 Case Report
Case Report
The Worst-Case Scenario of Electrolytes Imbalance, Cardiac Arrest, and Acquired Prolonged QT Interval: A Case Report and Literature Review
Maryam Al Arfaj1, Ali AL Qarni1,2,3, Muneera AlTaweel1,2* and Abdulrahman Al Omair1
1Department of Internal Medicine, King Abdulaziz Hospital, Ministry of National Guard Health Affairs, Saudi Arabia
2King Abdullah International Medical Research Center (KAIMRC), Ministry of National Guard Health Affairs, Saudi Arabia
3King Saud bin Abdulaziz University for Health Sciences, Ministry of National Guard Health Affairs, Saudi Arabia
Muneera AlTaweel, King Abdulaziz Hospital, Ministry of National Guard Health Affairs, Al-Ahsa, Saudi Arabia.
Received Date: May 01, 202; Published Date: May 12, 2023
Abstract
Long QT syndrome (LQTS) is the prolongation of QT interval in the electrocardiogram; it is usually classified as congenital or acquired. It is a life-threatening condition that could lead to malignant ventricular arrhythmias and sudden cardiac arrest. The most common causes for Acquired Long QT syndrome are medications like anti-arrhythmic, electrolyte imbalance, or endocrine disorders. Therefore, any patient having one of these conditions should be monitored carefully to prevent deleterious consequences. The central management for acquired LQTS is the correction of the underlying cause. However, recent studies showed that implantable cardioverter-defibrillator (ICD) could be helpful in the secondary prevention of such patients. Here we report a case of cardiac arrest due to acquired prolonged QT interval secondary to endocrine disorder and electrolytes imbalance.
Keywords:QT prolongation; Electrolytes imbalance; Cardiac arrest
Introduction
The term Long QT syndrome (LQTS) is defined by QT interval prolongation in electrocardiogram (ECG), more than 470 milliseconds in males, and 480 milliseconds in females, related to delayed cardiac repolarization [1]. This syndrome is linked to a higher risk of developing torsades de pointes (TdP), a lifethreatening cardiac arrhythmia characterized by polymorphic ventricular tachycardia [2]. It can be categorized into congenital or acquired. Patients with LQTS typically experience palpitations, syncope, seizures, and sudden cardiac death as primary symptoms [1]. Congenital LQTS is caused by mutations in the genes that code for cardiac ion channels and the proteins that interact with those channels. It affects 1 in 2000 people [3]. On the other hand, the prevalence of acquired LQTS is much less than congenital LQTS, and the most common causes are medications like anti-arrhythmic or anti-depressant. Chronic predisposing factors include structural heart diseases, metabolic and endocrine disorders like hypocalcemia, hypomagnesemia, under-replaced hypothyroidism, or acute electrolyte imbalance [4]. Cardiac arrest secondary to marked QT prolongation may occur under many conditions, but hypocalcemia is an extremely rare cause [5]. In this report, we present a 47-year-old woman post-cardiac arrest induced by persistent hypocalcemia and hypomagnesemia.
Case Report
A 47-year-old female known to have cured papillary thyroid carcinoma post-total thyroidectomy with iatrogenic hypoparathyroidism, on Calcium Carbonate 1200mg orally twice daily, Alfacalcidol 0.75 mcg and Cholecalciferol 2000 Units oral once daily, and primary hypothyroidism on levothyroxine 125 mcg oral once daily. She was also diagnosed with stage 2b Hodgkin’s lymphoma with mediastinum mass in 2003, chronic hepatitis B carrier, and C5/6 disc prolapse with radiculopathy. She has no background of any cardiac disease nor a family history of heart disease or sudden death, She was brought to the Emergency department (ED) on 19/12/2022 after she was found unresponsive at home. In ED, cardiopulmonary resuscitation (CPR) was done, and return of spontaneous circulation (ROSC) was achieved after 12 minutes; then, she was intubated and admitted to Intensive Care Unit (ICU).
Pulmonary Embolism was ruled out by chest CT scan, stroke was also ruled out by brain CT scan; septic workup was negative. In ICU, the patient was stabilized; she was then extubated three days later and transferred to our hospital under general medicine for continuity of care. When she was found to have prolonged QT interval on Electrocardiogram (ECG) and electrolytes imbalance (hypocalcemia, hypomagnesemia), she was referred to cardiology as prolonged QT interval was considered the most likely cause of cardiac arrest. She was also referred to endocrinology because of the suboptimal replacement of hypothyroidism and hypocalcemia.
The patient denied any history of chest pain or shortness of breath and no history of loss of consciousness before this event. However, she confirmed poor adherence to her medication. She also complained of palpitation on exertion. ; physical examination revealed a conscious, alert, and oriented patient, not in distress with normal vital signs(pulse rate of 89 beats per minute (bpm), which was regular, systolic blood pressure (SBP) 122 mmHg, diastolic blood pressure (DBP) 65 mmHg, , and respiratory rate 18 breath/minute, and temperature 36.8 °C and oxygen saturation was 98% on room air, cardiovascular, chest, abdomen, and lower limb exam was unremarkable Electrocardiogram (ECG) on admission (27/12/2022) showed Sinus Rhythm with global T wave inversion and prolonged QT (QTc:546 ms); there was no previous ECG to compare (Figure 1). Investigation showed low Adjusted Ca (1.89 mmol/L), low Potassium (3.1 mmol/L), normal Magnesium (0.68 mmol/L), normal Sodium (143 mmol/L), elevated TSH (11.49 mlU/L), and low Free T4 (9.0 pmol/L) (Table1). Electrolyte correction took place with intravenous (IV) Calcium Gluconate (10%) 1000 mg and oral Calcium Carbonate Tablet 1200 mg three times daily Levothyroxine 125 micrograms oral once daily was also resumed.
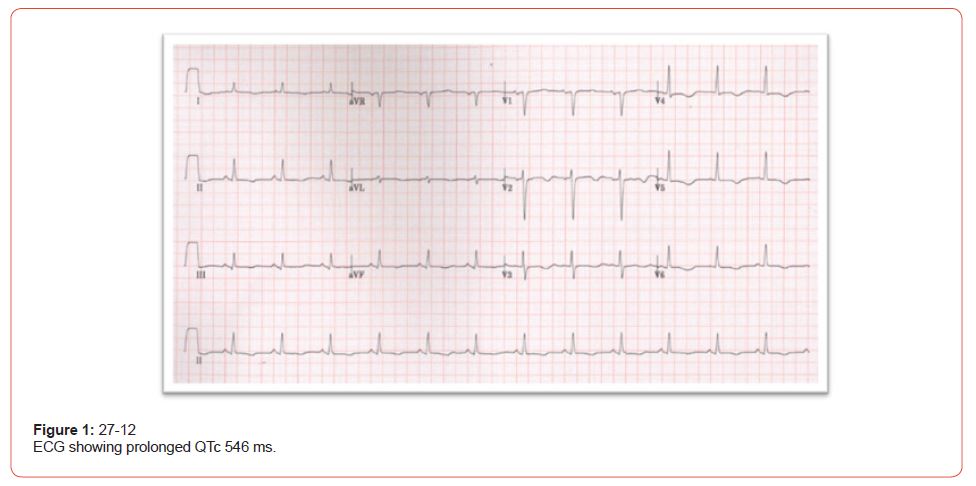
Table 1: Lab results.
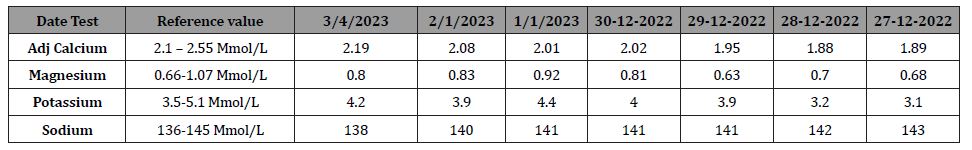
Two days later, the patient complained of palpitation; ECG was repeated and revealed Atrial fibrillation (AF) with prolonged QT (QTc 562) (Figure 2), Echocardiogram (ECHO) revealed mild Left Ventricular (LV) dysfunction, with an estimated Ejection fraction of 45%, mild Right Ventricle (RV) dilatation and Right Ventricular Systolic Pressure (RVSP) was 57mmhg, such parameters could be explained by the paroxysm of AF, causing mild impairment of LV systolic function followed by right side involvement. Holter’s finding also supports paroxysmal AF with short runs of supraventricular tachycardia (SVT)/AF. The patient was started on heart rate control medication Bisoprolol 2.5mg and anticoagulants. Repeated labs showed persistent hypocalcemia and hypomagnesemia(Adjusted Calcium 1.95 mmol/L and Magnesium 0.63 mmol/L), while Potassium was normal (3.9 mmol/L). An aggressive replacement was commenced with a goal for Potassium to be more than four mmol/L, Magnesium to be more than one mmol/L, and adjusted calcium to be 2 to 2.2 mmol/L to stabilize the cellular membrane potential and prevent arrhythmia.
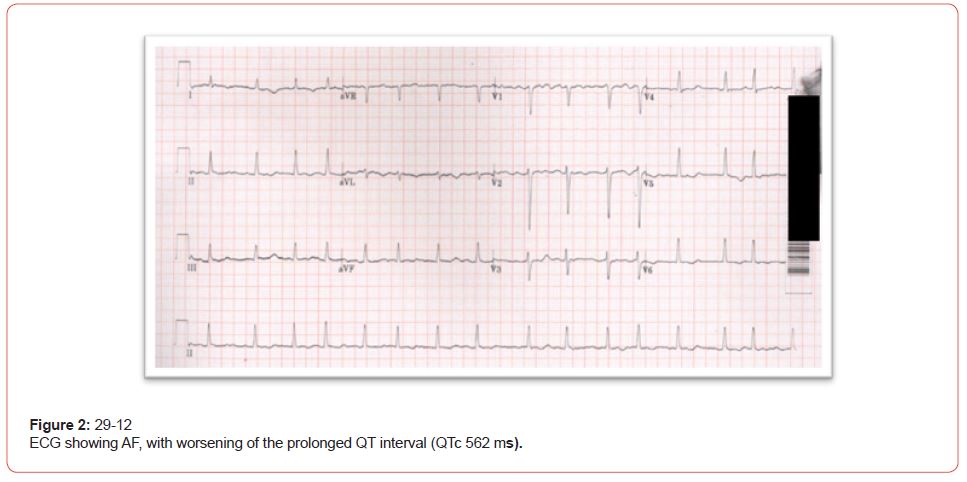
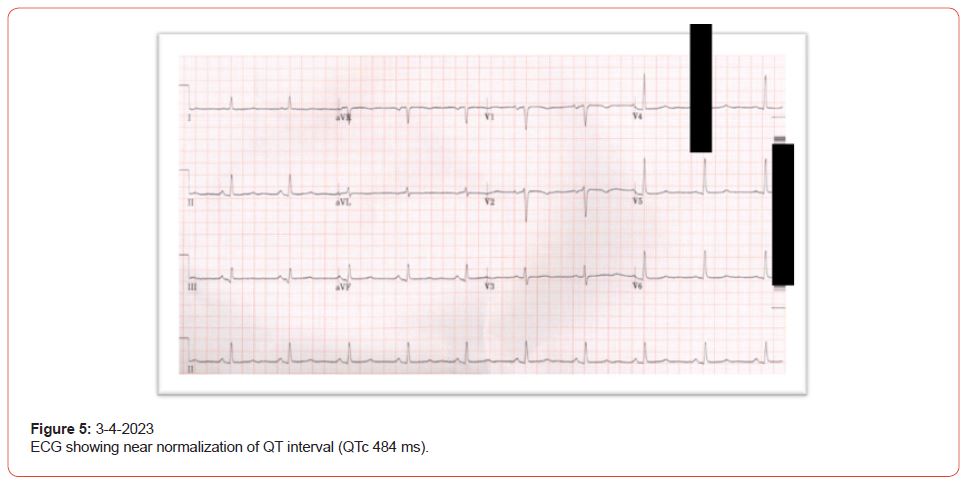
Three days post-admission to our hospital, the patient was stable; palpitation improved, and repeated labs showed; Adjusted Calcium 2.02 mmol/L, potassium 4.0 mmol/L, within target level, while magnesium at 0.82 mmol/L still below target. After the replacement of electrolytes, there was a gradual improvement in QTc duration, ECG showed (QTc 532) and AF (Figure 3). The cause of cardiac arrest was assumingly due to acquired prolonged QT interval secondary to hypocalcemia and hypomagnesemia, lack of immediate complete resolution of prolonged QT after correction of hypocalcemia and hypomagnesemia was thought to be due to under-replaced hypothyroidism as evident by the thyroid function. This assumption was confirmed 3 months later as QT interval became near normal after replacement of hypothyroidism (TSH was 1.9 mlU/L) (Figure 4 & 5) (Table 2).
Table 2: corrected QT interval.

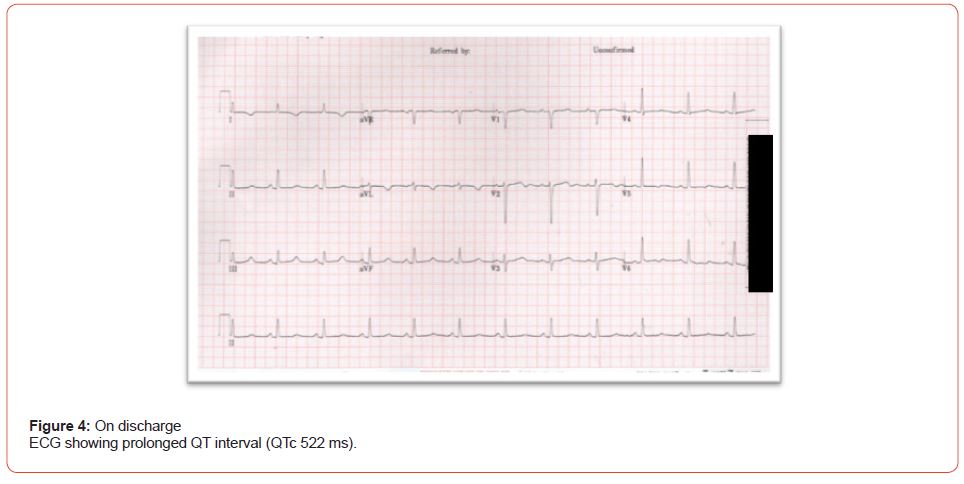
As she had a low CHA2DS2VASc score, there was no need for lifelong anticoagulation; she was discharged home on Calcium carbonate 1200mg three times per day orally, Alfacalcidol 1mcg orally once daily, levothyroxine to 150mcg orally once daily, and Cholecalciferol 50000IU twice per month. An implantable cardioverter defibrillator (ICD) for secondary prevention was considered as her adherence to electrolyte replacement as an outpatient was questionable, placing her at risk of tachyarrhythmia, Ventricular Tachycardia, or cardiac arrest. However, prior to discharge, the patient was educated about her diagnosis and the importance of strict adherence to medication to prevent such complications, which can be life-threatening in addition close follow-up with endocrinology and cardiology was arranged.
Discussion
The QT interval is prolonged either due to congenital or acquired causes; it is linked to a high incidence of malignant ventricular arrhythmias, which are characterized by rapid, irregular tachycardias that usually have a polymorphous configuration or a torsades de pointes pattern (TdP). Individuals with QT prolongation experience symptoms that range from dizziness to syncope and sudden death [1]. In our case, the patient presented with the worst presentation, which was cardiac arrest. On the other hand, there are other cases where the patients present with milder symptoms, like in Sukhjinder Nijjer study, the patient presented with five days history of palpitations and dyspnoea with no syncopal attacks [5]. Drugs, toxins, severe bradycardia, cardiac ischemia, and electrolyte imbalance are acquired causes of long QT syndrome (LQTS) [4]. This case report emphasizes the “worst-case scenario” of electrolyte disturbance (hypocalcemia, hypomagnesemia), which resulted in acquired long QT syndrome and cardiac arrest. Calcium plays many roles in the neuromuscular processes; hypocalcemia affects the cardiovascular system’s myocardial contractility and prolongs the QT interval, predisposing to ventricular arrhythmias [5]. A well-known cause of hypoparathyroidism and concomitant hypocalcemia is thyroid surgery due to parathyroid injury or removal [6]. Patients after thyroid surgery should have a longterm follow-up to reassure treatment adherence and to prevent such complications, like in our case where the main reason for the patient presentation was presumed to be poor adherence to hypoparathyroidism treatment which led 19 years later to hypocalcemia, followed by sudden cardiac arrest. In another case, hypocalcemia due to hypoparathyroidism occurred 23 years after the initial thyroid surgery [5]. It is possible that the presence of more than one cause of acquired LQTS creates a synergistic effect and predisposes the patient to the worst-case scenario i.e. cardiac arrest, in our case, hypocalcemia, hypomagnesemia and inadequately replaced hypothyroidism.
The principal management of patients with acquired LQTS is identifying and correcting the underlying cause by holding any precipitating drug or aggressive correction of any electrolyte imbalance such as hypomagnesemia, hypokalemia, hypocalcemia, or all, as in our case. Furthermore, medications that could prolong QT interval should be avoided [1]. The patient should be monitored closely till electrolytes are normalized and the QT interval returns to normal. In our case, the lack and delay of resolution of prolonged QT interval were due to suboptimal hypothyroidism replacement, which masked the immediate normalization of the QT interval on ECG with the rapid correction of electrolytes. It is well known that uncontrolled hypothyroidism can cause abnormalities in cardiac function like arrhythmias, prolonged QT interval, and in rare cases, torsade de pointes [7]. Therefore, it is important to search for all the factors that could lead to prolonged QT interval and correct it to get optimal results.
β-blockers are the primary therapy for the long-term management of congenital LQTS. Unfortunately, even when patients adhere to their prescribed β-blockers, the risk of cardiac events continues, particularly in symptomatic patients. Lack of adherence to β-blocker and using drugs that could prolong QT interval is the responsible cause for almost all life-threatening conditions in patients with LQTS; therefore, physicians tend to consider implantable cardioverter defibrillators (ICD) [8]. According to a recent study, ICD is indicated only for patients with congenital LQTS [9]. However, there is controversy about the long-term indication of ICD in patients with acquired LQTS who also had torsades de pointes and ventricular fibrillation, as currently there are no guidelines to use ICD in patients with acquired LQTS, partly because acquired LQTS is potentially reversible once the cause is corrected [10]. Yet, patients who survived cardiac arrest with acquired LQTS seem to be at ongoing risk of other life-threatening events after the first episode of cardiac arrest [11]. In Gerold Monnig’s study, during a 7-year follow-up, 44% of patients with acquired LQTS who had ICD implantation received appropriate ICD shocks that prevent lifethreatening conditions [12]. Therefore, we conclude that ICD can be secondary prevention and life-saving for patients with Acquired LQTS in addition to correcting the underlying cause.
Conclusion
In conclusion, diagnosis of acquired prolonged QT interval can be challenging when the main presenting symptoms are life-threatening arrhythmias or, in rare cases, like in our case, the patient could present with sudden cardiac arrest. Moreover, many causes could lead to acquired prolonged QT interval, like anti-arrhythmic medications, electrolyte imbalance, or endocrine disorders. A high index of suspicion is required as patients who were stable on treatment for many years can present with complications. Identifying prolonged QT interval on ECG is not enough for management and we have to search for the underlying cause so life-saving appropriate treatment and prevention can be provided. Prevention of symptoms is also challenging since there are no sufficient studies to support ICD implantation in patients with acquired prolonged QT syndrome, and it primarily depends on patient’s adherence to medications; therefore, education and patient engagement are crucial.
Take away message
1. Serious complications like cardiac arrest and ventricular arrhythmias could occur from acquired causes of prolonged QT syndrome and not only congenital causes.
2. Not only electrolyte imbalances like hypomagnesemia lead to LQTS, but hypocalcemia due to acquired hypoparathyroidism can lead to acquired LQTS with life-threatening complications, i.e., cardiac arrest even years after diagnosis.
3. Management of Acquired prolonged QT syndrome is by treating the underlying cause. However, ICD implantation can be lifesaving, although guidelines are lacking.
Study limitation
There was no baseline ECG to compare with and confirm the diagnosis during the first presentation.
Ethical approval
The study was approved by King Abdullah International Medical Research Center (KAIMRC), reference number NRA23A/013/04
Acknowledgement
None.
Conflict of Interest
No conflict of interest.
References
- Charles I Berul (2023) Acquired long QT syndrome: Clinical manifestations, diagnosis, and management.
- Moss AJ (1986) Prolonged QT-Interval Syndromes. JAMA: The Journal of the American Medical Association 256(21): 2985.
- Escande D, Drici MD, Barhanin J (2001) Molecular Basis of Inherited Long QT Syndromes and Cardiac Arrythmias. Heart Physiology and Pathophysiology pp. 1097-1105.
- Moss AJ (2003) Long QT Syndrome. JAMA 289(16): 2041-2044.
- Nijjer S, Ghosh AK, Dubrey SW (2010) Hypocalcaemia, long QT interval and atrial arrhythmias. BMJ case reports 2010: bcr0820092216.
- Tredici P, Grosso E, Gibelli B, Massaro MA, Arrigoni C, et al. (2011) Identification of patients at high risk for hypocalcemia after total thyroidectomy. Acta Otorhinolaryngol Ital 31(3): 144-148.
- Klein I, Danzi S (2007) Thyroid disease and the heart. Circulation 116(15): 1725-1735.
- Cho Y (2016) Management of Patients with Long QT Syndrome. Korean circulation journal 46(6): 747-752.
- Priori SG, Wilde AA, Horie M, Cho Y, Behr ER, et al. (2013) HRS/EHRA/APHRS expert consensus statement on the diagnosis and management of patients with inherited primary arrhythmia syndromes: document endorsed by HRS, EHRA, and APHRS in May 2013 and by ACCF, AHA, PACES, and AEPC in June 2013. Heart rhythm 10(12): 1932-1963.
- Kallergis EM, Goudis CA, Simantirakis EN, Kochiadakis GE, Vardas PE (2012) Mechanisms, risk factors, and management of acquired long QT syndrome: a comprehensive review. ScientificWorldJournal 2012: 212178.
- Mullally J, Goldenberg I, Moss AJ, Lopes CM, Ackerman MJ, et al. (2013) Risk of life-threatening cardiac events among patients with long QT syndrome and multiple mutations. Heart rhythm 10(3): 378-382.
- Mönnig G, Köbe J, Löher A, Wasmer K, Milberg P, et al. (2012) Role of implantable cardioverter defibrillator therapy in patients with acquired long QT syndrome: a long-term follow-up. Europace 14(3): 396-401.
-
Maryam Al Arfaj1, Ali AL Qarni1,2,3, Muneera AlTaweel1,2* and Abdulrahman Al Omair1. The Worst-Case Scenario of Electrolytes Imbalance, Cardiac Arrest, and Acquired Prolonged QT Interval: A Case Report and Literature Review. On J Cardio Res & Rep. 7(3): 2023. OJCRR.MS.ID.000662.
-
QT prolongation, Electrolytes imbalance, Cardiac arrest, Electrocardiogram, Anti-arrhythmic, Electrolyte imbalance, Endocrine disorders, Breath, Cardiovascular, Chest, Abdomen, Supraventricular tachycardia, Hypothyroidism, Tachyarrhythmia, Ventricular tachycardia
-

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.






