
Keywords:Body residues; Direct calorimetry; Ecotoxicology; Lumbriculus variegatus; Metabolic rate; Pentachlorophenol; Physiological trait; Pentachlorophenol
Introduction
Body residues with molar units have high toxicological relevance and the relationship between body residues and biological effects, including both lethal or sublethal effects, is of ecotoxicological importance especially helping to overcome difficulties related to bioavailability in different environmental compartments [1-3] by reducing variability in toxicity data [4].
In our previous studies we have used metabolic rate of aquatic organisms as a response metrics with the aim of determining body residues for chemicals acting as uncouplers of oxidative phosphorylation.
The results indicate that an acute exposure to uncoupling agents leads to dose-depending metabolic rate-enhancing effect at the whole organism level in different aquatic species under variable environmental conditions, if a critical internal threshold concentration for response development is achieved [5,6].
In this context more information is required for understanding the coupling of metabolic rate affecting factors, their directions and magnitude as a functional component of the organism’s metabolic machinery. From our perspective, environmental temperature is important since the ambient environmental temperature represents the most important external factor affecting metabolic rate. As a result, the initial change of metabolic rate in poikilotherms increases or decreases by a factor 2-3 per 10°C change within the thermal range they are able to get adapted [4]. At the same, 1.5 to 3-fold increase in overall metabolic rate is reported in aquatic organisms exposed to uncoupling agents [6,7].
Here environmental temperature was a relevant source of variability to be explored in the context given above and form the main focus of this study. Thus, metabolic rate of oligochaete worm Lumbriculus variegatus was measured, and effect of a known uncoupling agent pentachlorophenol (PCP) in 24h chemical exposures at two temperatures (2 and 20ºC) on metabolism was analyzed.
Material and Methods
The oligochaete worm L. variegatus was reared in the Aquatic Toxicology Laboratory at the University of Eastern Finland under conditions previously described [6]. The cold-acclimated culture (3±1°C) was formed three years before use in the experiments. The unlabeled PCP; originally 98% pure was obtained from Fluka (Buchs, Switzerland) and [14C]-labeled pentachlorophenol (PCP); specific activity 6.5 μCi/mmol from Sigma Chemical (St. Louis, MO, USA).
Two sets of experiments were run with L. variegatus; one set at 2°C, and another one at 20°C. At both temperatures, animals were exposed to a mixture of radiolabeled and nonlabelled PCP for 24 h in the beakers including controls and seven test concentration that ranged from 0.08 to 1.4 μM, and from 0.08 to 0.8 μM, for 2 and 20°C, respectively. During the last two exposure hours, animals were incubated in the closed microcalorimetric ampoule while their heat dissipation was continuously monitored.
The metabolic rate of individual test organisms was monitored directly as the rate of heat dissipation (=heat output) by using an isothermal heat conduction type microcalorimeter TAM 2277 (Thermal Activity Monitor, Thermometric, Järfälla, Sweden). The post-measurement treatments of samples, radioactivity measurement, heat output data acquisition and statistical analysis were done as described previously [5,6].
A bioconcentration factor (BCF) on a wet mass basis was calculated as the ratio of concentration in organisms after 24h (L. variegatus and the corresponding chemical concentration in the surrounding water; BCF= CB/CW (μmol/kg / μmol/ L).
Results and Discussion
After a 24-h exposure, the worms accumulated PCP body residues in the range of 0.045 to 0.66 (L. variegatus at 2°C) and 0.11to 0.64 (L. variegatus at 20°C) μmol/g wet weight (Table 1). This accounts for 1.7 to 6.3% (mean ± standard deviation [SD] 4.1± 1.1), 2.6 to 10.4 % (5.6± 1.6), of initially added PCP for L. variegatus at 2 and 20°C.
Table 1:Treatment-specific information of body size (wet mass), metabolic rate (μW/g), pentachlorophenol body residues (μmol/g), and bioconcentration factors for Lumbriculus variegatus (Lv) at two temperatures.
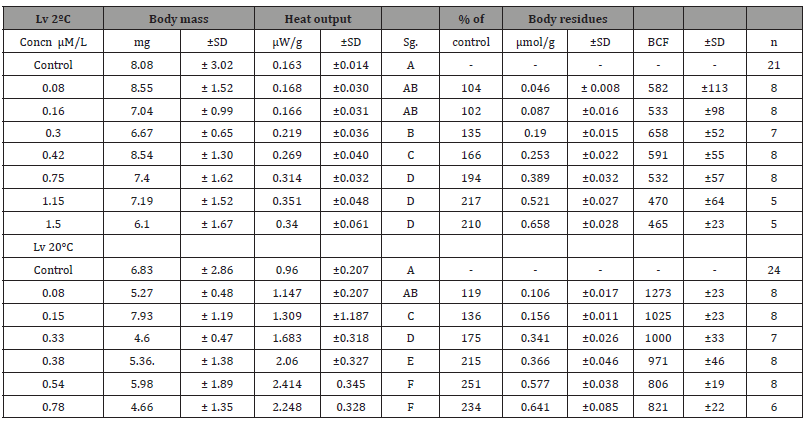
ab Significance; treatments with the same uppercase letter are not significantly different from one another (ANOVA , =0.05), cHeat output value as percentage of the relevant control
The observed body residues were significantly different among temperature (F1,108= 9.46, p= 0.003). As a result, BCFs (Table 1) varied a hundredfold between the treatments, and the highest BCF value of ca. 1300 was found in the worms exposed to PCP concentration of less than 0.4 μM at 20ºC. Above internal PCP threshold concentration of ca. 400 μmol/g gradual decrease of the BCF in the worms was observed.
At 2°C, heat output of unexposed worms varied from 0.91 to 2.1 μW/ individual (mean ±SD 1.26 ± 0.35, n=21) or 90.8 -196.6 μW/g (162.6 ± 14.3). The heat output showed an increased rate at a higher exposure temperature with the temperature coefficient Q10 of 3.1 (L. variegatus; control rate). At 20ºC, the highest individual level variation among treatments was recorded (2.8 to 16.8 μW/ individual (mean ±SD; 7.3± 3.8, n= 24) but it was reduced by using mass-scaled rate that has a range from 627.0 to 1355.0 μW/g (959.7 ± 207.0). At both temperatures after 24h PCP exposure, the test organisms responded to treatments of pentachlorophenol with a significant dose dependent increase in heat output by a maximum factor three for L. variegatus.
That physical, chemical, and biological factors affect metabolic rates has been known for a long time, but information about toxicant-induced variation in metabolism is limited although it is expected that within aquatic systems and organisms there will be variability between pollutant-exposed individuals in terms of physiological performance. That acute exposure to pentachlorophenol leads to a heat output-enhancing effect has also been known for a long time and therefore PCP is an appropriate model compound both in terms of physiological response [5-7] and the body residue- based approach [5-6]. Weak acid respiratory uncouplers, such as pentachlorophenol, exert their toxicity by disrupting the electrochemical proton gradient built up across the inner mitochondrial membrane. This leads to increased oxygen consumption and metabolic heat output. The current study observed that exposure to PCP significantly increased the heat output of exposed organisms. An attempt to form causal heat output-mediated link between tissue residues and PCP toxicity was also successful. Consequently, a direct link to the body residue concept i.e., link between bioaccumulation and ecotoxicity, was established.
Acknowledgement
Academy of Finland Research Council for Environmental and Natural Resources supported this research, project 63774.
Conflict of Interest
None.
References
- Penttinen OP, Kilpi Koski J, Jokela M, Toivainen K and Väisänen A (2008) Importance of dose metrics for lethal and sublethal sediment metal toxicity in the oligochaete worm Lumbriculus variegatus. J Soils Sediments 8: 59-66.
- Penttinen S, Malk V, Väisänen A, Penttinen OP (2011) Using the critical body residue approach to determine the acute toxicity of cadmium at varying levels of water hardness and dissolved organic carbon concentrations. Ecotoxicol Env Saf 74(5): 1151-1155.
- Barron MG, Hansen JA, Lipton J (2002) Association between contaminant tissue residues and effects in aquatic organisms. Rev Environ Contam Toxicol 173: 1-37.
- van der Heijden SA, Hermens JLM, Sinnige TL, Mayer P, Gilbert D (2015) Determining High-Quality Critical Body Residues for Multiple Species and Chemicals by Applying Improved Experimental Design and Data Interpretation Concept. Environ Sci Technol 49: 1879-1887.
- Penttinen OP, Kukkonen JVK (1998) Chemical stress and metabolic rate in aquatic invertebrates: Threshold, dose-response and mode of toxic action. Environ Toxicol Chem 17: 883-890.
- Penttinen OP, Kukkonen JVK (2000) Metabolic responses of Lumbriculus variegatus to respiratory uncoupler in cold and anoxic water. Environ Toxicol Chem 19: 2073-2075.
- McKim JM, Schmieder PK, Carlson RW, Hunt EP, Niemi GJ (1987) Use of respiratory-cardiovascular responses of rainbow trout (Salmo gairdneri) in identifying acute toxicity syndromes in fish. 1. Pentachlorophenol, 2.4-dinotrophenol, tricaine methanesulfonate and 1-octanol. Environ Toxicol Chem 6: 295-312.
-
Olli Pekka Penttinen* and Jussi VK Kukkonen. Importance of Dose Metrics for Understanding Sublethal Pentachlorophenol Toxicity in Aquatic Systems. Online J Ecol Environ Sci. 1(3): 2023. OJEES.MS.ID.000515.
-
Ecotoxicology, Ecophysiology, Direct calorimetry, Pentachlorophenol, Environmental temperature, Aquatic toxicology, Metabolic, Critical internal threshold, Oxidative phosphorylation, Physiological trait, Lumbriculus variegatus
-

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.






