 Research Article
Research Article
Synthesis, Characterization and Applications of Some Transition Metal Complexes of Nucleic Acid Constituents
MS Masoud 1*, M Sh Ramadan1, A M Sweyllam2 and M H Al-Saify3
1Department of Chemistry, Alexandria University, Egypt
2Department of Physics Department, Alexandria University, Egypt
3Sidi Kerir Petrochemicals Company, Egypt
Mamdouh S Masoud, Department of Chemistry, Faculty of Science, Alexandria University, Alexandria, Egypt.
Received Date: October 15, 2020; Published Date: January 27, 2021
Introduction
Molecular biology is an important in understanding the interactions between the various systems of a cell, including the interactions between deoxyribonucleic acid and ribonucleic acid, DNA and RNA, respectively and protein biosynthesis, (Figure 1). Bioinformatics or computational biology, is the interdisciplinary research field integrating biology with informatics, and is expected to a huge impact on the bioscientific, bioengineering and medical fields. There are many techniques in bioinformatics for DNA microarray data; how ever, these are mainly divided into fold-change analysis, clustering, classification, gentic network analysis, and simulation [1-3]. The introduction of 2H-, 13C-, and 15N-labeled oligonucleotide building blocks became paramount for structure elucidation of RNA and DNA molecules [4-8]. The pyrimidines (1) and purines (2) are of great importance [9-16], where in our laboratory, numerous papers have been published from the structural and coordination chemistry views [17-87] (Figure 2).



The pyrimidine nucleus is embedded in a large number of alkaloids, drugs, antibiotics, agrochemicals, and antimicrobial agents [88]. Many simple fused pyrimidine such as purines and pteridines are biologically active by themselves, or are essential components of very important naturally occurring substances (i.e., nucleic acids). Some pteridine derivatives are also used as anti-leukemic drugs [89], or potassium-conserving diuretics. Some fused thieno[ 3,2-d] pyrimidines serve as anti-allergy drugs, some act as fungicides. A very important biologically active pteridine system (fused pyrazino[2,3-d] pyrimidine) is present in folic acid (3) and several antibiotics. Pteridine was also found in riboflavin (6,7-(dimethyl- 9-(D-1-ribityl) isoalloxazine, vitamin B2(4), a growth-regulator for microbes and animals (Figure 3). The formation of caffeine (7) in the reaction of a diaminopyrimidine derivative (5) with formic acid is described as follows (Diagram 1) [90,91]. The chemistry, biological activity, and physical properties of fused pyrimidines are thoroughly reviewed in many articles and monographs [9,10,90]. Generally –ortho- amino(aminomethyl)aromatics would be the best starting materials for their synthesis, (Figure 4).
The manifestation of the purine system in natural products has been reviewed [92]. Fluorouracil (8)[93] or the human immunodeficiency virus (HIV) drug zidovudine(9) [94] compounds are of biological interest in chemotherapy. Ultrashort-acting barbiturates such as thiopental sodium(10)(Pentothal) [95] are often used as general anesthetics, whereas methylphenobarbital (11) [96] is used as antiepilepticum. Some diaminopyrimidines, such as pyrimethamine (12) [97] or trimethoprim (13) [98] are powerful antimalaria drugs used in combination with sulfonamides, (13) is also a potent antibacteriostaticum, whereas minoxidil (14) [99] is used as antihypertensivum. Sulfadiazine (15) [100] is one of the chemotherapeutics containing a pyrimidine moiety (Figure 5) [101].


Cisplatin, as one of the leading metal-based drugs, is widely used in the treatment of cancer. Significant side effects and drug resistance, however, have limited its clinical applications. Biological carriers conjugated to cisplatin analogs have improved specificity for tumor tissue, thereby reducing side effects and drug resistance. Platinum complexes with distinctively different DNA binding modes from that of cisplatin also exhibit promising pharmacological properties [102]. Metal centers, being positively charged, are favored to bind to negatively charged biomolecules; the constituents of proteins and nucleic acids offer excellent ligands for binding to metal ions. The pharmaceutical use of metal complexes therefore has excellent potential [103]. A lot of different coordination compounds and the mechanism of cytotoxic action have been discussed with regard to the development of new antitumor agents. Developing metal complexes as drugs, however, is not an easy task. Accumulation of metal ions in the body can lead to deleterious effects. Thusbiodistribution and clearance of the metal complexes as well as its pharmacological specificity are to be considered. Metal ions and metal coordination compounds are known to affect cellular processes in a dramatic way. This metal effect influences not only natural processes, such as cell division and gene expression, but also non-natural processes, such as toxicity, carcinogenicity, and antitumor chemistry. In chemotherapy, the key issue is killing the tumor cells, without causing too much harm to healthy cells.
Nucleic acids (NA) and their metal interactions belong to an advanced branch of experimental bio-inorganic chemistry, accurate theoretical studies in this field have yet become possible in the last decade, especially due to the development of density functional theory (DFT)-based quantum chemical techniques [104]. Computational studies, the architectural, thermodynamic, and kinetic propertiesare able to provide a simultaneous picture of the structure, stability, function and electronic properties of the metal-NA interactions, which is important tounderstand the biochemical functions. It is pretty challenging to relate gas phase data to properties observable in physiological DNA and RNA. Site-specific binding of metal ions obeys the rules of coordination chemistry. Thus, metal cations have to be surroundedwith properly situated ligand field to provide a realistic picture of theirinteractions. This adds another level of complexity to the models used for these systems and shows that metal cations are more sophisticated entitiesthan to treat them as charged spheres.
Metal-Nucleobase Interactions
The nominal charge of the polynucleotide chain is contributed by thephosphate group. Nevertheless, nucleobases themselves are excellent N and O donor ligands, forming well-defined binding sites primarily for divalentcations. Under physiological conditions, i.e. in aqueous solution, metal-cations are always present in a hydrated form. Thus, the relevant model for the site-specific binding of metal cations to nucleobases must always include the hydration shell of the metal cation. Then, the hydrated metal cation mayinteract with the nucleobase either in the (i) outer shell, (ii) inner shell, and occasionally, (iii) bidentate fashions (Figure 6(a-d)). While the geometry is largely determined by the coordination chemistry of the cation, the binding strength depends on the ligand type. Nucleobases offer several binding sites of various interaction strengths. Thus, it is important to determine the binding strength depending on themetalation site for a given base and cation. Moreover, various cations exhibit different affinity towards a given type of binding.
The inner and outer shell bindingof hydrated Mg2+ and Zn2+ cations to guanine are studied [105]. The thermo dynamical driving force of cation binding was estimated on the basis of three different thermodynamic cycles. The computed results reveal that the N7 position ofguanine has a greater affinity towards Zn2+ than Mg2+, while the O6 positionexhibits a similar propensity towards both cations. For the outer shell binding the gas-phase results were not accurate enough to unambiguously show preference towards either cations. Decomposition of the interaction energiesinto pairwise terms has revealed that inner shell binding of hydrated zinc to guanine can be viewed as a process in which the base is firstmetalated and then hydrated, as the dominant part of the total stabilization is due to the interaction between the bare cation and the nucleobase. In contrast, for the analogous complex with magnesium the metal-base term is reduced with respect to zinc, and, therefore, the complex is shifted more towards a complex of nucleobase with hydrated cation (Figure 6).
Nuclear magnetic resonance (NMR) shifts and spin-spin coupling constants for the hydrated magnesium guanine system have been studied [106], where the intermolecular spin-spin coupling constants 1J(X,O6) and 1J(X,N7) (X = Mg2+, Zn2+) can be successfully applied to differentiate between outer shell and inner shell binding modes. In contrast to Mg2+, which has a clear preference towards the hexacoordinated form, in biological systems Zn2+ might be present also in tetracoordinated complexes. An in-depth database analysis reveals that, in general, Mg2+ is more susceptible towards O-donor ligands while Zn2+ rather favors N and S-donors. Mn2+ represents a borderline between Zn2+ and Mg2+(107)and has a more balanced affinity for nitrogen and oxygen ligands and a ligand exchange rate 100 times faster than Mg2+,also Mn2+ is used as aparamagnetic probe in electron paramagnetic resonance(EPR) as well as NMR experiments.
Cd2+ is a much softer metal ion and therefore much more likely to interact through innersphere contacts with nucleobasenitrogens. In addition, it also has a higher inclination to form macrochelates [108,109]. Cd2+ is significantly larger than Mg2+, but its thiophilicity [110] makes it for metal-rescue experiments with phosphorothioates despite a seriouscaveat. Zn2+ is also very thiophilic and more similar in size to Mg2+, but engages in variablecoordination geometries [111]. Tl+ has been employed as a substitute for K+, making use either ofits thiophilicity or of the abundant spin 1/2 nucleus of 205Tl+.
Ribozyme catalysis is in many cases promoted by a variety of divalentmetal ions or even monovalent ions alone [112], on the other hand so far only a limited subset of metalions have been shown to be specifically required in naturally occurring ribozymes, a fact thatseems surprising in light of the diverse metal ion binding capabilities and selectivities of variousin vitro selected ribozymes and DNAzymes. The latter have been shown to discriminate against Mg2+, while selecting for Ca2+, Cu2+, Co2+, Zn2+, Mn2+, Pb2+,Ni2+ or a small subgroup of transition metal ions [113-119].
Metal-Phosphate Interactions
The nominally -1 negative charge of each nucleotide unit in NA is carried by the phosphate moiety. To a large extent the charge of NAs is compensated by monovalent cations partly condensed on the surface of the macromolecule. However, that since polar solvent efficiently screens the negative charge of phosphates, the individual phosphates for most of the time do not interact directly with an ion. Divalent metal cations specifically bound to the phosphate groups may have important catalytic functions, they can catalyze the hydrolysis of the phosphodiester linkage in RNA. Crystal database analyses show, that in contrast to the analogous carboxylates, metal binding to the phosphinyl (-PO2 --) fragment of the phosphate group does not take place in a symmetric fashion: i.e. in most published crystal structures the electron density maxima are outside the bisector of the O = P = O angle [20]. A thorough ab initio analysis of variously charged mono- and diphosphate derivatives with monoand divalent cations has shown that the asymmetric coordination of the metal cation is likely to be caused by the direct participation of polar particles, such as water, in the cation binding [121]. Biologically relevant models must include the proper number of ligated water molecules to represent the hydration sphere of the cation. Metal catalyzed hydrolysis of the phosphodiester bonds in NA is a long-disputed problem of bio-inorganic chemistry. The study suggested, that the process involves the cleavage of the C5’-O bonds of the sugar rather than that of the P-O bonds of the phosphate [122]. It has been found that catalytic activity of a metal cation is due to its ability to (i) shield the negative charge of the phosphate group and (ii) simultaneously weaken the C-O bonds in the model compounds. The role of the metal center is twofold: (i) via its hydration shell it stabilizes theintermediates and transition state complexes formed, and (ii) donates aproton from its hydration shell to facilitate the loss of the leaving group inthe rate determining step of the reaction. The mechanism of phosphodiesterhydrolysis is strongly dependent on the local RNA architecture and often requires cooperation of remote segments of the macromolecule [123]. Sometimes, even two cations might be active in the process. Thus, exploring themechanism of phosphodiester hydrolysis in biologically relevant systems is avery intricate and computationally challenging task.
Metal-Nucleotide Interactions
Combination of the N- and O-donor sites of nucleobases with the negatively charged phosphate oxygens gives rise to versatile metal-binding modes in nucleotide structures. Due to their distinct biological relevance, characterization of metal-nucleotide interactions correlated with N7-binding inpurine nucleotides. Hydrated zinc and magnesium group divalent cations bind to the N7position of purine nucleotides to form very strong H-bonds between the cation and anionic oxygen atoms of the phosphate group (Figure 7a). There by the water shell exerts a screening of the negative charge concentrated on thephosphate moiety and at the same time influences the backbone geometry. Thepairwise nucleotide-cation interaction provides the major contribution to the stabilization of these complexes, which can be characterized with a high non-additivity (17-28% of the total interaction energies). The non-additivity term expresses the shielding of the metal-nucleotide electrostatic attraction by the water shell and is larger for Zn2+ than for Mg2+ [124].As non-additivity and polarization effects are absented from standardforce fields, the force fields are not suitable to capture the energetics of the metal - nucleotide interactions with a satisfactory accuracy. It is parameterized to describe interaction of hydrated Zn2+ and Mg2+cations with 5’-guanosine-monophosphate and properly includes polarization and charge transfer terms necessary to treat this kind of systems [125]. Due to the high non-additivities, the pairwise interaction energies are notrepresentative enough for the strength of the metal-nucleobase and metal-phosphateinteractions. In contrast, the geometrical parameters well reflectthe balance of all contributing effects and can be used to indirectly evaluatebinding selectivity in these systems. From systematic changes in theinteratomic distances, electronic energies and interaction energies, the bindingselectivity of divalent cations towards N7-binding varies in the followingorder: Cu2+>> Zn2+ = Cd2+> Mg2+. In fact, the binding selectivity is the result of the balance between the water - cation and cation - nucleobase contributions. The binding selectivity in biological (solution) systems is determined by pretty tiny energy differences (on the scale of 3-10 kcal/mol), while the absolute gas phase binding energies are around 300 kcal/mol. This underlines the accuracy of the methods needed to evaluate such systems [125]. The gas phase data are typically dominated by the electrostatics which is eliminated in polar environment. However, specific non-electrostatic effects well reflected by thequantum mechanics (QM) calculations are often fully transferred into solution [124-126]. Simultaneous inner-shell binding to N7(G) and the phosphate oxygen (Figure 7b) attenuates the water-phosphate contacts. On the other hand, inner shell binding to the phosphate does not change the above established ranking of cations related to their selectivity towards N7-binding [126]. It has been found that inner shell binding of guanine to hexahydratedcation-phosphate complexes substantially weakens the metal-phosphate outer shell binding. Thus, the phosphate group recognizes when water is replaced by guanine in the hydration shell of the cation. However, it is unable to recognize the Mg2+ to Zn2+ substitution, assuming the metals adopt the same coordination mode [126]. These findings are of key importance at unraveling the principles of metal-phosphate Citation: MS Masoud , M Sh Ramadan, A M Sweyllam, M H Al-Saify. Synthesis, Characterization and Applications of Some Transition Metal Complexes of Nucleic Acid Constituents. Insi in Chem & Biochem. 1(4): 2021. ICBC. MS.ID.000516. DOI: 10.33552/ICBC.2021.01.000516. Page 6 of 25 recognition in NAs. The different modes for binding of ions to DNA are represented in Figure 8 (Figure 7).
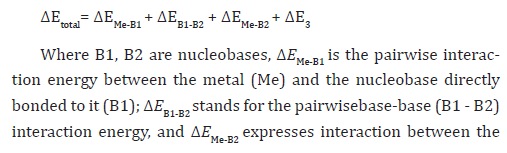
hydrated cation and the remote base(mostly long-range electrostatics). ΔE3 is the three-body(nonadditivity) term that reveals how the three interacting species cooperatewhen forming the complex, thus, polarization of the base pairing by the N7metal binding. ΔE3 and ΔEMe-B2 terms contribute to the enhancement of thebase pairing. Thepolarization enhancement (ΔE3) depends also on the polarity of the base pair, thus it is quite substantial for the GC and guanine-guanine (GG) base pairs and pretty small for the adenine-uracil(AU) pair. When the proximal base is replaced by a nucleotide, the metal binding is improved because of the favorable electrostatic contactsbetween the cation and the anionic phosphate group. On the contrary, thisreduces the three-body term.No significant differences were observed in the electron topology of theGC, GG and AU base pairs metalated with pentahydrated Mg2+ at N7. The polarization base pair enhancementupon cation binding is best interpreted on the basis of ‘’the electrostaticresponse of the complexes to the presence of the cation’’ [129].


Nucleic Acids and Metallodrugs
Introduction of cisplatin in cancer treatment ignited a series of experimental [130] and, later, theoretical studies aimed at describing its mechanism of action. Clinical application of platinum compounds is restricted due to their high cytotoxicity and the experimental research was recently shifted towards other less toxic DNA-binder metal-based drugs. These studies also benefit from findings of theoretical investigations on square planar platinum complexes,which are also of interest for the basic bioinorganic chemistry. The action of metal-based drugs, on the other hand, usually depends on the irreversible association of metal ion complexes to DNA. Following the historical discovery of the anticancer properties of cisplatin, cis-(NH3)2PtCl2 [131], many other Pt2+, but also Pt4+, Ru3+, Ga3+ and Ti4+ complexes have been investigated with regard to their anticancer activity [132].
Platinum Coordination Compounds
Structure-activity relationships for a class of platinum coordination compounds confirmed that only those compounds having cisgeometry block cell growth. The most active complex, cisplatin (16), (Figure 9) exhibits antitumor activity, whereas its trans isomer showed no such activity. Many derivatives of cisplatin also inhibit growth, and these compounds have at least one N-H group, which is responsible for important hydrogen-bond donor properties, either in the approach of the biological target or the final structure. Most of the well-known platinum anticancer complexes have the general formula cis-[PtX2(NHR2)2], in which R = organic fragment and X = leaving group, such as chloride or (chelating bis) carboxylate. So, Pt is most useful clearly relates to ligand-exchange kinetics, which is quite slow for Pt compounds, affording them a high kinetic stability and results in ligand-exchange reactions of minutes to days, rather than microseconds to seconds for many other coordination compounds. Pt(II) has a strong thermodynamic preference for binding to S-donor ligands. For that reason, one would predict that platinum compounds would perhaps never reach DNA, with many cellular platinophiles (S-donor ligands, such as glutathione, methionine) as competing ligands in the cytosol. The so-called kinetic trans effect should be mentioned, which is responsible for ligand-exchange reactions on metalsions. The rule can be quite simply formulated as: ligands located trans to another ligand with a strong trans effect (such as many soft ligands) are more rapidly substituted than ligands in cispositions. Despite the success of cisplatin, however, it lacks selectivity for tumor tissue, which leads to severe side effects. These include renal impairment, neurotoxicity and ototoxicity (loss of balance/hearing), which are only partially reversible when the treatment is stopped. With long-term or high-dose therapy, severe anemia may develop. Modified versions of cisplatin, leading to second and third generation platinum-based drugs have been synthesized over the past 30 years. Several platinum complexes (17-23), (Figure 9) are in clinical trials, but some of these new complexes have not yet demonstrated such significant advantages over cisplatin. The second-generation platinum drug carboplatin, [Pt(C6H6O4) (NH3)2](17), has fewer toxic side effects than cisplatin and is more easily used in combination therapy. Its low reactivity allows a higher dose to be administered. Carboplatin is used more for ovarian cancer treatment. Cis-aminedichloro(2-methylpyridine)platinum( II) (ZD0437)(19) derivatives have improved antitumor activity whereas oxaliplatin(21) is known to be most effective in colon cancer treatment. The so-called JM-216(22), a Pt(IV) compound, has been in routine clinical use as an orally administered drug. A representative trinuclear complex, BBR3464 (23), has entered a phase II clinical trial and exhibits activity against pancreatic, lung and melanoma cancers (Figure 9).
Combining drugs with different modes of action often synergizes their effects, so the scientists will continue to probe the various metallopharmaceutical mechanisms in the hope that together they might yield an even wider range of effective chemotherapeutic agents [133,134]. Combination therapy could be the way forward in the fight against cancer.The anticancer efficacy of cisplatin is also influenced by the efficiency of cisplatin-DNA adduct removal by the cellular repair machinery, with nucleotide excision repair being a major pathway. The repair of platinum-DNA crosslinks is retarded when the DNA is bound to the histones in a nucleosome core particle [135,136]. The human mismatch-repair protein, hMSH2, also binds specifically to DNA containing cisplatin adducts and displays selectivity for the DNA adducts of therapeutically active platinum complexes [137]. So, hMSH2 mediates cisplatin toxicity. Contribution of individual adducts to antitumor activity and toxicity of platinum complexes still remains to be examined. The formation of adducts following the reaction of cis-diaminedichloroplatinum(II) (cisplatin) with various DNA nucleotides was investigated [138]. Formation and conversion of distinct species were confirmed. The potential applications comprised studies of novel platinum complexes, investigations of platinum-adduct formation with DNA, and determination of platinum-DNA adducts in cells [139].
A systematic study on the proton affinities of platinated adenines hasrevealed that the gas-phaseprotonation energies depend primarily on theoverall charge and to a much lesser extent on the location of the metal [140]. Thecharge-dependence is enormous, yet it appears to be completely abolished inaqueous solution, where the corresponding pKa values are essentiallyindependent on the charge of the metal adduct in the range of charges +3 to –1.In contrast, the subtle variations in the interplay between different cationbinding and protonation sites stem from molecular orbital effects andappears to be translated from the gas phase to solution. (Figure 10) summarizes the computed gas-phase proton affinities as a function of the total charge as well as the position of the proton and platinum moiety. For a given platinum and proton position one can recognize an almost linear relationship between the proton affinity and the charge of the platinum adduct. For a given total charge of the metal adduct the maximum is reached if the platinum is at N7 and the proton binds to N1. This is in qualitative agreement with experimental observations from solution studies and at the same time illustrates that site-dependence of theprotonation energy stems from molecular orbital effects, because it is independent on the total charge of the metal-adduct (Figure 10).
The improvement of the protonation energy is larger for the N4-platinated cytosine than for N6-mercurated adenine (Figure 11(a-b)) and can be correlated with the shift of the π-electron density from the nucleobase to the metal center in the major tautomeric forms. Metal-induced shift of the protonation equilibria and thereby stabilization of the rare tautomeric forms may give rise to stabilization of mispairs in DNA [141]. Stabilization of the N1-protonated form of adenine by N6-metalation with Hg2+ gives rise to the formation of stable AH+.C and AH+.G base pairs. Interbase proton transfer processes may also be accelerated by amino-platination. For example, proton transfer from the N1 position of guanine to N3 of cytosine results in the formation of a CH+.G- ion pair. The energy difference between the ion pair and canonical forms of this base pair is 24 kcal/mol in the gas-phase. Due to the stabilization of the protonated (i.e. “rare tautomeric”) form in N4-platinated cytosines, however, this energy difference can be reduced to the half by metalation [140]. N7-metalation also changes the gas-phase tautomericequilibria of purine bases. However, the effect vanishes for neutral metal adducts [142]. Charged metal adducts stabilize the major (amino) and minor (imino) forms of guanine and adenine, respectively (Figure 11(c- d). As the shift of tautomericequilibria in these gas-phase structures is due to electrostatic effects, they are not expected to occur in solution or crystal (Figure 11).
Studies on Cisplatin Binding to Nucleobases
Enhancement of the base pairing by N7 binding of [Pt(NH3)3]2+ is much larger than that caused by the inner shell binding of hydrated Mg2+ or Zn2+cations [142]. The electronic changes stimulated by Pt-coordination in the structure of nucleobases implicate even more pronounced changes in the stability of platinated base pairs. A correlation has been found between the N1-acidity of N7 platinated guanines and the strength of the GC base pairs in the gasphase [143]. The enhancement of the base pair is caused primarily by polarization effects, while the N1-acidity is mostly influenced by ionic-electrostatic forces with some additional polarization effects. While the computed gas-phase N1-deprotonation energies can be correlated with the solution pKavalues [144]. Similar observations were made for the AT Hoogsteen pair platinated at N1(A)-position [145]. While the computed interaction energy for the AT Hoogsteen pair is –14.1 kcal/mol,platination enhances its strength to –18.8 kcal/mol, primarily due to the long-range electrostatic attraction between the charged Pt2+ cation and thymine. When including, however, a nitrate anion coordinated in pseudoaxial position to Pt2+ (Figure 12) the base pair stability dropped down to –11.6 kcal/mol, because of the compensation of the charge on Pt2+. This suggests that the buffer anion determines the experimental outcome (Figure 12).
Cisplatin is the first anticancer drug successfully employed in the clinical treatment of various kinds of cancers, such as small cell lung, ovarian, testicular, head and neck tumors. From chemical point of view it is a squareplanar molecule (Figures(13-14)) whose hydrolysis results in the formation of the active form suitable to bind to DNA [146,147]. Cisplatin binds to the G-reach segments of DNA in such a way to form intrastrand cross-links between two adjacent guanines. The antitumor activity of the drug is associated with the bending of the DNA as a consequence of the cross-link formation. The binding was modeled by classicalmolecular dynamics (MD)simulation [148]. In fact, the binding is a ligand exchange reaction between the hydrated form of cisplatin and guanine, in which the water ligand is replaced by the nucleobase. The ligand exchange reaction proceeds via a trigonalbipyramidal transition state and is kinetically controlled. A comparative study on cisplatin binding to guanine and adeninehas shown, that for guanine (Figure 15), the binding process is energetically more favored both from kinetic and thermodynamic points of view [149]. The extrastabilization of the transition state with guanine is attributed to (i) a verystrong H-bond between the ammine ligands of the Pt-moiety and O6 ofguanine as well as (ii) the significantly stronger electronic interactionbetween the Pt and the guanine ligand as compared to that between Pt andadenine. In addition, the diaqua complex has been found to exhibit a higherselectivity towards guanine.There are controversial views on the role of π-back-donation in cisplatin- nucleobasebinding [149]. A study on hypothetical square planar Pt-complexescontaining CO-ligands concludes that, even with the strongest π-acids, π-back-donation may operate exclusively in the zero oxidation state. Thus, Ithardly has any role in stabilizing the cisplatin-nucleobase adducts. Inaddition, it is suggested, that non-coplanarity of the cisplatin and nucleobasemoieties also impedes the π-interactions in these systems. On the contrary, arecent orbital analysis of guanine complexes cross-linked with cisplatinreports on a rare binding combination between the dxy atomic orbital of Ptand the antibondingπ*-orbital of guanine [150]. In complex biological matrices, such as the cell, there are severalpotential targets of cisplatin binding. In fact, Pearson’s theory predict alarger binding affinity of cisplatin towards S-donor ligands [151]. A comprehensive theoreticalrationalon the competition of S- and N-donor ligands in cisplatinbindinghas been given by Deubel [151,152]. From an analysis of the Pt-L binding energiesin various [Pt(NH3)3L]2+ (L = ligand) complexes,the intrinsicbinding affinity of cisplatin is higher towards N-donors than S-donors in thegas-phase. However, solvation effects, particularly in polar solvents,strongly alter the gas-phase trends, and eventually in water S-donors becomemore favorable targets than the N-donor ligands.The kinetic control of cisplatin binding is determined by three factors: (i) the nucleophile, (ii) the substituents of the nucleophile, (iii) the environment, i.e. salvation effects (Figure 13-15).
Besides cisplatin, the second-generation drug, carboplatin (diamine[ 1,1-cyclobutanedicarboxylato(2-)]-O,O´-platinum(II)) (17), Figure(9) has been introduced into oncotherapy. The observed pharmacokinetic differences between cisplatin and carboplatin depend primarily on the slower rate of conversion of carboplatin to reactive species. Studies on the interaction of carboplatin with DNA indicate that the reaction proceeds via ring-opening in carboplatin and subsequent binding with DNA constituents. Replacement of the chloride groups in the cisplatin molecule by cyclobutanedicarboxylate ligand significantly diminished the nephrotoxic effects of the formed carboplatin, without affecting its antitumor potency. The synthesis of “the second generation platinum drugs” with improved toxicological profiles and “third generation drugs” overcoming cisplatin resistance.








Based on mechanistic findings, coordination chemists are designing and synthesizing new compounds. A novel DNA binding metal compound with antitumor activity and clinical efficacy must fulfill the following key requirements: (1) good intrinsic properties, including saline solubility and enough stability to arrive intact at the cellular target; (2) efficient transport properties in blood and through membranes; (3) efficient DNA-binding properties but slow reactivity with proteins; (4) the ability to differentiate between cancerous and normal cells; and (5) activity against tumors that are, or have become, resistant to cisplatin and derivatives. This latter requirement usually implies a structure that is distinct from cisplatin- type species.
Non-Platinum Anticancer Agents
Titanium complexes such as Titanocene dichloride had been recognized as active anticancer drug against breast and gastrointestinal carcinomas [153]. Gold complexes also show anticancer activity. The target site of Au complexes is mitochondria not DNA. Certain gold complexeswith aromatic bipyridyl ligands have shown cytotoxicity against cancer cells [154]. The 2-[(dimethylamino) methyl] phenyl gold(III) complex has also proven to be antitumor agent against human cancers [155]. Gold nanoparticles when used in combination with radio therapy or chemotherapy enhance DNA damage and make the treatment target specific [156]. Lanthanum has also been used to treat various forms of cancer [157]. Some complexes of Mn(III)induce tumor selective apoptosis of human cells are studied [158]. Many complexes were studied which showedanti-proliferative effects in human ovarian cancers. Ruthenium complexes with oxidation state +2 or +3 showantitumor activity against metastasis cancers. Ruthenocenederivatives act as anti-estrogen [153]. The relative binding of ruthenocene derivatives were very high and even better than hydroxyl tamoxifen which is novel antagonist for estrogen [159]. Ruthenocene complexes with aromatic ligands represent a relatively new group of compounds with antitumor activity. Ru(III)-(imidazole, indazole and arene) complexes exhibit anticancerproperties [153,160]. Ru thenocene-cymene complexes have shown to damage DNA by forming monofunctional adducts selectively with guanine bases [161]. Many of Ru complexes exhibit anti-estrogen properties similar to that observed for novel anti-estrogen Tamoxifen [162]. Ferrocifenesexhibit anticancer activity against hormone dependentand hormone independent breast cancers [153]. The ferrocene derivatives having hydroxyl group inphenyl ring and have high affinity for estrogen receptor.Many organometallic analogues of tamoxifen used as avehicle for introducing other cytotoxic agents to thecancer cells [163]. Normally, cancers are diagnosed at a stage of thedisease when some anatomical changes occur in thebody in the form of well defined tumors. These massescan be removed by surgery however this therapy is notsuitable for treatment of small or hidden tumors. Nanotechnologyoffers potential solutions to this problem forthe treatment of various types of cancers. Silica gold nanoshellstechnology for thermalablative therapy of cancer are used [164]. Nanoshells absorb light in the near infrared which can beused to deliver a therapeutic dose of heat by usingmoderately low exposures of extra corporeally appliednear-infrared (NIR) light. It has been reported that silver nanoparticles exhibit anti-proliferativeactivity [153]. Several examples ofnanoshell-based diagnostic and therapeutic approachesincluding the development of nanoshellbioconjugates formolecular imaging are described [165]. Mercaptopurines are well known anti leukemic drugs but their use has been hampered by theirshort half life. This problem has been overcome by theuse of gold nanoparticles in combination with mercaptopurines. Conjugation therapy of 6-mercaptopurine with goldnanoparticles not only enhanced its anti-leukemic andanti-inflammatory activity but also reduces the quantity ofdose and side effects of the drug [166].
A lot of new non-platinum metal-based anticancer complexes have been developed,especially those of V, Mo and Sn. Biological studies have shown that DNA is the primary cellular target for these metal-based drugs [167-169]. Intensive efforts of various research groups in recent years have contributed significantly toward an improved understanding of the mechanism of the antitumor activity of platinum complexes [170-172]. However, significantly less is known about the antitumor mechanisms of non-platinum metal-based complexes. The aqueous coordination chemistry of non-platinum metal-based anticancer complexes with nucleotides and their DNA binding modes have great importance and is valuable in understanding the mechanism of antitumor activity, as well as laying the foundation for rational design of more active and less toxic metal drugs. The reactions of some typical antitumor agents, Cp2TiCl2 (Cp=η5-C5H5), Cp2ZrCl2, cis-RuIICl2(DMSO)3(DMSO)( cis-RDT),(CH3)2SnCl2, (C2H5)2SnCl2 and (C2H5)2SnCl2(phen) (phen=Phenanthroline), with DNA and its components, and their coordination chemistry investigations in aqueous solution with nucleotides, as well as their bindings to DNA under near physiological condition were studied. The probe, [trans-en2Os (η2-H2)]2+,bind readily to a variety of biomolecules [173] such as nucleotides with ligand, L, substituted by these biomolecules, that results in the direct coordination of OsII to the donor atoms(such as N,O) of the biomolecules (Diagram 2).
When 2’-deoxyguanosine-5’-monophosphate (dGMP) was added to solution of the probe in D2O at 25°C each solute at 0.01 mol/L, the phosphate oxygen and N7 of dGMP both coordinated to the probe, the former being dynamically preferred(K=3×102) and the interaction was complete after 10 min, while the latter is thermodynamically preferred (K=2.9×103) and binding was complete after 24h.UV, circular dichroism(CD), fluorescence spectra and cyclic voltammetry( CV) are used for investigation of the interaction of two typical metal complexes, Cp2TiCl2 and Et2SnCl2, with calf thymus DNA and salmon DNA. There are two types of binding sites between DNA and Cp2TiCl2, one is the phosphate group of DNA and the other is the base nitrogen rings of DNA whereas only the phosphate group on DNA can binds to Et2SnCl2.The Cp2TiCl2 and (cis-RDT) are binded to dGMP(orDNA) by the metal center coordinating with the phosphate oxygen and N7 of dGMP, and the binding modes of Cp2ZrCl 2, (C2H5)2SnCl2 and (C2H5)2SnCl2(phen) are similar, they all only bind to dGMP (or DNA) by the metal center coordinating with the phosphate oxygen. It is very interesting to relate the binding modes with their anticancer activities.Cp2TiCl2 and cis-RDT are highly active anticancer agents with a wide antitumor spectrum, while Cp2ZrCl 2, (C2H5)2SnCl2 and (C2H5)2SnCl2(phen) are not good anticancer agents, (C2H5)2SnCl2and (C2H5)2SnCl2(phen) can only inhibit P388 cell growth and Cp2ZrCl2 has no activity at all. Studies on the binding modes of other high active anticancer metal agent, such as Cp2Mo- Cl2, also revealed that it can bind with phosphate group and base N7 of DNA or nucleotides [174,175]. Although binding of metal ions to the sugar ring oxygen atoms has rarely been observed. The role of phosphate under physiological conditions seems to be only secondary for coordination; nevertheless, it is quite important for hydrogen bonding, and the binding between metal ion and phosphate might be the first step of metal agents-DNA(or nucleotides) interactions, just as in the case of Cp2TiCl2-DNA interaction. Interactions between antimetastatic agents trans-RuCl2(DMSO)4 and 5’-dGMP also forms N7, α PO chelation [176]. So that the binding modes with DNA of a metal complex seem play a key role in its anticancer activity. It seems all highly active anticancer metal drugs have the ability to bind with both phosphate group and nitrogen sites on bases of DNA. The chemical activities of a compound are determined by its molecular structure. So further analysis is required for the relationships between the structure of metal anticancer agents, the binding modes with DNA and their anticancer activities.
Current and Future Developments
Possibilities for research in the coming decade with new techniques, which follow the reactions of Pt complexes and nucleic acids and proteins, will allow the detection of otherwise invisible intermediate products. The need for new platinum antitumor drugs was underscored by the usefulness of cisplatin and carboplatin in chemotherapy and the resistance of many tumors to these compounds. Combinatorial chemistry could aid in the search for cisplatin analogs if fast, high-throughput assays were available. The goal is to develop rapid cell-based assays suitable for high-throughput screening that accurately predicts the cytotoxicity of platinum complexes. The next stage in drug design is likely to be the development of dedicated drugs that comprise the transport (through the membranes), survival in the cell, binding to the DNA, and eventually, excretion from the body with minimum side effects. In this process, both metal coordination and hydrogen bonding will be key factors at the molecular level. Recent advances in medicinal inorganic chemistry demonstrate significant prospects for the utilization of metal complexes as drugs, presenting a flourishing arena for inorganic chemistry. Significant progress in platinum based anticancer agents has been achieved, based in part on a mechanistic understanding of the DNA-binding and pharmacological effects of cisplatin. DNAs are the target molecules for most of the metal anticancer agents in human body. The anticancer nature is the coordination of metal ions with DNA molecules. i.e. the direct chelation of the metal ions with certain nucleophilic groups in DNA (such as oxygen sites from phosphates and nitrogen as well as oxygen sites from bases), causing the DNAs’ damage in cancer cells, the DNAs were hindered during the processes of replication or transcription, the growing and division of the cancer cells were stopped, and resulted in their death.
When drug molecules (pre-anticancer molecules) enter into an organism, they will first undergo a series of processes including hydrolysis, transport and membrane-crossing, and then reach the nearby of the target DNA molecule and form active intermediates which interact with DNA molecules directly and exert the anticancer activity. These active intermediates have a general cis-form of two-water-binding transitional state:
[cis-AnM(H2O)2]m+,
where A is stably binding hydrophobic group, n=1, 2 or more and M is metal ion.
The number of water molecules binding to M must not be fewer than two and they must be lie in the ortho-position of the structure.
The functions of the hydrophobic ligand A are:
(1) caring the whole molecule to cross membranes (including cell membranes and nucleus membranes), go through the bilipid bilayers.
(2) making the metal ion to move to the nearby of the base’s cyclic-nitrogen sites and form covalent bonding.
Metal anticancer complexes are often electrophilic and may react with many cellular components, such as simple ions and molecules like Cl-,(HPO4)2-,OH- and H2O; amino acids , peptides and polyphosphates like histidine(His), methionine(Met), cysteine( Cys), glutathione, metallothionein and ATP. From the view of the coordination chemistry, metal complexes (including those of Pt) can bind to several types of possible biomolecules in the cell. But only the binding on DNA which lead to cell death is considered the most important. In the case of platinum complexes, it is quite clear that in the cells, after the relatively slow hydrolysis, cis- Pt have a preference for DNA over proteins and other molecules. L-methionine increases the rate of reaction of 5’-GMP with cisplatin and that S-bound L-HMet in the adduct [Pt(dien)(L-HMet-S)]2+ (dien= 1,5-diamino-3-azapentane ) can be replaced by N7 of 5’GMP [177,178]. A methionine-contaning protein or peptide could transport and transfer some platinum to DNA [178].
Thus, in a very simple model, the action process of a metal anticancer agent in an organism may be briefly summarized into following equations:

DNA-D is DNA-drug complex.
DNA molecule is a two-pole molecule, its surface is a negatively- charged backbone of phosphatepentose chains. In the inside of double helix there exist hydrophobic bases stacking layer by layer. For exerting its potency, the drug molecules must build with the phosphate groups of DNA at first, and then, with the help of DNA’ conformational dynamic changes(partial unwinding of the double helix), the lipophilic groups of the drug molecule may be drawn by DNA’s hydrophobic sections, the nitrogen sites on DNA molecules may be exposed, the metal atom could invade into the internal part of DNA and coordinates with the bases. Oxygen site on the phosphate group has a higher negative charge relatively, it is a good donor with high electronegativity; so its action with the metal atom was caused mainly by static electricity, forming electrovalency, belongs to charge-controlling reaction.
Through the study on hydrolysis mechanism and relationship between structure and activity of metal anticancer agents, the following three points are key if the metal anticancer agents have activities:
(1) Appropriate hydrolysis rates of a complex.

(3) Forming of the coordination both with the oxygen of phosphate groups and with nitrogen of bases.
The molecules with high anticancer activities should not only produce active intermediates by the proper hydrolysis rates, but also bind to both oxygen of phosphate groups and nitrogen of base in DNA, thus showing anticancer activity
The two pole complementary principle(TPCP) has generalized the molecular structure, action modes and steric selectivity for metal anticancer agents. TPCP includes three aspects:
1) Two-pole complement in molecular structures
The drug molecules with anticancer activity always have two poles of hydrophilicity and hydrophobicity, positive and negative charges in their structures. Correspondingly, they will present easily- leaving groups and stable keeping groups in a solution. Such twopole structures can lead the drug molecules not only to be dissolved in water and transported to the surface of the cell membranes, but also to cross the membranes by going through the lipid bilayers and arrive at the nearby of the target molecules.
2) Two-pole complement in the receptor-substrate action mode
The interaction between the drug molecule and its target molecule is always executed by forming an active intermediate which binds with oxygen sites (electrovalently) on phosphate groups and nitrogen sites (covalently) on purines, pyrimidines of DNA backbone through charge-controlling and orbital-controlling. That is a two-pole complement of electrovalent and covalent action modes.
3) Two-pole complement in the symmetry of the receptor- substrate system
The interaction characteristics of a chiral drug molecule with DNA behave as using the left hand enantiomer of the drug molecule binding with the right hand DNA, forming a two-pole complementary complex chirally.
Site-Specific Binding of Cations to Nucleic Acids
Interaction of metal cations with NAs is a very complex network of numerous contributing effects. The recently published approach by Petrov et al. represents an interesting step towards understanding the binding of metal cations to NAs in its complexity [179]. They divide the total binding free energy into four contributions on the basis of a thermodynamic cycle approach. At first, interaction energy of the gas-phase system, describing the binding site is determined. In the second step the gas-phase system is immersed into water using e.g. a continuum solvation models (COSMO)-polarized continuum model (CPCM). Interaction between the RNA and the metal cation is considered in the next step. The free energy change upon addition of diffuse electrolyte ions is calculated. The summation of these four contributions gives the total binding free energy in solution. The uniqueness of quantum chemical methods to simultaneously capture thestructure, energy and electronic properties can be fruitfully exploited atstudying the interaction of metal cations with NAs.When applying quantum chemistry to metal ions, it is critically importantto distinguish between ionic electrostatic effects and (molecular orbital,polarization) non-electrostatic effects. While the ionic effects are usually notvisible in polar environment due to solvent screening the non-electrostaticeffects are typically well expressed. Consideration of gas phase experimentalor QM data without at least a qualitative inclusion of the solvent effectsleads to conclusions that are not relevant outside the gas phase.
In NAs, a great variety of binding sites are available for the cations,represented by the heteroatoms of nucleobases and the charged oxygens ofthe phosphate groups. Electronic effects control the intrinsic propensity of acation towards a given type of binding, which cannot be accounted for bystandard force field calculations. Site-specific coordination of metal cationscauses sensible changes in the protonation and tautomericequilibria ofnucleobases and base pair strengths. QM calculations represent a dedicatedtool to study such physical properties. Gas-phase properties can be projectedinto solution either by using continuum solvent techniques or by following the charge dependence of the gas-phase results. To understand mechanism of action of DNA-binder metallodrugs, beyond the NAs, one has to consider interaction with other components ofthe biological matrix.Assessing the strength of metal-NA interactions calls for combininginformation from gas-phase quantum chemical calculations with othertheoretical approaches used to describe solvation effects as well as polymer-cationinteractions. The state-of-the-art approach [179] elaborated by Petrov et al. illustrates the way of computational quantum chemistry towards moreextended systems, such as RNA and DNA.
Metallated Nucleic Acids for Nanotechnology
DNA, as a self-assembling molecule, has long been of interest for nanotechnological applications. The addition of metal ions can switch nucleic acids between two conformations(e.g., from singleto double-strand, or from hairpin to duplex), which has been taken advantage of designing biosensors for metal ions [180-183] and recently even logical AND and OR gates [184]. Furthermore, metal ions can modify the physico-chemical properties of nucleic acids and thereby extend their natural functional repertoire. While DNA alone possesses only marginal conductivity, one idea is that the incorporation of metal ions to natural or artificial nucleotides in double- stranded nucleic acid structures might yield useful molecular wires or magnets [185].
Incorporation of metal ions such as Zn2+, Co2+, or Ni2+ into DNA yielding so-called M-DNA has been proposed to make DNA more conductive [186-188]. There is still some controversy about this [189] as well as about the definite structure of M-DNA and the situation of metals therein [190-192] (190–192). In contrast, the structure of Hg2+- and Ag+-mediated base pairs, where the metal replaces the imino protons at the N3 position in U-U/T-T base pairs or stabilizes C-C mismatches, respectively, has been well established by NMR measurements and UV melting studies [180,193-195]. The binding of an Au3+ ion in the center of a G-C base pair has been observed in the crystal structure of an RNA [196].
The incorporation of artificial nucleotides that can serve as metal ion binding sites can helpto fine-tailor the conformation and stabilities of nucleic acid structures. The incorporation of Ag+ ions into imidazole andtriazole base pairs aredemonstrated [194,197]. Clearly, with artificial nucleotides one can also further extend the range of possible metal binding sites. Examples are Cu2+-hydroxypyridone base pairs [198] and salen metal base pairs (Cu2+, Mn2+/3+, Fe3+, Ni2+, and VO2+) [199,200]. In a few cases artificial metal ion binding nucleotides have also been incorporated in polynucleotides with modified backbones like PNA (peptide nucleic acid) and GNA (glycol nucleic acid).
Structural Characterization of Metal Ion Binding Sites
The range of metal ions that have been observed to interact with nucleic acids in one or another specific context is very wide. The structural characterization of their binding sites, their precise localization, and the number and type of associated ligands in solution have been studied by a combination of chemical, biochemical and spectroscopic methods.
Chemical and biochemical methods
Well-established chemicalandbiochemical methods are used to map metal ion binding sites through backbone cleavage. However, information on structural features of the interaction sites can be inferred only indirectly, by studying the effect on ribozyme kinetics or conformation of mutated functional groups or the substitution of hard ligands or metals by soft ones. Nonetheless, many metal ion binding sites could be predicted by these methods, which only later on were confirmed in X-ray crystal structures. The strength of chemical and biochemical methods lies in the identification and characterization of catalytically important metal ion binding sites in ribozymes and their applicability also to very large molecules.
a. Metal ion-induced hydrolytic cleavage
Many metal ions catalyze the cleavage of phosphodiester bonds. The cleavage pattern in a large RNA thus holds information about the location of metal ion binding sites. The cleavage depends on local geometry and is mainly thought to take place through an “inline” nucleophilic attack of the metal at a 2’-hydroxyl group that is followed by transesterification (Figure 16) [201,202]. A2’,3’-cyclic phosphate and a 5’ hydroxyl group result of this cleavage, analogous to the reactioncatalyzed by the small phosphodiester-cleaving ribozymes. The cleavage rates have been shownto be pH dependent, thus indicating the involvement of the metal hydroxides [203]. Mg2+ has avery low cleavage capacity at neutral pH (the pKa of Mg(H2O)6 2+ is 11.4andconsequently, transition metal and lanthanide ions are normally used as probes. Pb2+ is muchmore efficient than Zn2+ and Mn2+ [204], but all three have been used to localize metal-RNAinteraction sites [205-208]. Lanthanide(III) ions are especially suitable and employed frequentlyas probes because their charge makes them bind rather tightly and pKa values close to neutralfacilitate efficient cleavage [209-211] (Figure 16).
b. Metal ion-induced radical cleavage
Alternatively, metal ion binding sites can be probed with Fe2+ [212], taking advantage of thefenton reaction. In the presence of H2O2, Fe2+ is oxidized to Fe3+ generating short- lived hydroxyl radicals that will lead to backbone cleavage in the near proximity of the Fe2+/3+ binding sites. Fe3+ is then reduced again to Fe2+ by sodium ascorbate.
c. Mutational approaches to determine metal ligands
Once putative binding sites have been determined, various nucleotide analogs [213] can help to identify more details about a coordination site. Functional groups of a specific nucleobase,Figure( 8), sugar or phosphate moiety are modified or removed and the effect can be monitored, e.g., by looking at the metal ion cleavage pattern. Alternatively, the metal ion binding sites that are important for catalysis or essential for structural integrity can be inferred from the catalytic competence or overall conformation of the mutated nucleic acid as determined in enzymatic or electrophoretic mobility shift assays, respectively.Nucleotide analogue interference mapping (NAIM) [214] is an efficient way to systematically probe the importance of RNA functional groups [215]. In combination with metal ion switch experiments this method is also useful in establishing putative metal ion binding sites [216-219].
d. Metal ion switch experiments
The divergent ligand preferences of different kinds of metal ions are the basis of metal ionswitch experiments. Mg2+ and Mn2+ prefer the harder oxygen ligands, while Cd2+, Zn2+ and Pb2+ display a pronounced preference for the softer aromatic-nitrogen and especially sulfur sites [220,221]. Mutations that exchange oxygen for sulfur or nitrogen groups can significantly suppress Mg2+ binding, but the adverse effects can often be rescued by Cd2+ or Mn2+. The method is especially useful in identifying catalytically involved metal ions [222] and is usually thought to be limited to the analysis of directly coordinated metal ions.
Spectroscopic methods
I. Electron paramagnetic resonance
EPR methods require the sample to be frozen. In solution at room temperature, EPR can provide information on the populations of free and bound metal ions and thereby binding affinities as well as cooperativities but structural details are not accessible [223-225]. At low temperature in a frozen sample, hyperfine interactions between the unpaired electron spin of a paramagnetic metal ion and the spin of the nuclei are able to hint at the coordination environment of the metal ion. Two more advanced applications, electron-nuclear double resonance spectroscopy(ENDOR) and electron spin echo envelope modulation(ESEEM) can even reveal details about the number and type (14N/15N, 1H/2H, 31P) of the immediate coordinating partners within a radius of 6–7 Å [226,227]. Since EPR is limited to paramagnetic metal ions, Mn2+ is a popular object of study due to its similarities with Mg2+. Both its electron and nuclear spin are 5/2, but degeneration leads to a spectrum with only 6 characteristic main lines. The exchange of metal ion-coordinated water molecules for RNA ligands is accompanied by subtle perturbations of the spectrum. As signals from differently bound metal ions will always overlay, an unambiguous analysis usually requires sample conditions with only one prominent metal ion binding site. A single tightly bound Mn2+ ion can, for example, be observed in a background of monovalent ions in the Hammerhead ribozyme: ESEEM spectroscopy allowed the precise localization of the metal ion at a site with specifically 15N-labeled guanine, the determination of ligand sites and also of the hydration level [228]. In a different approach, multiple Mn2+ binding sites in the Diels-Alder ribozyme were gradually silenced by Cd2+, allowing thus their individual characterization [225].
II. Lanthanide (III) luminescence
The luminescence of lanthanide (III) metal ions is sensitive to the direct coordinationenvironment and thus can yield information on the metal ion binding pocket.
III. X-ray absorption spectroscopy
The coordination environment of tightly bound transition metal ions can be characterized indetail in solution by X-ray absorption spectroscopy (XAS) methods like X-ray absorption near edge structure (XANES)and extended X-ray absorption fine structure(EXAFS). XANES is sensitive to the average oxidation state of the metal in the sample, while EXAFS allows thededuction of the number and type of ligands as well as coordination geometry and metal-ligand atom distances. Detection is possible in dilute solution and metal ions that are classified as spectroscopically silent (like Na+, K+, Mg2+, or Ca2+, Cu+, and Zn2+) are accessible [229]. Nucleic acids are not optimally suited for XAS investigations, considering thepredominantly weak interactions and the coexistence of many metal ion binding sites in mostconstructs. Nonetheless, higher-affinity binding sites, like the ones in the G-quadruplexchannel [230] or in a short RNase P helix P4 model [231] can be characterized in remarkable detail, notto forget complexes of kinetically more inert metal ions [232,233].
IV. Vibrational spectroscopies
Metal ion binding is reflected in changes of the vibrational bands of nucleobase, sugar, andphosphate constituents, which can be measured by infrared (IR) and Raman spectroscopy. WhileIR depends upon oscillating dipole moments and is influenced by all types of non-symmetricallybonded atoms, Raman signals derive from the inelastic scattering of photons and occur whenthere are changes in polarizability. The latter are therefore especially sensitive to electron-rich ormultiply bonded groups. In addition, water absorbs in the IR range but is not observed in Ramanspectra. Thus, the two methods can yield useful complementary information on a fast timescale and in all physical states [234,235]. Discrete vibrational bands for base, sugar and phosphategroups can be observed, albeit only as an average signal of all conformational states in thesample. Isotopic labeling is an approach to partly alleviate this problem [236,237]. FT-IR (Fourier-transform infrared) spectroscopy and Raman spectroscopy have beenemployed to semiquantitatively follow metal ion-induced conformational changes [238,239] and identify primary binding sites for a variety of metal ions in RNA and DNA [240- 245]. ByRaman spectroscopy the largest metal ion-dependent changes are observed in the phosphodiestersignals. The symmetric stretching of non-bridging phosphate oxygens has recently beenproposed to contain quantitative information about the degree of innersphere coordinated metalions in RNA [246]. Raman spectroscopy in solution requires ratherconcentrated and highly pure samples, restricting this method to the analysis of smallerconstructs. Raman crystallography or microscopy is an approach that can significantly increasesignal intensity and reduce background signals. It allowed the identification of inner- andoutersphere coordinated metal ions in the hepatitis delta virus (HDV) ribozyme [247-249].
V. Nuclear magnetic resonance methods
Nuclear magnetic resonance (NMR) spectroscopy has provided the three-dimensional structuresof hundreds of RNA and DNA molecules. Its restriction in terms of molecular size comparedto X-ray crystallography, is outweighed by the singular capacity to reveal not only structure andconformation, but also local and global dynamics of macromolecules in solution in a quantitativemanner. The big advantage compared to other spectroscopic methods in solution is the resolution of the individual nuclei at almost every single position in a polynucleotide chain, allowing thesimultaneous site-specific characterization of multiple metal ion binding sites. Most approaches are based on indirect observations of metal ion induced changes in thenucleic acid binding sites. The most abundant isotopes of hydrogen and phosphorus (1H and 31P)both have nuclear spins of 1/2 as it is the case with 15N and 13C. The latter are widely used inisotopically enriched nucleotides instead of the natural isotopes 14N and 12C. But also the directobservation of NMR-active metal nuclei can help in the structural and thermodynamiccharacterization of metal binding sites [250,251]. Metal ion-nucleic acid interactions can haverepercussions on chemical shifts, relaxation properties and scalar couplings of involved NMRactive nuclei [252]. In addition, nuclear overhauser effects(NOEs) to the protons of popularmetal ion mimics, [Co(NH3)6]3+ and NH4 + , can denote coordination sites [253].
• Chemical shift perturbations
Chemical shifts are the most straightforwardlymeasurable factor in a NMR experiment. Binding of metal ions can influence thechemical shift in two ways: Either through direct deshielding of a nucleus or throughshielding/deshielding effects upon conformational changes of the neighborhood that result froma binding event. The latter effect is especially prevalent for 1H shifts and reduces the accuracywith which a binding site can be localized, but on the other hand it is also a highly sensitive flagto define the binding pocket. In the case of smaller structures that do not undergo significantconformational changes upon metal ion interaction, 1H shift perturbations can be used as goodindicators of metal ion binding sites [254- 256]. Imino proton shifts are mostly well resolvedand can often be monitored satisfactorily in 1D spectra. However, the observable ones areusually part of a Watson-Crick hydrogen bonding pattern and then not in close proximity to themetal ion binding atom. In addition, the chemical shift of imino protons is very sensitive totemperature and accessibility of bulk water, which may falsify results. Chemical shifts induced by [Co(NH3)6]3+ are slightly more pronounced due to the higher chargeand associated 10 times stronger binding affinity [252]. Unfortunately, exchange rates ofcommonly employed metal ions are often in the intermediate regime on the NMR time scaleleading to a general broadening of the lines already at lower concentrations that can impede theanalysis of chemical shift changes.
With 31P, nucleic acids contain a second highly sensitive and abundant spin 1/2 nucleus inaddition to protons, whose only disadvantage is the low signal dispersion. Most 31P resonancescluster in the small region between –3 to –1 ppm and only a few with non-standard backbonetorsion angles can be resolved from the rest [257]. 31P resonances can be unambiguouslyassigned by the site-specific incorporation of a non-bridging 17O that efficiently broadens thesignal of the adjacent 31P [258]. Coordination of Cd2+ to a phosphorothioate, for example, will lead to an upfield shift of the 31P resonance by a few ppm in the hammerhead ribozyme [259,260]. However, such experiments have to be interpreted with care because theintroduction of a sulfur atom might also create a new or specially shifted binding site. Considering the unfavorable NMR characteristics of oxygen isotopes, 15N is the onlynucleus in nucleic acids that can serve as a probe for the detection of an innersphere coordinationsite. In addition, 15N is straightforwardly observable by NMR spectroscopy. This explains whywith 15N, compared to 1H, 13C and 31P, much higher chemical shift perturbations can beobserved. Direct coordination to nucleobasenitrogens is confirmed in Hg2+-mediated thyminebase pairs in DNA by 30 ppm 15N downfield shifts [193] as well as by Ag+ coordination toimidazole nucleotides [194]. Also the more labile coordination of Cd2+ and Zn2+ ions to N7induces 20 ppm shifts in 1D 15N experiments [261-263]. An initial broadening of the 15Nresonance is attributed to the exchange between free and bound states. At higher, saturating Cd2+concentrations the peak grows sharper again.
• Paramagnetic effects
The large magnetic moment of an unpaired electron of aparamagnetic metal ion species efficiently relaxes nuclei in their immediate environment [231,264,265]. The effect is strictly distance-dependent (relative to r–6). Paramagnetic line broadening information caneven be included as weak distance restraints in molecular dynamics calculations [256]. Mn2+exchanges very fast between the free and bound form and can therefore effectively relax a wholemolecule at substoichiometric concentrations. μM amounts are usually enough to detect site-specificlinebroadening effects. Mn2+ is most widely used because of its likeness to Mg2+ [266-268], but also Co2+ and Ni2+ are suitable paramagnetic probes [118,269].
• Nuclear overhauser effect cross-peaks to [Co(NH3)6]3+ and NH4+
[Co(NH3)6]3+ and NH3+ is a mimic for[Mg(H2O)6]2+ [270] and has been used in a wide range of studies[271-274]. Similarly, NH4 + can be used as a substitute for monovalent metal ions [253]. Both compoundspossess protons that are amenable for direct observation of inter-molecular NOE crosspeaksto nucleic acid protons in a radius smaller than 6 Å. In the most common casetumbling of the cation in the binding site and exchange with the unbound state are fast on thechemical shift time scale and all [Co(NH3)6]3+ + protons resonate at a single frequency. G-quadruplexstructures are an exception, displaying a binding site with on-off rates slow enoughto allow observation of separate resonances for free and bound ammonium [275,276]. The tightbinding even significantly slows down the usually fast proton exchange of ammonium [253]. It isadvantageous to employ 15N-labeled NH4 + because it not only avoids the quadrupolar14Nnucleus, but also allows for heteronuclearsingle quantum coherence (HSQC) experiments that detect the exchange of ionsbetween binding sites and the solvent [277]. Structural information, however, can be inferred inboth fast and slow exchanging cases. From the NOE cross-peaks of [Co(NH3)6]3+ or NH4 + weakdistance restraints can be extracted and integrated in molecular dynamics simulations to inferbinding sites [252].
• Direct detection of NMR-active metal isotopes
A good number of the metal ionsthat have been studied in association with nucleic acids have at least one NMR-active isotope. Unfortunately many of them, including the biologically most relevant 23Na+, 39K+, and 25Mg2+,have half-integer spins > 1/2. Fast quadrupolar relaxation usually restricts their use in solutionNMR experiments to the study of kinetic and thermodynamic features by line shape analysis [278-282] and pushes them more into the field of solid state NMR, whichis better suited to handle the large quadrupolar effects.Spin 1/2 isotopes are available in 52Fe, 107Ag, 109 Ag, 111 Cd, 113 Cd, 19 5Pt, 199 Hg, 203 Tl, 205 Tl, and 207 Pb. Many have wide chemical shift ranges and can give useful information about coordination geometry and ligand atom identity, as has been shown in proteins [283,284]. Innucleic acids, however, their use is most often restricted by the problem of weak binding and fastexchange. An exception is again the high-affinity binding of monovalent metal ions to G-quadruplexstructures that even permitted the direct observation of separate peaks for the boundand unbound species of 23Na+, 39K+, and 85Rb+, which was not thought possible so far [250,285]. 205Tl+ is a very useful NMR substitute for K+ and Na+ due to its high natural abundance (70%) and high sensitivity [286]. Such direct couplings could not be observed between 113Cd2+ and 15N upon coordination ofthe metal ion to an N7 in the tandem G·A base-pair motif of the hammerhead ribozyme [261,287], which is attributed to the fast exchange of Cd2+. The coordination of Ag+ ionsbetween imidazole nucleotide analogs in a DNA duplex on the other hand is
Determination of Binding Kinetics and Affinities
Metal ions interact with nucleic acids on a very wide range of time scales. Fast exchange (ms toμs range) dominates the dynamic interactions of the majority of alkali and alkaline earth metalions. Metal complexes effective in cancer treatment are characterized by particularly slow kinetics that can be in the order of hours or days [288]. Mg2+ is special with regard to its water-ligand exchange rate of ~2×105 s–1 being four orders of magnitude slower than that of most of the main group metal ions, but also significantly slower than many transition metal ions [289]. Two challenges are faced in the thermodynamic analysis of metal ion binding to nucleic acids. Firstly, metal ion binding is inextricably interwoven with the folding and structural stability of nucleic acids and the one cannot be studied without affecting the other [290]. Secondly, usually there are only few specific binding sites with higher affinity and they coexist with a large background of weak, transient electrostatic interactions that are not easily accounted for in simple binding polynomials [291]. Nucleic acid conformation will not significantly change any more upon addition of the metal ion to be analyzed and that all unspecific interaction sites will be saturated by the monovalent ion. Metal ion affinities have been inferred from a range of different observables, like thermodynamic stabilities of the nucleic acid fold measured by UV melting curves [292-295] or the thermodynamics of folding of the hammerhead ribozyme by isothermal titration calorimetry (ITC) [296]. Apparent metal affinity constants have been determined from observed rate constants of ribozyme catalysis in many cases [297-301]. An approach based on the gas-phase fragmentation of metal-nucleic acid complexes in electrospray ionization mass spectrometry (ESI-MS) was recently applied todetermine binding affinities to the thrombin binding aptamer [302]. In addition, the ratio of bound and unbound metal ions, as determined by fluorescent indicators, atomic emissionspectroscopy (AES), NMR, EPR or lanthanide luminescence, in dependence of metal ionconcentration, yields information on binding affinities.
Analysis of the dependence of the bound and free metal ionconcentrations on the total metal ion concentration can help todisentangle them to some extent to determine classes of binding sites with similar affinities andtheir occupancies and give information about binding cooperativity. There is usually also a restriction to simpler modelmolecules with one or only few strong binding sites. While the size restriction is also true for NMR, this method has one advantage over all the others: It allows to separately monitor bindingevents at several individual binding sites in a molecule and to determine intrinsic binding affinities. So-called “ion counting” methods detect the free metal ion concentration in a sample, from which the number of ions strongly bound to the nucleic acid can be deduced. This can be useful to establish the number of strong divalent metal ion binding sites in a high-salt background [213], but also to evaluate the affinities of different metal ions with respect to each other. Fluorescent indicators have been widely used to determine free metal ion concentrations [303-305]. Recent methodological progress has made AES a very valuable tool in this regard, able to detecta far wider range of mono- and divalent cations and also anions, thereby more completelyaccounting for the ion cloud around polynucleotides [306,307].
Biological Importance of Pyrimidine
Antimicrobial activity
Microbes cause various types of disease like pneumonia, amoebiasis, typhoid, malaria, cough and cold infections and some severe diseases like tuberculosis, influenza, syphilis, and AIDS as well. Flucytosine is a fluorinated pyrimidine used as nucleosidalanti- fungal agent for the treatmentof serious systemic infections caused by susceptible strains of candida and Cryptococcus (Diagram 3) [308].





Anti-inflammatory activity
Anti-inflammatory refers to the property of a substance or treatment that reduces inflammation. Due to remarkable pharmacological efficiency of pyrimidine derivatives, anextensive research has been focused on anti-inflammatory activity of pyrimidine nucleus.
Anticancer activity
Cancer is an idiopathic disease and doctors and scientists are constantly trying to evolve new effective drugs for its treatment. There is no other disease which parallels cancer indiversity of its origin, nature and treatments. One of the early metabolites prepared for cancertreatment was 5-fluorouracil(5-FU) [309], a pyrimidine derivative. 5-Thiouracil also exhibits some useful antineoplastic activities (Diagram 4) [310].
Antineoplastic compounds possessing guanine nucleus likes azathioprine, mercaptopurine, thioguanine, and tegafur [311-314] have been discovered. These drugs stop the use of regular cellular metabolites. Several anti-metabolites like mopidamol, nimustine, raltitrexed, uramustine, and trimetrexate have been studied . A pyrimidine antimetabolitegemcitabinehas antitumor activity against murine solid tumor [315-320].
Antitubercular activity
Tri-substituted pyrimidines have their in vitro anti-malarial activity against Plasmodium falciparum in the range of 0.25- 2μg/ml and anti-tubercular activity against Mycobacterium tuberculosis at a concentration of 12.5 μg/ml Diagram 5 [321].
Central Nervous System (CNS) Activity (Sedative/hypnotic)
A wide variety of barbiturates are used as sedative, hypnotics and classified as drugs having short, intermediate and long duration of action [322]. Allobarbital, aprobarbital, phenobarbital, secobarbital, and pentobarbital are frequently used hypnotic barbiturates [323]. Hexobarbital, cyclobarbital and propallylonal are used sedatives hypnotics [324].
Antihyperlipidemic activity
2-substituted-6-phenyl and 7-phenyl thieno[3,2-d] pyrimidin- 4-ones are synthesized [325] through cyclocondensation of the corresponding thiophenoaminoesters with a variety of nitriles in the presence of dry hydrogenchloride gas. Antihyperlipidemic activity has been reported in a few thienopyrimidines Diagram 6.
However extensive studies on the structural chemistry for some of the nucleic acid complexes for the solid and solution state complexes. The studies were mainly focused to the dielectric, electrical conductivity, potentiometric and thermal measurements [326-330].
Acknowledgement
None.
Conflict of Interest
No conflict of interest.
References
- M Schena, D Shalon, RW Davis, PO Brown (1995) Sceince 270: 467
- S Yamakawa, K,o, A Chisaka, K Yashida, A Shinmyo, T Kohchi (2004) J Biosci Bioeng 98: 140.
- N Mera, H Aoyagi, S Nakasona, K Iwasaki, H Saiki (2004) J Biosci Bioeng 97: 169.
- B Goswami, B L Gaffney, R A Jones (1993) J Am Chem Soc 115: 3832.
- C Wang, H Gao, B.L Gaffney, R A Jones (1991) J Am Chem Soc 113: 5486.
- B L Gaffney, C Wang, R A Jones (1992) J Am Chem Soc 114: 4047.
- Y S Rhee, C Wang, BL Gaffney, R A Jones (1993) J Am Chem Soc 115: 12607.
- X Zhang, B L Gaffney, RA Jones (1998) J Am Chem Soc 120: 615.
- WLF Armarego (1967) In “The Chemistry of Heterocyclic Compounds, Fused Pyrimidines”, Part I: “Quinazolines”, D.J Brown, Ed.; Vol 24/1, Interscience Publishers: New York– London – Sydney.
- M Legraverend (2008) Tetrahedron 64: 8585.
- W B Parker (2009) Chem Rev 109(7): 2880.
- S Ostrowski (2001) Polish J Chem 75, 1661, Jordan J Chem 4(1): 1(2009).
- A C Tella, J A Obaleye (2010) Int J Chem Sci 8(3): 1675.
- J Cieplik, M Stolarczyk, J Pluta, O Gubrynowicz, I Bryndal, et al. (2011) Acta Poloniae Pharmaceutica, Drug Research 68(1): 57.
- L Zhuo, K Kou, Y Wang, H Chen (2015) Designed monomers, polymers 18(1): 42.
- Y Liang, S F Wnuk (2015) Molecules 20: 4874.
- M S Masoud, A M Heiba, F M Ashmawy (1983) Trans Met Chem 8: 124.
- M S Masoud, T.M Salem, Z Zaki (1984) XXIII International Conference on Coordiantion Chemistry, Colorado, USA, p. 372.
- A A Hasanein, M S Masoud, A M Heiba (1985); (1987) Current Science 54(2): 1165; J Chem Soc Pak 9(2): 199.
- M S Masoud, S S Haggag (1985) 30th IUPAC Congress, Manchester 9-13 September (1985); 8th International symposium on solute-solute-solvent interaction, p 210, p 303, University of Regensburg, 9-10 August (1987); 3rd National Symposium on Grystallography, Cairo, Academy of Scientific Research, Technology, National Committe of Technology, 26-27 January (1988); 5th World Conference on Thermal Analysis, Corfu, Greece, May 16-17 (1988); 9th Arabian Chemistry Conference, Kwuit, 15-19 December (1990); Thermochimica Acta, 196,221 (1992).
- M S Masoud, N A Ibrahim, S A AbouAli, G Y Ali, I M Abed, Ind J Chem 25A, 389.
- M S Masoud, S A Abou Ali, G Y Ali, IM Abed (1987) Thermochim Acta 122, 209.
- M S Masoud, M E Kassem, Y Abd El-Aziz, S Massoud (1987) Xth Conference on Solid State Science, Applications, p 46.
- M A El-Dessouky, M S Masoud, F A Ali, S A Abou El-Enein, Affinidad (1988) 416, August 321; 2nd Chemistry Conference, Fac of Sci Alex Univ 28-30 June, 178,196 (1988); Trans Met Chem 15, 443 (1990); XXVII Colloqium spectroscopicum international, Bergen, Norway, 9-14 June (1991).
- M S Masoud, M Khater, N El-Guindi (1988) J Iraqi Chem Soc 13, 177.
- M S Masoud, E.M.Soliman, A E El-Kholy, E A Khalil (1988) Thermochem Acta 136: 1.
- M S Masoud, Z M Zaki (1988) Trans Met Chem 13: 321; Bull Fac Sci Mansoura Univ 17(1): 71 (1990).
- M S Masoud, E M Soliman, A M Heiba (1989) Trans Met Chem 14: 175.
- M S Masoud, S Abou El-Enein (1989) Thermochim Acta 140, 365.
- M S Masoud, E A Khalil, A R Youssef (1990) Synth React Inorg Met.-Org Chem 20(6): 793.
- M S Masoud, S S Haggag, E M Soliman, M El-Shabasy (1991) J Mater Sci 26: 1109.
- M S Masoud, S A Abou El-Enein, E El-Shereafy, J Therm Anal 37: 365 (1991).
- M S Masoud, E A Khalil, Polish J Chem 65: 933 (1991).
- M S Masoud, M E Kassem, Y Abd El-Aziz, S M Khalil (1991) Bull Fac Sci Mansoura University 18(1), 105.
- M S Masoud, E A Khalil, S S Haggag, Pak J Sci Ind Res 35,480 (1992); Nucleosides, Nucleotides, Nucleic Acids 25(1), 73 (2006).
- M S Masoud, S A Abou El-Enein, O F Hafez (1992) J Therm Anal 38: 1365.
- M S Masoud, M M El-Essawi, A M Amr, XXI Congress on Molecular Spectroscopy, EUCMOS XXI, Technical University of Vienna, Austria, 23-28 August (1992); Anal Proc 29: 370.
- E A Khalil, M S Masoud, A El-Marghany (1993) Pak J Sci Ind Res 36: 68.
- M S Masoud, E El-Shereafy, E A Khalil, O H Abd El-Hamid (1993) Egypt J Anal Chem 2(1): 95.
- M S Masoud, Z M Zaki, F M Ismail, A K Mohamed (1994) Z für Phys Chem Bd 185, S 223.
- M S Masoud, O H El-Hamid, Z M Zaki (1994) Trans Met Chem 19: 21.
- M S Masoud,S S Haggag, Z M Zaki, M EI-Shabasy (1994) Spectrosc Lett 27(6): 775.
- S A Abou EI-Enein, M S Masoud, A El-Khatib, S Abd El-Aziz (1994) Alexandria Engineering J 33(3): D103.
- M S Masoud, O.F Hafez, N.A Obeid (1994) Pak J Sci Ind Res 37(10): 421.
- M S Masoud, H.M El-Nahas, S S Haggag Pak (1995) J Sci Ind Res 38(3-4): 108.
- M S Masoud, S.S Haggag, O H Abd El-Hamid, Revue Roumaine De Chimie(1996) 41(1-2), June 21-27.
- M S Masoud, A K Ghonaim, A A Mahmoud, 1st International Conference on Basic Sciences, Advanced Technology, Faculty of Science, Assiut University, "A 5-1,AP8", "P 24, 46",10-11 Nov.(1996); 2nd Mediterranean Basic Conference on Analytical Chemistry 23-28 Nov Robat (Morocco) (1997); 36th Annual Eastern Analytical Symposium & Exposition, Somerset, New Jersey, USA; No 525, p.27,16- 21 Nov (1997)
- M S Masoud, M M Ghonaim, O A El-Wahab (1996) Egypt J Appl Sci 11(8): 302.
- M S Masoud, R H A El-Sayed, A H Mostafa, N H Abd El Moneium (1999) The 5th International Conference of Chemistry, its Role in Development, Department of Chemistry, Faculty of Science, El Mansura University.
- M S Masoud, S A Abou El- Enein, H M Kamel (1999) 3rd International Scientific Conference (Science, Development, Environment), Faculty of Science, Al-Azhar University, p.72, 22- 25 March; Ind J Chem 41A: 297.
- M S Masoud, A El Marghany, S K El Sadany, N G Ali (1999) The 5th International Conference of Chemistry, its Role in Development, Department of Chemistry, Faculty of Science, El Mansura University, 19-22 April.
- M S Masoud, A A Hasanein, A K Ghonaim, E A Khalil, A A Mahmoud (1999) Z für Phy Chem Bd 209 S, 223.
- M S Masoud, A M Hindawy, R H Ahmed (1999) Pak J Sci Ind Res 42(1): 11.
- M S Masoud, E A Khalil, A.A Ibrahim, A El-Marghany (2007) Z für Phys Chem Bd 211S, 13 (1999); Spectrochim Acta 67A, 662.
- M S Masoud, S A Abou El-Enein, NA Obeid (2001) Z für Phys Chem 215: 7 867.
- M S Masoud, A K Ghonaium, R H Ahmed, A A Mahmoud, A E Ali (2001) Z für Phys Chem 215(4): 531.
- M S Masoud, A K Ghonaim, R H Ahmed S A Abou El-Enein, A A Mohmoud (2002) J Coord Chem 55(1): 79.
- M S Masoud, A A.Soayed, A E Ali, O K Sharsherah (2003) J Coord Chem 56(8): 725.
- M S Masoud, A A Soayed, A E Ali (2004) Spectrochim Acta, 60A: 1907.
- M S Masoud, S A Abou El-Enein, M Ayad, A S Goher (2004) Spectrochim Acta 60A: 77.
- M S Masoud, A M Hindawey, E A Khalil, A M Ramadan (2005) Bull Fac Sci Assuit University 33(2B), l-8 (2004); Can J Anal Sci Spectrosc 50(6): 297.
- M E Mahmoud, M S Masoud, N N Maximous (2004) Mikrochim Acta 147(1-2): 111.
- M S Masoud, A E Ali, M A Shaker, M Abdul Ghani (2004) Spectrochim Acta 60A: 3155.
- M S Masoud, E A Khalil, A M Hafez, A F El-Husseiny (2005) Spectrochim Acta 61A: 989.
- M S Masoud, E A Khalil, O H Abd El-Hamid, A A Soayed (2005) The Egyptian Science Magazine 2(2): 33.
- M S Masoud, M F Amira, S A El-Moneim, G M Moghazy, A A Abou-Hagar, et al. (2005) The Egyptian Science Magazine 2(4): 88.
- M S Masoud, T S Kasem, M A Shaker, A A Ali (2006) J Therm Anal Calorim 84(3): 549.
- M S Masoud, A A Soayed, A S El-Kholany, Proceeding of the 3rd international Chemical & Environmental Engineering Conference, 16-18 May (2006) J of Radioanalytical, Nuclear Chemistry 1-8 (2011).
- M S Masoud, E A Khalil, A M Ramadan, Y M Gohar, A Sweyllam (2007) Spectrochim Acta 67A: 669.
- M S Masoud, M Sh Ramadan, MH Al-Saify (2006) Chem-04, Faculty of Science, Cairo University, p.50: 5-8. International Conference on Science, Technology, Prauge, Czech Republic: 5-6.
- M S Masoud, M F Amira, A.M Ramadan, Gh M El-Ashry (2008) Spectrochim.Acta, 69A, 230.
- H H Hammud, K H Bouhadir, M S Masoud, A M Ghannoum, SA Assi ((2008)) Journal of Solution Chemistry 37(7): 895.
- M S Masoud, S A Abou El-Enein, AM Ramadan, A S Goher (2008) J Anal Appl pyrolysis 81(1): 45.
- M S Masoud, M Sh Ramadan, A El-Samahy, I Mahmoud, M H Al-Saify (2008) Chem-05, Faculty of Science, Cairo University, p.48, 3-5 March.
- M T Zaworotko, H H.Hammud, G Mc- Manus, A M Ghannoum, A Kabbani, et al. (2009) J Chem Crystall 39: 853.
- M S Masoud, A El-Merghany, M Y Abd El-Kaway (2009) International Conference on Chemistry, its Role in Development, Department of Chemistry, Faculty of Science, University of Mansoura, (16-19) April (2007); Synthesis, Reactivity in Inorganic, Metal-Organic, Nano-Metal Chemistry, 39 (9): 537.
- M S Masoud, A El-Merghany, A M Ramadan, M Y Abd El-Kaway (2010) J Therm Anal.Calorim 101(3): 839.
- M S Masoud, M K Awad, M A Shaker, M M T El-Tahawy (2010) Corrosion Science 52(7): 2387.
- M S Masoud, M.A Shaker, A E Ali, G S Elasala (2012) Seventh International Scientific Conference, Environment, Development, Nanotechnology, Faculty of Science, Al-Azhar University, Cairo, Egypt, 170: 22-24, Spectrochim Acta, 90A: 93.
- M S Masoud, A.M Hindawy, A A Soayed, M Y Abd El-Kaway (2012) J Fluid Phase Equilibria, 312: 37 (2011); Research Conference for Egyptian Faculties of Science, Alexandria University, Session 6, OR-11 (18-19) May (2011); Spectrochim Acta, 92A, 15: 256.
- M S Masoud, A A Soayed, A F El-Husseiny (2012) Spectrochim Acta 99A: 365.
- N Z Shaban, M S Masoud, M A Mawlawi, D Awad, O M Sadek (2012) J Physiol Biochem 68(4): 475.
- M S Masoud, M Y Abd El-Kaway (2012) Spectrochim Acta 102A: 175.
- M S Masoud, A El-Marghany, A Orabi, A E Ali, R Sayed (2013) Spectrochim Acta 107A: 179.
- M K Awad, M S Masoud, M A Shaker, A E Ali, M M T El-Tahawy (2013) Research on Chemical Intermediates 39(6): 2741.
- M S Masoud, A E Ali, M Y Abd El-Kaway (2014) J Therm Anal Calorim 116(1): 183.
- M S Masoud, M F El-Shahat, A S Elkholany (2014) Spectrochim Acta, 127A: 216.
- D J Brown (1994) In “The Chemistry of Heterocyclic Compounds, The Pyrimidines”, Taylor, E.C Ed.; Vol 23, J Wiley & Sons: New York – Chichester – Brisbane – Toronto –Singapore.
- A Albert (1985) In “Selective Toxicity: The Physico-Chemical Basis of Therapy”, 7th ed N.B Chapman; J.E Hall Eds.; London, p 347.
- J H Lister (1996) In “The Chemistry of Heterocyclic Compounds, The Purines”, Supplement I; Wiley – Interscience: New York – Chichester – Brisbane – Toronto – Singapore.
- B Bobrański, Z Synowiedzki (1948) J Am Pharm Assoc Sci Edu 37: 62.
- H Rosenmeyer (2004) Chem Biodiv 1: 361.
- D K Goette (1981) J Am Acad Dermatol 4: 633.
- J Darbyshire, M Foulkes, R Peto, W Duncan, A Babiker, et al. (2004) “The Cochrane Library”, John Wiley & Sons, Chichester, Issue 2.
- P L Dordoni, L Frassanito, M F Bruno, R Proirtti, R De Cristofaro, et al. (2004) Br J Haematol 125: 79.
- M J Eadie, W D Hooper (2002) “Other Barbiturate-Methylphenobarbital”, In “Antiepileptic Drugs”, Eds E H Levy, R Mattson, B.S Meldrum, E Perucca, Lippincott-Williams, Wilkins, Philadelphia.
- H M McIntosh (2001) from “The Cochrane Library”, Update Software Ltd Issue 4.
- A M Joffe, J D Farley, D Linden, G Goldsand (1989) Am J Med 87: 332.
- T F Cash (1999) Br J Dermatol 141: 398.
- E Petersen, D R Schmidt (2003) Expert Rev Anticancer Ther 1: 175.
- R Schaumann (1996) In “Houben-Weyl: Hatarenes IV (Six-, Higher-Membered Ring Systems with Two or More Heteroatoms)”, Thieme-Interscience.
- C Orvig, M J Abrams (1999) Chem Rev 99: 2201.
- H Sakurai, Y Kojima, Y Yoshikawa, K Kawabe, H Yasui (2002) Coord Chem Rev 226: 187.
- W Koch, M C Holthausen (2002) “A Chemist’s Guide to Density Functional Theory”, 2nd ed Wiley-VCH, Weinheim, New York, Chicester.
- J E Šponer, V Sychrovský, P Hobza, J Šponer (2004) Phys Chem Chem Phys 6: 2772.
- V Sychrovský, J Šponer, P Hobza (2004) J Am Chem Soc 126: 663.
- C W Bock, A K Katz, G D Markham, J P Glusker (1999) J Am Chem Soc 121: 7360.
- E Freisinger, R K O Sigel (2007) Coord Chem Rev 251: 1834.
- H Sigel, R Griesser (2005) Chem Soc Rev 34: 875.
- B Knobloch, B Nawrot, A Okruszek, R K O Sigel (2008) Chem Eur J 14: 3100.
- H Sigel, R B Martin (1994) Chem Soc Rev 23: 83.
- M Roychowdhury-Saha, D H Burke (2006) RNA 12: 1846.
- D N Frank, N R Pace (1997) Proc Natl Acad Sci 94: 14355.
- W Wang, L P Billen, Y Li (2002) Chem Biol 9: 507.
- P J Bruesehoff, J Li, A J Augustine, Y Lu (2002) Comb Chem High T Scr 5: 327.
- J Li, W C Zheng, A H Kwon, Y Lu (2000) Nucleic Acids Res 28: 481.
- T Pan, B Dichtl, O C Uhlenbeck (1994) Biochemistry 33: 9561.
- H P Hofmann, S Limmer, V Hornung, M Sprinzl (1997) RNA 3: 1289.
- M Zivarts, Y Liu, R R Breaker (2005) Nucleic Acids Res 33: 622.
- R S Alexander, Z F Kanyo, L E Chirlian, D W Christianson (1990) J Am Chem Soc 112: 933.
- B Schneider, M.Kabeláč, P Hobza (1996) J Am Chem Soc 118: 12207.
- V V Murashov, J Leszczynski (1999) J Phys Chem 103B: 8391.
- M J Fedor (2002) Curr Opin Struct Biol 12: 289.
- J Šponer, M Sabat, L Gorb, J Leszczynski, B Lippert, et al. (2000) J Phys Chem 104B: 7535
- N Gresh, J E Šponer, N Špačková, J Šponer (2003) J Phys Chem 107B: 8669
- L Rulíšekand, J Šponer (2003) J Phys Chem 107B: 1913.
- E H S Anwander, M M Probst, B M Rode (1990) Biopolymers 29: 757
- J Šponer, J V Burda, M Sabat, J Leszczynski, P Hobza (1998) J Phys Chem 102A: 5951
- J Munoz, J Šponer, P Hobza, M Orozco, F J Luque (2001) J Phys Chem 105B: 6051.
- B Lippert (1999) “Cisplatin: chemistry, biochemistry of a leading anticancer drug “, Wiley-VCH, Weinheim, New York, Chichester, USA.
- B Rosenberg, L Vancamp, J E Trosko, V H Mansour (1969) Nature 222: 385.
- A M Pizarro, P J Sadler (2009) “In Nucleic Acid-Metal Ion Interactions”, Ed N V Hud, Royal Society of Chemistry, Cambridge, UK.
- K Hasegawa, K Mori, N Ishii, Y Aoki (2003) Chem Abstr 2478640.
- J M Essigmann R G Croy, K J Yarema, M Morningstar (2002) US 5882941 (1999); US 6500669 (2002).
- D Wang, R Hara, G Singh, A Sancar, S J Lippard (2003) Biochem 42: 6747.
- N Carte, F Legendre, E Leize (2000) Anal Biochem 284: 77.
- J A Mello, S Acharya, R Fishel, J M Essigmann (1996) Chem Biol 3, 579 (1996).
- U Warnke, J Gysler, B Hofte (2001) Electrophoresis 22: 97.
- S Yokoyama (1997) US 5646011; US 5846725 (1998); US 6046044 (2000).
- J E Šponer, J Leszczynski, F Glahe, B Lippert, J Šponer (2001) Inorg Chem 40, 3269.
- J Šponer, J E Šponer, L Gorb, J Leszczynski, B Lippert (1999) J Phys Chem 103A, 11406 (1999).
- J V Burda, J Šponer, J Leszczynski (2001) J Biol Inorg Chem 5, 178 (2000); Phys Chem Chem Phys 3, 4404.
- J V Burda, J Šponer, J.Hrabáková, M Zeizinger, J Leszczynski (2003) J Phys Chem 107B: 5349.
- R K O Sigel, E Freisinger, B Lippert (2000) J Biol Inorg Chem 5: 287
- K S Schmidt, J Reedijk, K Weisz, E M B Janke, J E Šponer, et al. (2002) Inorg Chem 41: 2855.
- J V Burda, M Zeizinger, J Leszczynski (2005) J Comput Chem 26: 907
- L A S Costa, W R Rocha, W B De Almeida, H F Dos Santos (2004) Chem Phys Lett 387: 182
- M A Elizondo-Riojas, J Kozelka (2001) J Mol Biol 314: 1227.
- M H Baik, R A Friesner, S J Lippard (2003) J Am Chem Soc 124: 4495 (2002); J Am Chem Soc 125: 14082 (2003); Inorg Chem 42: 8615
- J V Burda,, J Leszczynski (2003) Inorg Chem 42: 7162.
- D V Deubel (2004) J Am Chem Soc 124: 5834 (2002); J Am Chem Soc 126: 5999.
- J K C Lau, D V Deubel (2005) Chem - Eur J 11: 2849.
- S Rafique, M Idrees, A Nasim, H Akbar, A Athar (2010) Biotechnol Mol Biol Rev 5(2): 38.
- G Marcon, S Carotti, M Coronnello, L Messori, E Mini, et al. (2002) J Med Chem 45: 1672.
- L Messori, F Abbate, G Marcon, P Orioli, M Fontani, et al. (2000) J Med Chem 43: 3541.
- Y Zheng, L Sanche (2009) Radiat Res 172(1): 114.
- S Kapoor (2009) J Cell Biochem 106(2): 193.
- K I Ansari, J D Grant, S Kasiri, G Woldemariam, B Shrestha, S S Mandal (2009) J Inorg Biochem 103(5): 818 (2009).
- M J Clarke (2005) Coord Chem 236: 299.
- M Galanski, V B Arion, M A Jakupec, B K Keppler (2003) Curr Pharm 9: 2078.
- C S Allardyce, P J Dyson, D J Ellis, S L Heath (2001) Chem commun 1396.
- P Pigon, S Top, A Vessières, M.Huché, E A.Hillard, et al. (2005) J Med Chem 48:
- H C Kiat, K L Weng, J Gerard, L Lawrence, T Siden, et al. (2006) J Organomet Chem 69: 9.
- L R.Hirsch, J B Jackson, A Lee, N J Halas, J L West (2003) Anal Chem 75: 2377.
- C Loo, A Lin, L Hirsch, M H Lee, J Barton, et al. (2004) Cancer Res Treat 3(1): 33.
- P Podsiadlo, V A Sinani, J H Bahng, N W Kam, J Lee, et al. (2008) Langmuir 24(2): 568.
- A K Saxena, F Huber (1989) Coord Chem Rev 95: 109.
- P Koepf-Maier, H Kopef (1987) Chem Rev 87: 1137.
- G Sava, S Zorzet, T Giraldi, G MestroniandG Zassinovich (1984) Eur J Cancer Clin Oncol 20: 841.
- T Boulikas, A Pantos, E.Bellis, P Christofis (2007) Cancer Therapy 5: 537.
- A -M Florea, D Büsselberg (2011) Cancers (Basel) 3(1): 1351.
- X Wang, X Wang, Z Guo (2015) Acc Chem Res 48(9): 2622.
- Z W Li, H Taube (1992) Science 256: 210.
- P Umapathy (1989) Coord Chem Rev 95: 129.
- L Y Kuo, M G Kanatzidis, M Sabat, A L Tipton, T J Marks (1991) J Am Chem Soc 113: 9027.
- E Alessio, Y Xu, S Cauci, G Mestroni, F Quadrifoglio, et al. (1989) J Am Chem Soc 111: 7068.
- K J Barnham, M I Djuran, P S Murdoch, P J Sadler (1994) J Chem Soc Chem Commun 721.
- K J Barnham, M I Djuran, P S Murdoch, J D Ranford, P J Sadler (1996) J Chem Soc Dalton Trans 3721 (1995); Inorg Chem 35: 1065.
- A S Petrov, G Lamm, G R Pack (2005) Biopolymers 77: 137.
- A Ono, S Cao, H Togashi, M Tashiro, T Fujimoto, et al. (2008) Chem Commun 4825.
- A Ono, H Togashi (2004) Angew Chem Int Ed 43: 4300.
- J Liu, Y Lu (2007) Angew Chem Int Ed 46: 7587.
- X Liu, Y Tang, L Wang, J Zhang, S Song, et al. (2007) Adv Mat 19: 1471.
- R Freeman, T Finder, I Willner (2009) Angew Chem Int Ed 48: 7818.
- T Carell, C Behrens, J Gierlich (2003) Org Biomol Chem 1: 2221.
- J S Lee, L J Latimer, R S Reid (1993) Biochem Cell Biol 71: 162.
- P Aich, S L Labiuk, L W Tari, L J T Delbaere, W J Roesler, K J Falk, R P Steer, J S Lee (1999) J Mol Biol 294: 477.
- A Rakitin, P Aich, C Papadopoulos, Y Kobzar, A S Vedeneev, et al. (2001) Phys Rev Lett 86: 3670.
- F Moreno-Herrero, P Herrero, F Moreno, J Colchero, C Gómez-Navarro, et al. (2003) Nanotechnology 14: 128.
- M Fuentes-Cabrera, B G Sumpter, J E Šponer, J Šponer, L Petit, et al. (2007) J Phys Chem 111B: 870.
- S S Alexandre, J M Soler, L Seijo, F Zamora (2006) Phys Rev 73B: 205112
- S L Labiuk, L T J Delbaere, J S Lee (2003) J Biol Inorg Chem 8: 715.
- Y Tanaka, S Oda, H Yamaguchi, Y Kondo, C Kojima, et al. (2007) J Am Chem Soc 129: 244.
- S Johannsen, N Megger, D Böhme, R K O Sigel, J Müller (2010) Nature Chem 2: 229.
- Y Miyake, H Togashi, M Tashiro, H Yamaguchi, S Oda, et al. (2006) J Am Chem Soc 128: 2172.
- E Ennifar, P Walter, P Dumas (2003) Nucleic Acids Res 31: 2671.
- D Böhme, N Düpre, D A Megger, J Müller (2007) Inorg Chem 46: 10114.
- K Tanaka, A Tengeiji, T Kato, N Toyama, M Shiro, et al. (2002) J Am Chem Soc 124: 12494.
- G H Clever, C Kaul, T Carell (2007) Angew Chem Int Ed 46: 6226.
- G H Clever, K Polborn, T Carell (2005) Angew Chem Int Ed 44: 7204.
- F H Westheimer (1968) Acc Chem Res 1: 70.
- G A Soukup, R R Breaker (1999) RNA 5: 1308.
- R S Brown, J C Dewan, A Klug (1985) Biochemistry 24: 4785.
- R Breslow, DL Huang (1991) Proc Natl Acad Sci 88: 4080.
- C Werner, B Krebs, G Keith, G Dirheimer (1976) Biochim Biophys Acta 432 161.
- R S Brown, B A Hingerty, J C Dewan, A Klug (1983) Nature 303: 543.
- J Ciesiolka, W D Hardt, J Schlegl, V A Erdmann, R K Hartmann (1994) Eur J Biochem 219: 49.
- M Hertweck, M W Müller (2001) Eur J Biochem 268: 4610.
- J Ciesiolka, T Marciniec, W J Krzyzosiak (1989) Eur J Biochem 182: 445.
- R K O Sigel, A Vaidya, A M Pyle (2000) Nat Struct Biol 7: 1111.
- R K O Sigel, A M Pyle (2003) Met Ions Biol Syst 40: 477.
- C Berens, B Streicher, R Schroeder, W Hillen (1998) Chem Biol 5: 163.
- R Das, K J Travers, Y Bai, D Herschlag (2005) J Am Chem Soc 127: 8272.
- I T Suydam, S A Strobel (2009) Methods Enzymol 468: 3.
- M C Erat, R K O Sigel (2011) Met Ions Life Sci 9: 37.
- E L Christian, M Yarus (1993) Biochemistry 32: 4475.
- S Basu, S A Strobel (2001) Method 23: 264.
- J A Jansen, T J McCarthy, G A Soukup, J K Soukup (2006) Nature Struct Mol Biol 13: 517.
- V D Sood, T L Beattie, R A Collins (1998) J Mol Biol 282: 741.
- C P Da Costa, D Krajewska, A Okruszek, W J Stec, H Sigel (2002) J Biol Inorg Chem 7: 405.
- C P Da Costa, A Okruszek, H Sigel (2003) Chem Bio Chem 4: 593.
- J K Frederiksen, R Fong, J A Piccirilli (2009) “In Nucleic Acid-Metal Ion Interactions”, Ed N V Hud, Royal Society of Chemistry, Cambridge, UK.
- J L Leroy, M Guéron (1977) Biopolymers 16: 2429.
- T E Horton, D R Clardy, V J DeRose (1998) Biochemistry 37: 18094.
- N Kisseleva, S Kraut, A Jaschke, O Schiemann (2007) HFSP J 1: 127.
- L Hunsicker-Wang, M Vogt, V J DeRose (2009) Methods Enzymol 468: 335.
- V J DeRose (2009) “In Nucleic Acid-Metal Ion Interactions”, Ed N V Hud, Royal Society of Chemistry, Cambridge UK.
- M Vogt, S Lahiri, C G Hoogstraten, R D Britt, V J DeRose (2006) J Am Chem Soc 128: 16764.
- J E Penner-Hahn (2005) Coord Chem Rev 249: 161.
- I V Smirnov, F W Kotch, I J Pickering, J T Davis, R H Shafer (2002) Biochemistry 41: 12133.
- K S Koutmou, A Casiano-Negroni, M M Getz, S Pazicni, A J,rews, et al. (2010) Proc Natl Acad Sci 107: 2479.
- -K Teo, P Eisenberger, J Reed, J K Barton, S J Lippard (1978) J Am Chem Soc 100: 3225.
- A P Hitchcock, C J L Lock, W M C Pratt (1982) Inorg Chim Acta 66: 45.
- J M Benevides, S A Overman, G J Thomas (2005) J Raman Spec 36: 279.
- T Theophanides (2000) “In Infrared, Raman Spectroscopy of Biological Materials”, Ed H.-U.Gremlich, B Yan, M Dekker, New York.
- Y Chen, N V Eldho, K T Dayie, P R Carey (2010) Biochemistry 49: 3427.
- H Takeuchi, H Murata, I Harada (1988) J Am Chem Soc 110: 392.
- H A Tajmir-Riahi, T Theophanides (1985) J Biomol Struct Dyn 2, 995 (1985) ; J Biomol Struct Dyn 3: 537.
- S Adam, P Bourtayre, J Liquier, E Taillandier (1986) Nucleic Acids Res 14: 3501.
- M Langlais, H A Tajmir-Riahi, R Savoie (1990) Biopolymers 30: 743.
- J Duguid, V A Bloomfield, J Benevides, G J Thomas (1995) Biophys J 65, 1916 (1993); Biophys J 69: 2623.
- H Arakawa, J F Neault, H A Tajmir-Riahi (2001) Biophys J 81: 1580.
- J Stangret, R Savoie (2002) Phys Chem Chem Phys 4: 4770.
- S Nafisi, Z Norouzi (2009) DNA Cell Biol 28: 469.
- S Nafisi, A Sobhanmanesh, K Alimoghaddam, A Ghavamzadeh, H A Tajmir-Riahi (2005) DNA Cell Biol 24: 634.
- E L Christian, V E,erson, P R Carey, M E Harris (2010) Biochemistry 49: 2869.
- H Chen, B Gong, P C Bevilacqua, P R Carey, B L Golden (2009) Biochemistry 48: 1498.
- B Gong, J H Chen, E Chase, D M Chadalavada, R Yajima, et al. (2007) J Am Chem Soc 129: 13335.
- B Gong, Y Chen, E L Christian, J H Chen, E Chase, et al. (2008) J Am Chem Soc 130: 9670.
- A Wong, R Ida, G Wu (2005) Biochem Biophys Res Commun 337: 363.
- M L Gill, S A Strobel, J P Loria (2005) J Am Chem Soc 127” 16723.
- R L Gonzalez Jr, I Tinoco Jr (2002) J Mol Biol 289, 1267 (1999); Methods Enzymol 338: 421.
- J Feigon, S E Butcher, L D Finger, N V Hud (2001) Methods Enzymol 338: 400.
- R K O Sigel, D G Sashital, D L Abramovitz, A G Palmer, S E Butcher, A M Pyle (2004) Nat Struct Mol Biol 11: 187.
- M C Erat, O Zerbe, T Fox, R K O Sigel (2007) Chem Bio Chem 8: 306.
- S E Butcher, F H T Allain, J Feigon (2000) Biochemistry 39: 2174.
- S S Wijmenga, B N M van Buuren (1998) Prog Nucl Mag Res Sp 32: 287.
- B A Connolly, F Eckstein (1984) Biochemistry 23: 5523.
- M Maderia, L M Hunsicker, V J DeRose (2000) Biochemistry 39: 12113.
- E M Osborne, W L Ward, M Z Ruehle, V J DeRose (2009) Biochemistry 48: 10654.
- Y Tanaka, C Kojima, E H Morita, Y Kasai, K Yamasaki, A Ono, M Kainosho, K Taira (2002) J Am Chem Soc 124: 4595.
- G Wang, B L Gaffney, R A Jones (2004) J Am Chem Soc 126: 8908.
- G W Buchanan, J B Stothers (1982) Can J Chem 60: 787.
- I Bertini, C Luchinat (1986) “NMR of Paramagnetic Molecules in Biological Systems “,Benjamin/Cummings Pub Co Menlo Park, CA (1986).
- I Bertini, C Luchinat, G Parigi(2001) “Solution NMR of Paramagnetic Molecules – Applications to Metallobiomolecules, Models”, Elsevier Science B.V Amsterdam.
- J H Davis, T R Foster, M Tonelli, S E Butcher (2007) RNA 13: 76.
- J Noeske, H Schwalbe, J Wöhnert (2007) Nucleic Acids Res 35: 5262.
- D O Campbell, P Bouchard, G Desjardins, P Legault (2006) Biochemistry 45: 10591.
- R E Hurd, E Azhderian, B R Reid (1979) Biochemistry 18: 4012.
- J A Cowan (1993) J Inorg Biochem 49: 171.
- M Schmitz, I Tinoco (2000) Jr RNA 6: 1212.
- G Colmenarejo, I Tinoco Jr (1999) J Mol Biol 290: 119.
- J S Kieft, I Tinoco Jr Structure 5: 713.
- S Rüdisser, I Tinoco Jr (2000) J Mol Biol 295: 1211.
- P Schultze, N V Hud, F W Smith, J Feigon (1999) Nucleic Acids Res 27: 3018.
- F W Smith, J Feigon (1993) Biochemistry 32: 8682.
- N V Hud, P Schultze, V Sklenár, J Feigon (1999) J Mol Biol 285: 233.
- D M Rose, C F Polnaszek, R G Bryant (1982) Biopolymers, 21: 653.
- S S Reid, J A Cowan (1990) Biochemistry, 29, 6025.
- E Berggren, L Nordenskiöld, W H Braunlin (1992) Biopolymers 32: 1339.
- J A Cowan, H W Huang, L Y Hsu (1993) J Inorg Biochem 52: 121.
- L A Wright, L E Lerner (1994) Biopolymers 34: 691.
- L M Utschig, J W Bryson (1995) T V O'Halloran, Science 268: 380.
- J E Coleman (1993) Methods Enzymol 227: 16.
- R Ida, G Wu (2008) J Am Chem Soc 130: 3590.
- J P,ré, H R Mäcke (2003) J Inorg Biochem 97: 315.
- Y Tanaka, Y Kasai, S Mochizuki, A Wakisaka, E H Morita, et al. (2004) J Am Chem Soc 126: 744.
- J Reedijk (2008) Platinum Met Rev 52: 2.
- L Helm, A E Merbach (1999) Coord Chem Rev 187: 151.
- V K Misra, D E Draper (2002) J Mol Biol 317: 507.
- D E Draper (2004) RNA 10: 335.
- M J Serra, J D Baird, T Dale, B L Fey, K Retatagos, E Westhof (2002) RNA 8: 307.
- Y V Bukhman, D E Draper (1997) J Mol Biol 273: 1020.
- R Shiman, D E Draper (2000) J Mol Biol 302: 79.
- L G Laing, T C Gluick, D E Draper (1994) J Mol Biol 237: 577.
- C Hammann, A Cooper, D M J Lilley (2001) Biochemistry 40: 1423.
- S O Shan, A Yoshida, S Sun, J A Piccirilli, D Herschlag (1999) Proc Natl Acad Sci 96: 12299.
- S Wang, K Karbstein, A Peracchi, L Beigelman, D Herschlag (1999) Biochemistry 38: 14363.
- P M Gordon, E J Sontheimer, J A Piccirilli (2000) Biochemistry 39: 12939.
- S I Nakano, D J Proctor, P C Bevilacqua (2001) Biochemistry 40: 12022.
- A K Brown, J Li, C M B Pavot, Y Lu (2003) Biochemistry 42: 7152.
- J M Wilcox, D L Rempel, M L Gross (2008) Anal Chem 80: 236.
- D Grilley, A M Soto, D E Draper (2009) Methods Enzymol 455: 71.
- H Krakauer (1971) Biopolymers 10: 2459.
- R Römer, R Hach (1975) Eur J Biochem 55: 271.
- Y Bai, M Greenfeld, K J Travers, V B Chu, J Lipfert, et al. (2007) J Am Chem Soc 129: 14981.
- M Greenfeld, D Herschlag (2009) Methods Enzymol 469: 375.
- A Polak, H J Scholer (1975) Chemotherapy 21: 113.
- A Cox (1968) Quart Rev 22, 934.
- O N Al Safarjalani, X J Zhou, R H Ras, J Shi, R F Schinazi, et al. (2005) Cancer Chemother Pharmacol 55: 541.
- G B Elion (1967) Fed Proc 26: 898.
- J H Burchenal (1953) Blood 8: 965.
- B D Clarkson (1970) Cancer 5: 227.
- S A Giller, R A Zhuk, M I U Lidak (1967) Dokl Akad Nauk SSR 176: 332.
- J L Ambrus, I Stadler, M Kulaylat, A Koreshi, S Akhtar (1996) J Med Chem 27: 21.
- M Weller, B Muller, R Koch, M Bamberg, P Krauseneck (2003) J Clin Oncol 21: 3276.
- M Horton (2005) Clin Cancer Res 11: 1884.
- B J Kennedy, J L Torkelson, E Torlakovic (1999) Cancer 85: 2265.
- J R Bertino (1979) Biochem Pharmacol 28: 1983.
- L W Hertel, G B Border, J S Kroin, S M Rinzel, G A Poore, et al. (1990) Cancer Res 50: 4417.
- A Agrawal, K Srivastava, S K Puri (2005) Bio Org Med Chem Lett 15: 5218.
- T C Daniels, E C Jorgensen (1982) “Central nervous system depressents in Wilson, Gisvoids Textbook of Organic medicinal, Pharmaceutical Chemistry”,ed R F Doerge, , J B Lippen-Cott, Philadelphia 33.
- D S Threlkeld (1998) Facts, Comparisons: 269.
- J Vida, J Yevich (2003) “Sedative hypnotics, In Burger's Medicinal Chemistry, Drug Discover” , 6thed D J Abrahim, John Wiley, New Jersy (6): 203.
- C J Shishoo, U S Pathak, K S Jain, I T Devani, M T Chhabria (1994) Chem Inform 25(38): 436.
- M S Masoud, M Sh Ramadan, A El-Samahy, I Mahmoud, M H Al-Saify (2017) Advanced Materials Science 2(4): 1.
- M S Masoud, M Sh Ramadan, M H Al-Saify (2018) Pharm Anal Chem 4(1): 1.
- M S Masoud, M Sh Ramadan, A H El-Sayed, A M Sweyllam, M H Al-Saify (2018) Arc Org Inorg Chem Sci 3(5): 410.
- M S Masoud, M F Fathalla, R H A Mohamed, M S Hassan (2019) Asian J of Chem 31(3): 733.
- M S Masoud, A S El-Kholany, M F El-Shahat (2019) J of Chemical, Pharmaceutical Research 11(12):
- M Schena, D Shalon, RW Davis, PO Brown (1995) Sceince 270: 467
- S Yamakawa, K,o, A Chisaka, K Yashida, A Shinmyo, T Kohchi (2004) J Biosci Bioeng 98: 140.
- N Mera, H Aoyagi, S Nakasona, K Iwasaki, H Saiki (2004) J Biosci Bioeng 97: 169.
- B Goswami, B L Gaffney, R A Jones (1993) J Am Chem Soc 115: 3832.
- C Wang, H Gao, B.L Gaffney, R A Jones (1991) J Am Chem Soc 113: 5486.
- B L Gaffney, C Wang, R A Jones (1992) J Am Chem Soc 114: 4047.
- Y S Rhee, C Wang, BL Gaffney, R A Jones (1993) J Am Chem Soc 115: 12607.
- X Zhang, B L Gaffney, RA Jones (1998) J Am Chem Soc 120: 615.
- WLF Armarego (1967) In “The Chemistry of Heterocyclic Compounds, Fused Pyrimidines”, Part I: “Quinazolines”, D.J Brown, Ed.; Vol 24/1, Interscience Publishers: New York– London – Sydney.
- M Legraverend (2008) Tetrahedron 64: 8585.
- W B Parker (2009) Chem Rev 109(7): 2880.
- S Ostrowski (2001) Polish J Chem 75, 1661, Jordan J Chem 4(1): 1(2009).
- A C Tella, J A Obaleye (2010) Int J Chem Sci 8(3): 1675.
- J Cieplik, M Stolarczyk, J Pluta, O Gubrynowicz, I Bryndal, et al. (2011) Acta Poloniae Pharmaceutica, Drug Research 68(1): 57.
- L Zhuo, K Kou, Y Wang, H Chen (2015) Designed monomers, polymers 18(1): 42.
- Y Liang, S F Wnuk (2015) Molecules 20: 4874.
- M S Masoud, A M Heiba, F M Ashmawy (1983) Trans Met Chem 8: 124.
- M S Masoud, T.M Salem, Z Zaki (1984) XXIII International Conference on Coordiantion Chemistry, Colorado, USA, p. 372.
- A A Hasanein, M S Masoud, A M Heiba (1985); (1987) Current Science 54(2): 1165; J Chem Soc Pak 9(2): 199.
- M S Masoud, S S Haggag (1985) 30th IUPAC Congress, Manchester 9-13 September (1985); 8th International symposium on solute-solute-solvent interaction, p 210, p 303, University of Regensburg, 9-10 August (1987); 3rd National Symposium on Grystallography, Cairo, Academy of Scientific Research, Technology, National Committe of Technology, 26-27 January (1988); 5th World Conference on Thermal Analysis, Corfu, Greece, May 16-17 (1988); 9th Arabian Chemistry Conference, Kwuit, 15-19 December (1990); Thermochimica Acta, 196,221 (1992).
- M S Masoud, N A Ibrahim, S A AbouAli, G Y Ali, I M Abed, Ind J Chem 25A, 389.
- M S Masoud, S A Abou Ali, G Y Ali, IM Abed (1987) Thermochim Acta 122, 209.
- M S Masoud, M E Kassem, Y Abd El-Aziz, S Massoud (1987) Xth Conference on Solid State Science, Applications, p 46.
- M A El-Dessouky, M S Masoud, F A Ali, S A Abou El-Enein, Affinidad (1988) 416, August 321; 2nd Chemistry Conference, Fac of Sci Alex Univ 28-30 June, 178,196 (1988); Trans Met Chem 15, 443 (1990); XXVII Colloqium spectroscopicum international, Bergen, Norway, 9-14 June (1991).
- M S Masoud, M Khater, N El-Guindi (1988) J Iraqi Chem Soc 13, 177.
- M S Masoud, E.M.Soliman, A E El-Kholy, E A Khalil (1988) Thermochem Acta 136: 1.
- M S Masoud, Z M Zaki (1988) Trans Met Chem 13: 321; Bull Fac Sci Mansoura Univ 17(1): 71 (1990).
- M S Masoud, E M Soliman, A M Heiba (1989) Trans Met Chem 14: 175.
- M S Masoud, S Abou El-Enein (1989) Thermochim Acta 140, 365.
- M S Masoud, E A Khalil, A R Youssef (1990) Synth React Inorg Met.-Org Chem 20(6): 793.
- M S Masoud, S S Haggag, E M Soliman, M El-Shabasy (1991) J Mater Sci 26: 1109.
- M S Masoud, S A Abou El-Enein, E El-Shereafy, J Therm Anal 37: 365 (1991).
- M S Masoud, E A Khalil, Polish J Chem 65: 933 (1991).
- M S Masoud, M E Kassem, Y Abd El-Aziz, S M Khalil (1991) Bull Fac Sci Mansoura University 18(1), 105.
- M S Masoud, E A Khalil, S S Haggag, Pak J Sci Ind Res 35,480 (1992); Nucleosides, Nucleotides, Nucleic Acids 25(1), 73 (2006).
- M S Masoud, S A Abou El-Enein, O F Hafez (1992) J Therm Anal 38: 1365.
- M S Masoud, M M El-Essawi, A M Amr, XXI Congress on Molecular Spectroscopy, EUCMOS XXI, Technical University of Vienna, Austria, 23-28 August (1992); Anal Proc 29: 370.
- E A Khalil, M S Masoud, A El-Marghany (1993) Pak J Sci Ind Res 36: 68.
- M S Masoud, E El-Shereafy, E A Khalil, O H Abd El-Hamid (1993) Egypt J Anal Chem 2(1): 95.
- M S Masoud, Z M Zaki, F M Ismail, A K Mohamed (1994) Z für Phys Chem Bd 185, S 223.
- M S Masoud, O H El-Hamid, Z M Zaki (1994) Trans Met Chem 19: 21.
- M S Masoud,S S Haggag, Z M Zaki, M EI-Shabasy (1994) Spectrosc Lett 27(6): 775.
- S A Abou EI-Enein, M S Masoud, A El-Khatib, S Abd El-Aziz (1994) Alexandria Engineering J 33(3): D103.
- M S Masoud, O.F Hafez, N.A Obeid (1994) Pak J Sci Ind Res 37(10): 421.
- M S Masoud, H.M El-Nahas, S S Haggag Pak (1995) J Sci Ind Res 38(3-4): 108.
- M S Masoud, S.S Haggag, O H Abd El-Hamid, Revue Roumaine De Chimie(1996) 41(1-2), June 21-27.
- M S Masoud, A K Ghonaim, A A Mahmoud, 1st International Conference on Basic Sciences, Advanced Technology, Faculty of Science, Assiut University, "A 5-1,AP8", "P 24, 46",10-11 Nov.(1996); 2nd Mediterranean Basic Conference on Analytical Chemistry 23-28 Nov Robat (Morocco) (1997); 36th Annual Eastern Analytical Symposium & Exposition, Somerset, New Jersey, USA; No 525, p.27,16- 21 Nov (1997)
- M S Masoud, M M Ghonaim, O A El-Wahab (1996) Egypt J Appl Sci 11(8): 302.
- M S Masoud, R H A El-Sayed, A H Mostafa, N H Abd El Moneium (1999) The 5th International Conference of Chemistry, its Role in Development, Department of Chemistry, Faculty of Science, El Mansura University.
- M S Masoud, S A Abou El- Enein, H M Kamel (1999) 3rd International Scientific Conference (Science, Development, Environment), Faculty of Science, Al-Azhar University, p.72, 22- 25 March; Ind J Chem 41A: 297.
- M S Masoud, A El Marghany, S K El Sadany, N G Ali (1999) The 5th International Conference of Chemistry, its Role in Development, Department of Chemistry, Faculty of Science, El Mansura University, 19-22 April.
- M S Masoud, A A Hasanein, A K Ghonaim, E A Khalil, A A Mahmoud (1999) Z für Phy Chem Bd 209 S, 223.
- M S Masoud, A M Hindawy, R H Ahmed (1999) Pak J Sci Ind Res 42(1): 11.
- M S Masoud, E A Khalil, A.A Ibrahim, A El-Marghany (2007) Z für Phys Chem Bd 211S, 13 (1999); Spectrochim Acta 67A, 662.
- M S Masoud, S A Abou El-Enein, NA Obeid (2001) Z für Phys Chem 215: 7 867.
- M S Masoud, A K Ghonaium, R H Ahmed, A A Mahmoud, A E Ali (2001) Z für Phys Chem 215(4): 531.
- M S Masoud, A K Ghonaim, R H Ahmed S A Abou El-Enein, A A Mohmoud (2002) J Coord Chem 55(1): 79.
- M S Masoud, A A.Soayed, A E Ali, O K Sharsherah (2003) J Coord Chem 56(8): 725.
- M S Masoud, A A Soayed, A E Ali (2004) Spectrochim Acta, 60A: 1907.
- M S Masoud, S A Abou El-Enein, M Ayad, A S Goher (2004) Spectrochim Acta 60A: 77.
- M S Masoud, A M Hindawey, E A Khalil, A M Ramadan (2005) Bull Fac Sci Assuit University 33(2B), l-8 (2004); Can J Anal Sci Spectrosc 50(6): 297.
- M E Mahmoud, M S Masoud, N N Maximous (2004) Mikrochim Acta 147(1-2): 111.
- M S Masoud, A E Ali, M A Shaker, M Abdul Ghani (2004) Spectrochim Acta 60A: 3155.
- M S Masoud, E A Khalil, A M Hafez, A F El-Husseiny (2005) Spectrochim Acta 61A: 989.
- M S Masoud, E A Khalil, O H Abd El-Hamid, A A Soayed (2005) The Egyptian Science Magazine 2(2): 33.
- M S Masoud, M F Amira, S A El-Moneim, G M Moghazy, A A Abou-Hagar, et al. (2005) The Egyptian Science Magazine 2(4): 88.
- M S Masoud, T S Kasem, M A Shaker, A A Ali (2006) J Therm Anal Calorim 84(3): 549.
- M S Masoud, A A Soayed, A S El-Kholany, Proceeding of the 3rd international Chemical & Environmental Engineering Conference, 16-18 May (2006) J of Radioanalytical, Nuclear Chemistry 1-8 (2011).
- M S Masoud, E A Khalil, A M Ramadan, Y M Gohar, A Sweyllam (2007) Spectrochim Acta 67A: 669.
- M S Masoud, M Sh Ramadan, MH Al-Saify (2006) Chem-04, Faculty of Science, Cairo University, p.50: 5-8. International Conference on Science, Technology, Prauge, Czech Republic: 5-6.
- M S Masoud, M F Amira, A.M Ramadan, Gh M El-Ashry (2008) Spectrochim.Acta, 69A, 230.
- H H Hammud, K H Bouhadir, M S Masoud, A M Ghannoum, SA Assi ((2008)) Journal of Solution Chemistry 37(7): 895.
- M S Masoud, S A Abou El-Enein, AM Ramadan, A S Goher (2008) J Anal Appl pyrolysis 81(1): 45.
- M S Masoud, M Sh Ramadan, A El-Samahy, I Mahmoud, M H Al-Saify (2008) Chem-05, Faculty of Science, Cairo University, p.48, 3-5 March.
- M T Zaworotko, H H.Hammud, G Mc- Manus, A M Ghannoum, A Kabbani, et al. (2009) J Chem Crystall 39: 853.
- M S Masoud, A El-Merghany, M Y Abd El-Kaway (2009) International Conference on Chemistry, its Role in Development, Department of Chemistry, Faculty of Science, University of Mansoura, (16-19) April (2007); Synthesis, Reactivity in Inorganic, Metal-Organic, Nano-Metal Chemistry, 39 (9): 537.
- M S Masoud, A El-Merghany, A M Ramadan, M Y Abd El-Kaway (2010) J Therm Anal.Calorim 101(3): 839.
- M S Masoud, M K Awad, M A Shaker, M M T El-Tahawy (2010) Corrosion Science 52(7): 2387.
- M S Masoud, M.A Shaker, A E Ali, G S Elasala (2012) Seventh International Scientific Conference, Environment, Development, Nanotechnology, Faculty of Science, Al-Azhar University, Cairo, Egypt, 170: 22-24, Spectrochim Acta, 90A: 93.
- M S Masoud, A.M Hindawy, A A Soayed, M Y Abd El-Kaway (2012) J Fluid Phase Equilibria, 312: 37 (2011); Research Conference for Egyptian Faculties of Science, Alexandria University, Session 6, OR-11 (18-19) May (2011); Spectrochim Acta, 92A, 15: 256.
- M S Masoud, A A Soayed, A F El-Husseiny (2012) Spectrochim Acta 99A: 365.
- N Z Shaban, M S Masoud, M A Mawlawi, D Awad, O M Sadek (2012) J Physiol Biochem 68(4): 475.
- M S Masoud, M Y Abd El-Kaway (2012) Spectrochim Acta 102A: 175.
- M S Masoud, A El-Marghany, A Orabi, A E Ali, R Sayed (2013) Spectrochim Acta 107A: 179.
- M K Awad, M S Masoud, M A Shaker, A E Ali, M M T El-Tahawy (2013) Research on Chemical Intermediates 39(6): 2741.
- M S Masoud, A E Ali, M Y Abd El-Kaway (2014) J Therm Anal Calorim 116(1): 183.
- M S Masoud, M F El-Shahat, A S Elkholany (2014) Spectrochim Acta, 127A: 216.
- D J Brown (1994) In “The Chemistry of Heterocyclic Compounds, The Pyrimidines”, Taylor, E.C Ed.; Vol 23, J Wiley & Sons: New York – Chichester – Brisbane – Toronto –Singapore.
- A Albert (1985) In “Selective Toxicity: The Physico-Chemical Basis of Therapy”, 7th ed N.B Chapman; J.E Hall Eds.; London, p 347.
- J H Lister (1996) In “The Chemistry of Heterocyclic Compounds, The Purines”, Supplement I; Wiley – Interscience: New York – Chichester – Brisbane – Toronto – Singapore.
- B Bobrański, Z Synowiedzki (1948) J Am Pharm Assoc Sci Edu 37: 62.
- H Rosenmeyer (2004) Chem Biodiv 1: 361.
- D K Goette (1981) J Am Acad Dermatol 4: 633.
- J Darbyshire, M Foulkes, R Peto, W Duncan, A Babiker, et al. (2004) “The Cochrane Library”, John Wiley & Sons, Chichester, Issue 2.
- P L Dordoni, L Frassanito, M F Bruno, R Proirtti, R De Cristofaro, et al. (2004) Br J Haematol 125: 79.
- M J Eadie, W D Hooper (2002) “Other Barbiturate-Methylphenobarbital”, In “Antiepileptic Drugs”, Eds E H Levy, R Mattson, B.S Meldrum, E Perucca, Lippincott-Williams, Wilkins, Philadelphia.
- H M McIntosh (2001) from “The Cochrane Library”, Update Software Ltd Issue 4.
- A M Joffe, J D Farley, D Linden, G Goldsand (1989) Am J Med 87: 332.
- T F Cash (1999) Br J Dermatol 141: 398.
- E Petersen, D R Schmidt (2003) Expert Rev Anticancer Ther 1: 175.
- R Schaumann (1996) In “Houben-Weyl: Hatarenes IV (Six-, Higher-Membered Ring Systems with Two or More Heteroatoms)”, Thieme-Interscience.
- C Orvig, M J Abrams (1999) Chem Rev 99: 2201.
- H Sakurai, Y Kojima, Y Yoshikawa, K Kawabe, H Yasui (2002) Coord Chem Rev 226: 187.
- W Koch, M C Holthausen (2002) “A Chemist’s Guide to Density Functional Theory”, 2nd ed Wiley-VCH, Weinheim, New York, Chicester.
- J E Šponer, V Sychrovský, P Hobza, J Šponer (2004) Phys Chem Chem Phys 6: 2772.
- V Sychrovský, J Šponer, P Hobza (2004) J Am Chem Soc 126: 663.
- C W Bock, A K Katz, G D Markham, J P Glusker (1999) J Am Chem Soc 121: 7360.
- E Freisinger, R K O Sigel (2007) Coord Chem Rev 251: 1834.
- H Sigel, R Griesser (2005) Chem Soc Rev 34: 875.
- B Knobloch, B Nawrot, A Okruszek, R K O Sigel (2008) Chem Eur J 14: 3100.
- H Sigel, R B Martin (1994) Chem Soc Rev 23: 83.
- M Roychowdhury-Saha, D H Burke (2006) RNA 12: 1846.
- D N Frank, N R Pace (1997) Proc Natl Acad Sci 94: 14355.
- W Wang, L P Billen, Y Li (2002) Chem Biol 9: 507.
- P J Bruesehoff, J Li, A J Augustine, Y Lu (2002) Comb Chem High T Scr 5: 327.
- J Li, W C Zheng, A H Kwon, Y Lu (2000) Nucleic Acids Res 28: 481.
- T Pan, B Dichtl, O C Uhlenbeck (1994) Biochemistry 33: 9561.
- H P Hofmann, S Limmer, V Hornung, M Sprinzl (1997) RNA 3: 1289.
- M Zivarts, Y Liu, R R Breaker (2005) Nucleic Acids Res 33: 622.
- R S Alexander, Z F Kanyo, L E Chirlian, D W Christianson (1990) J Am Chem Soc 112: 933.
- B Schneider, M.Kabeláč, P Hobza (1996) J Am Chem Soc 118: 12207.
- V V Murashov, J Leszczynski (1999) J Phys Chem 103B: 8391.
- M J Fedor (2002) Curr Opin Struct Biol 12: 289.
- J Šponer, M Sabat, L Gorb, J Leszczynski, B Lippert, et al. (2000) J Phys Chem 104B: 7535
- N Gresh, J E Šponer, N Špačková, J Šponer (2003) J Phys Chem 107B: 8669
- L Rulíšekand, J Šponer (2003) J Phys Chem 107B: 1913.
- E H S Anwander, M M Probst, B M Rode (1990) Biopolymers 29: 757
- J Šponer, J V Burda, M Sabat, J Leszczynski, P Hobza (1998) J Phys Chem 102A: 5951
- J Munoz, J Šponer, P Hobza, M Orozco, F J Luque (2001) J Phys Chem 105B: 6051.
- B Lippert (1999) “Cisplatin: chemistry, biochemistry of a leading anticancer drug “, Wiley-VCH, Weinheim, New York, Chichester, USA.
- B Rosenberg, L Vancamp, J E Trosko, V H Mansour (1969) Nature 222: 385.
- A M Pizarro, P J Sadler (2009) “In Nucleic Acid-Metal Ion Interactions”, Ed N V Hud, Royal Society of Chemistry, Cambridge, UK.
- K Hasegawa, K Mori, N Ishii, Y Aoki (2003) Chem Abstr 2478640.
- J M Essigmann R G Croy, K J Yarema, M Morningstar (2002) US 5882941 (1999); US 6500669 (2002).
- D Wang, R Hara, G Singh, A Sancar, S J Lippard (2003) Biochem 42: 6747.
- N Carte, F Legendre, E Leize (2000) Anal Biochem 284: 77.
- J A Mello, S Acharya, R Fishel, J M Essigmann (1996) Chem Biol 3, 579 (1996).
- U Warnke, J Gysler, B Hofte (2001) Electrophoresis 22: 97.
- S Yokoyama (1997) US 5646011; US 5846725 (1998); US 6046044 (2000).
- J E Šponer, J Leszczynski, F Glahe, B Lippert, J Šponer (2001) Inorg Chem 40, 3269.
- J Šponer, J E Šponer, L Gorb, J Leszczynski, B Lippert (1999) J Phys Chem 103A, 11406 (1999).
- J V Burda, J Šponer, J Leszczynski (2001) J Biol Inorg Chem 5, 178 (2000); Phys Chem Chem Phys 3, 4404.
- J V Burda, J Šponer, J.Hrabáková, M Zeizinger, J Leszczynski (2003) J Phys Chem 107B: 5349.
- R K O Sigel, E Freisinger, B Lippert (2000) J Biol Inorg Chem 5: 287
- K S Schmidt, J Reedijk, K Weisz, E M B Janke, J E Šponer, et al. (2002) Inorg Chem 41: 2855.
- J V Burda, M Zeizinger, J Leszczynski (2005) J Comput Chem 26: 907
- L A S Costa, W R Rocha, W B De Almeida, H F Dos Santos (2004) Chem Phys Lett 387: 182
- M A Elizondo-Riojas, J Kozelka (2001) J Mol Biol 314: 1227.
- M H Baik, R A Friesner, S J Lippard (2003) J Am Chem Soc 124: 4495 (2002); J Am Chem Soc 125: 14082 (2003); Inorg Chem 42: 8615
- J V Burda,, J Leszczynski (2003) Inorg Chem 42: 7162.
- D V Deubel (2004) J Am Chem Soc 124: 5834 (2002); J Am Chem Soc 126: 5999.
- J K C Lau, D V Deubel (2005) Chem - Eur J 11: 2849.
- S Rafique, M Idrees, A Nasim, H Akbar, A Athar (2010) Biotechnol Mol Biol Rev 5(2): 38.
- G Marcon, S Carotti, M Coronnello, L Messori, E Mini, et al. (2002) J Med Chem 45: 1672.
- L Messori, F Abbate, G Marcon, P Orioli, M Fontani, et al. (2000) J Med Chem 43: 3541.
- Y Zheng, L Sanche (2009) Radiat Res 172(1): 114.
- S Kapoor (2009) J Cell Biochem 106(2): 193.
- K I Ansari, J D Grant, S Kasiri, G Woldemariam, B Shrestha, S S Mandal (2009) J Inorg Biochem 103(5): 818 (2009).
- M J Clarke (2005) Coord Chem 236: 299.
- M Galanski, V B Arion, M A Jakupec, B K Keppler (2003) Curr Pharm 9: 2078.
- C S Allardyce, P J Dyson, D J Ellis, S L Heath (2001) Chem commun 1396.
- P Pigon, S Top, A Vessières, M.Huché, E A.Hillard, et al. (2005) J Med Chem 48:
- H C Kiat, K L Weng, J Gerard, L Lawrence, T Siden, et al. (2006) J Organomet Chem 69: 9.
- L R.Hirsch, J B Jackson, A Lee, N J Halas, J L West (2003) Anal Chem 75: 2377.
- C Loo, A Lin, L Hirsch, M H Lee, J Barton, et al. (2004) Cancer Res Treat 3(1): 33.
- P Podsiadlo, V A Sinani, J H Bahng, N W Kam, J Lee, et al. (2008) Langmuir 24(2): 568.
- A K Saxena, F Huber (1989) Coord Chem Rev 95: 109.
- P Koepf-Maier, H Kopef (1987) Chem Rev 87: 1137.
- G Sava, S Zorzet, T Giraldi, G MestroniandG Zassinovich (1984) Eur J Cancer Clin Oncol 20: 841.
- T Boulikas, A Pantos, E.Bellis, P Christofis (2007) Cancer Therapy 5: 537.
- A -M Florea, D Büsselberg (2011) Cancers (Basel) 3(1): 1351.
- X Wang, X Wang, Z Guo (2015) Acc Chem Res 48(9): 2622.
- Z W Li, H Taube (1992) Science 256: 210.
- P Umapathy (1989) Coord Chem Rev 95: 129.
- L Y Kuo, M G Kanatzidis, M Sabat, A L Tipton, T J Marks (1991) J Am Chem Soc 113: 9027.
- E Alessio, Y Xu, S Cauci, G Mestroni, F Quadrifoglio, et al. (1989) J Am Chem Soc 111: 7068.
- K J Barnham, M I Djuran, P S Murdoch, P J Sadler (1994) J Chem Soc Chem Commun 721.
- K J Barnham, M I Djuran, P S Murdoch, J D Ranford, P J Sadler (1996) J Chem Soc Dalton Trans 3721 (1995); Inorg Chem 35: 1065.
- A S Petrov, G Lamm, G R Pack (2005) Biopolymers 77: 137.
- A Ono, S Cao, H Togashi, M Tashiro, T Fujimoto, et al. (2008) Chem Commun 4825.
- A Ono, H Togashi (2004) Angew Chem Int Ed 43: 4300.
- J Liu, Y Lu (2007) Angew Chem Int Ed 46: 7587.
- X Liu, Y Tang, L Wang, J Zhang, S Song, et al. (2007) Adv Mat 19: 1471.
- R Freeman, T Finder, I Willner (2009) Angew Chem Int Ed 48: 7818.
- T Carell, C Behrens, J Gierlich (2003) Org Biomol Chem 1: 2221.
- J S Lee, L J Latimer, R S Reid (1993) Biochem Cell Biol 71: 162.
- P Aich, S L Labiuk, L W Tari, L J T Delbaere, W J Roesler, K J Falk, R P Steer, J S Lee (1999) J Mol Biol 294: 477.
- A Rakitin, P Aich, C Papadopoulos, Y Kobzar, A S Vedeneev, et al. (2001) Phys Rev Lett 86: 3670.
- F Moreno-Herrero, P Herrero, F Moreno, J Colchero, C Gómez-Navarro, et al. (2003) Nanotechnology 14: 128.
- M Fuentes-Cabrera, B G Sumpter, J E Šponer, J Šponer, L Petit, et al. (2007) J Phys Chem 111B: 870.
- S S Alexandre, J M Soler, L Seijo, F Zamora (2006) Phys Rev 73B: 205112
- S L Labiuk, L T J Delbaere, J S Lee (2003) J Biol Inorg Chem 8: 715.
- Y Tanaka, S Oda, H Yamaguchi, Y Kondo, C Kojima, et al. (2007) J Am Chem Soc 129: 244.
- S Johannsen, N Megger, D Böhme, R K O Sigel, J Müller (2010) Nature Chem 2: 229.
- Y Miyake, H Togashi, M Tashiro, H Yamaguchi, S Oda, et al. (2006) J Am Chem Soc 128: 2172.
- E Ennifar, P Walter, P Dumas (2003) Nucleic Acids Res 31: 2671.
- D Böhme, N Düpre, D A Megger, J Müller (2007) Inorg Chem 46: 10114.
- K Tanaka, A Tengeiji, T Kato, N Toyama, M Shiro, et al. (2002) J Am Chem Soc 124: 12494.
- G H Clever, C Kaul, T Carell (2007) Angew Chem Int Ed 46: 6226.
- G H Clever, K Polborn, T Carell (2005) Angew Chem Int Ed 44: 7204.
- F H Westheimer (1968) Acc Chem Res 1: 70.
- G A Soukup, R R Breaker (1999) RNA 5: 1308.
- R S Brown, J C Dewan, A Klug (1985) Biochemistry 24: 4785.
- R Breslow, DL Huang (1991) Proc Natl Acad Sci 88: 4080.
- C Werner, B Krebs, G Keith, G Dirheimer (1976) Biochim Biophys Acta 432 161.
- R S Brown, B A Hingerty, J C Dewan, A Klug (1983) Nature 303: 543.
- J Ciesiolka, W D Hardt, J Schlegl, V A Erdmann, R K Hartmann (1994) Eur J Biochem 219: 49.
- M Hertweck, M W Müller (2001) Eur J Biochem 268: 4610.
- J Ciesiolka, T Marciniec, W J Krzyzosiak (1989) Eur J Biochem 182: 445.
- R K O Sigel, A Vaidya, A M Pyle (2000) Nat Struct Biol 7: 1111.
- R K O Sigel, A M Pyle (2003) Met Ions Biol Syst 40: 477.
- C Berens, B Streicher, R Schroeder, W Hillen (1998) Chem Biol 5: 163.
- R Das, K J Travers, Y Bai, D Herschlag (2005) J Am Chem Soc 127: 8272.
- I T Suydam, S A Strobel (2009) Methods Enzymol 468: 3.
- M C Erat, R K O Sigel (2011) Met Ions Life Sci 9: 37.
- E L Christian, M Yarus (1993) Biochemistry 32: 4475.
- S Basu, S A Strobel (2001) Method 23: 264.
- J A Jansen, T J McCarthy, G A Soukup, J K Soukup (2006) Nature Struct Mol Biol 13: 517.
- V D Sood, T L Beattie, R A Collins (1998) J Mol Biol 282: 741.
- C P Da Costa, D Krajewska, A Okruszek, W J Stec, H Sigel (2002) J Biol Inorg Chem 7: 405.
- C P Da Costa, A Okruszek, H Sigel (2003) Chem Bio Chem 4: 593.
- J K Frederiksen, R Fong, J A Piccirilli (2009) “In Nucleic Acid-Metal Ion Interactions”, Ed N V Hud, Royal Society of Chemistry, Cambridge, UK.
- J L Leroy, M Guéron (1977) Biopolymers 16: 2429.
- T E Horton, D R Clardy, V J DeRose (1998) Biochemistry 37: 18094.
- N Kisseleva, S Kraut, A Jaschke, O Schiemann (2007) HFSP J 1: 127.
- L Hunsicker-Wang, M Vogt, V J DeRose (2009) Methods Enzymol 468: 335.
- V J DeRose (2009) “In Nucleic Acid-Metal Ion Interactions”, Ed N V Hud, Royal Society of Chemistry, Cambridge UK.
- M Vogt, S Lahiri, C G Hoogstraten, R D Britt, V J DeRose (2006) J Am Chem Soc 128: 16764.
- J E Penner-Hahn (2005) Coord Chem Rev 249: 161.
- I V Smirnov, F W Kotch, I J Pickering, J T Davis, R H Shafer (2002) Biochemistry 41: 12133.
- K S Koutmou, A Casiano-Negroni, M M Getz, S Pazicni, A J,rews, et al. (2010) Proc Natl Acad Sci 107: 2479.
- -K Teo, P Eisenberger, J Reed, J K Barton, S J Lippard (1978) J Am Chem Soc 100: 3225.
- A P Hitchcock, C J L Lock, W M C Pratt (1982) Inorg Chim Acta 66: 45.
- J M Benevides, S A Overman, G J Thomas (2005) J Raman Spec 36: 279.
- T Theophanides (2000) “In Infrared, Raman Spectroscopy of Biological Materials”, Ed H.-U.Gremlich, B Yan, M Dekker, New York.
- Y Chen, N V Eldho, K T Dayie, P R Carey (2010) Biochemistry 49: 3427.
- H Takeuchi, H Murata, I Harada (1988) J Am Chem Soc 110: 392.
- H A Tajmir-Riahi, T Theophanides (1985) J Biomol Struct Dyn 2, 995 (1985) ; J Biomol Struct Dyn 3: 537.
- S Adam, P Bourtayre, J Liquier, E Taillandier (1986) Nucleic Acids Res 14: 3501.
- M Langlais, H A Tajmir-Riahi, R Savoie (1990) Biopolymers 30: 743.
- J Duguid, V A Bloomfield, J Benevides, G J Thomas (1995) Biophys J 65, 1916 (1993); Biophys J 69: 2623.
- H Arakawa, J F Neault, H A Tajmir-Riahi (2001) Biophys J 81: 1580.
- J Stangret, R Savoie (2002) Phys Chem Chem Phys 4: 4770.
- S Nafisi, Z Norouzi (2009) DNA Cell Biol 28: 469.
- S Nafisi, A Sobhanmanesh, K Alimoghaddam, A Ghavamzadeh, H A Tajmir-Riahi (2005) DNA Cell Biol 24: 634.
- E L Christian, V E,erson, P R Carey, M E Harris (2010) Biochemistry 49: 2869.
- H Chen, B Gong, P C Bevilacqua, P R Carey, B L Golden (2009) Biochemistry 48: 1498.
- B Gong, J H Chen, E Chase, D M Chadalavada, R Yajima, et al. (2007) J Am Chem Soc 129: 13335.
- B Gong, Y Chen, E L Christian, J H Chen, E Chase, et al. (2008) J Am Chem Soc 130: 9670.
- A Wong, R Ida, G Wu (2005) Biochem Biophys Res Commun 337: 363.
- M L Gill, S A Strobel, J P Loria (2005) J Am Chem Soc 127” 16723.
- R L Gonzalez Jr, I Tinoco Jr (2002) J Mol Biol 289, 1267 (1999); Methods Enzymol 338: 421.
- J Feigon, S E Butcher, L D Finger, N V Hud (2001) Methods Enzymol 338: 400.
- R K O Sigel, D G Sashital, D L Abramovitz, A G Palmer, S E Butcher, A M Pyle (2004) Nat Struct Mol Biol 11: 187.
- M C Erat, O Zerbe, T Fox, R K O Sigel (2007) Chem Bio Chem 8: 306.
- S E Butcher, F H T Allain, J Feigon (2000) Biochemistry 39: 2174.
- S S Wijmenga, B N M van Buuren (1998) Prog Nucl Mag Res Sp 32: 287.
- B A Connolly, F Eckstein (1984) Biochemistry 23: 5523.
- M Maderia, L M Hunsicker, V J DeRose (2000) Biochemistry 39: 12113.
- E M Osborne, W L Ward, M Z Ruehle, V J DeRose (2009) Biochemistry 48: 10654.
- Y Tanaka, C Kojima, E H Morita, Y Kasai, K Yamasaki, A Ono, M Kainosho, K Taira (2002) J Am Chem Soc 124: 4595.
- G Wang, B L Gaffney, R A Jones (2004) J Am Chem Soc 126: 8908.
- G W Buchanan, J B Stothers (1982) Can J Chem 60: 787.
- I Bertini, C Luchinat (1986) “NMR of Paramagnetic Molecules in Biological Systems “,Benjamin/Cummings Pub Co Menlo Park, CA (1986).
- I Bertini, C Luchinat, G Parigi(2001) “Solution NMR of Paramagnetic Molecules – Applications to Metallobiomolecules, Models”, Elsevier Science B.V Amsterdam.
- J H Davis, T R Foster, M Tonelli, S E Butcher (2007) RNA 13: 76.
- J Noeske, H Schwalbe, J Wöhnert (2007) Nucleic Acids Res 35: 5262.
- D O Campbell, P Bouchard, G Desjardins, P Legault (2006) Biochemistry 45: 10591.
- R E Hurd, E Azhderian, B R Reid (1979) Biochemistry 18: 4012.
- J A Cowan (1993) J Inorg Biochem 49: 171.
- M Schmitz, I Tinoco (2000) Jr RNA 6: 1212.
- G Colmenarejo, I Tinoco Jr (1999) J Mol Biol 290: 119.
- J S Kieft, I Tinoco Jr Structure 5: 713.
- S Rüdisser, I Tinoco Jr (2000) J Mol Biol 295: 1211.
- P Schultze, N V Hud, F W Smith, J Feigon (1999) Nucleic Acids Res 27: 3018.
- F W Smith, J Feigon (1993) Biochemistry 32: 8682.
- N V Hud, P Schultze, V Sklenár, J Feigon (1999) J Mol Biol 285: 233.
- D M Rose, C F Polnaszek, R G Bryant (1982) Biopolymers, 21: 653.
- S S Reid, J A Cowan (1990) Biochemistry, 29, 6025.
- E Berggren, L Nordenskiöld, W H Braunlin (1992) Biopolymers 32: 1339.
- J A Cowan, H W Huang, L Y Hsu (1993) J Inorg Biochem 52: 121.
- L A Wright, L E Lerner (1994) Biopolymers 34: 691.
- L M Utschig, J W Bryson (1995) T V O'Halloran, Science 268: 380.
- J E Coleman (1993) Methods Enzymol 227: 16.
- R Ida, G Wu (2008) J Am Chem Soc 130: 3590.
- J P,ré, H R Mäcke (2003) J Inorg Biochem 97: 315.
- Y Tanaka, Y Kasai, S Mochizuki, A Wakisaka, E H Morita, et al. (2004) J Am Chem Soc 126: 744.
- J Reedijk (2008) Platinum Met Rev 52: 2.
- L Helm, A E Merbach (1999) Coord Chem Rev 187: 151.
- V K Misra, D E Draper (2002) J Mol Biol 317: 507.
- D E Draper (2004) RNA 10: 335.
- M J Serra, J D Baird, T Dale, B L Fey, K Retatagos, E Westhof (2002) RNA 8: 307.
- Y V Bukhman, D E Draper (1997) J Mol Biol 273: 1020.
- R Shiman, D E Draper (2000) J Mol Biol 302: 79.
- L G Laing, T C Gluick, D E Draper (1994) J Mol Biol 237: 577.
- C Hammann, A Cooper, D M J Lilley (2001) Biochemistry 40: 1423.
- S O Shan, A Yoshida, S Sun, J A Piccirilli, D Herschlag (1999) Proc Natl Acad Sci 96: 12299.
- S Wang, K Karbstein, A Peracchi, L Beigelman, D Herschlag (1999) Biochemistry 38: 14363.
- P M Gordon, E J Sontheimer, J A Piccirilli (2000) Biochemistry 39: 12939.
- S I Nakano, D J Proctor, P C Bevilacqua (2001) Biochemistry 40: 12022.
- A K Brown, J Li, C M B Pavot, Y Lu (2003) Biochemistry 42: 7152.
- J M Wilcox, D L Rempel, M L Gross (2008) Anal Chem 80: 236.
- D Grilley, A M Soto, D E Draper (2009) Methods Enzymol 455: 71.
- H Krakauer (1971) Biopolymers 10: 2459.
- R Römer, R Hach (1975) Eur J Biochem 55: 271.
- Y Bai, M Greenfeld, K J Travers, V B Chu, J Lipfert, et al. (2007) J Am Chem Soc 129: 14981.
- M Greenfeld, D Herschlag (2009) Methods Enzymol 469: 375.
- A Polak, H J Scholer (1975) Chemotherapy 21: 113.
- A Cox (1968) Quart Rev 22, 934.
- O N Al Safarjalani, X J Zhou, R H Ras, J Shi, R F Schinazi, et al. (2005) Cancer Chemother Pharmacol 55: 541.
- G B Elion (1967) Fed Proc 26: 898.
- J H Burchenal (1953) Blood 8: 965.
- B D Clarkson (1970) Cancer 5: 227.
- S A Giller, R A Zhuk, M I U Lidak (1967) Dokl Akad Nauk SSR 176: 332.
- J L Ambrus, I Stadler, M Kulaylat, A Koreshi, S Akhtar (1996) J Med Chem 27: 21.
- M Weller, B Muller, R Koch, M Bamberg, P Krauseneck (2003) J Clin Oncol 21: 3276.
- M Horton (2005) Clin Cancer Res 11: 1884.
- B J Kennedy, J L Torkelson, E Torlakovic (1999) Cancer 85: 2265.
- J R Bertino (1979) Biochem Pharmacol 28: 1983.
- L W Hertel, G B Border, J S Kroin, S M Rinzel, G A Poore, et al. (1990) Cancer Res 50: 4417.
- A Agrawal, K Srivastava, S K Puri (2005) Bio Org Med Chem Lett 15: 5218.
- T C Daniels, E C Jorgensen (1982) “Central nervous system depressents in Wilson, Gisvoids Textbook of Organic medicinal, Pharmaceutical Chemistry”,ed R F Doerge, , J B Lippen-Cott, Philadelphia 33.
- D S Threlkeld (1998) Facts, Comparisons: 269.
- J Vida, J Yevich (2003) “Sedative hypnotics, In Burger's Medicinal Chemistry, Drug Discover” , 6thed D J Abrahim, John Wiley, New Jersy (6): 203.
- C J Shishoo, U S Pathak, K S Jain, I T Devani, M T Chhabria (1994) Chem Inform 25(38): 436.
- M S Masoud, M Sh Ramadan, A El-Samahy, I Mahmoud, M H Al-Saify (2017) Advanced Materials Science 2(4): 1.
- M S Masoud, M Sh Ramadan, M H Al-Saify (2018) Pharm Anal Chem 4(1): 1.
- M S Masoud, M Sh Ramadan, A H El-Sayed, A M Sweyllam, M H Al-Saify (2018) Arc Org Inorg Chem Sci 3(5): 410.
- M S Masoud, M F Fathalla, R H A Mohamed, M S Hassan (2019) Asian J of Chem 31(3): 733.
- M S Masoud, A S El-Kholany, M F El-Shahat (2019) J of Chemical, Pharmaceutical Research 11(12):
-
MS Masoud , M Sh Ramadan, A M Sweyllam, M H Al-Saify. Synthesis, Characterization and Applications of Some Transition Metal Complexes of Nucleic Acid Constituents. Insi in Chem & Biochem. 1(4): 2021. ICBC. MS.ID.000516.
-
Antitumor Activity, Novel Azole compound, Carcinoma, Swiss Albino Mice.
-

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.
- Introduction
- Metal-Nucleobase Interactions
- Metal-Phosphate Interactions
- Metal-Nucleotide Interactions
- Interaction of Metal Cations with Base Pairs
- Nucleic Acids and Metallodrugs
- Platinum Coordination Compounds
- Studies on Cisplatin Binding to Nucleobases
- Non-Platinum Anticancer Agents
- Current and Future Developments
- Site-Specific Binding of Cations to Nucleic Acids
- Metallated Nucleic Acids for Nanotechnology
- Structural Characterization of Metal Ion Binding Sites
- Determination of Binding Kinetics and Affinities
- Biological Importance of Pyrimidine
- Acknowledgement
- Conflict of Interest
- References






