 Research Article
Research Article
‘’ZINC ‘’ The Friendly and Forgotten Micronutrient
Khajik Sirob Yaqob*
Specialist in child’s nutrition and growth with interest in pediatrics neurology, Iraq
Khajik Sirob Yaqob, Specialist in child’s nutrition and growth with interest in pediatrics neurology,0 Associate membership and fellowship of the RCPCH, Membership of Oxford university hospitals, Iraq.
Received Date: August 02, 2019; Published Date: August 26, 2019
Introduction
Zinc is the second most abundant trace mineral after iron, and is important in the protein metabolism and synthesis, in DNA metabolism, and in stabilization of cell membrane [1].
It is essential for functions of various cellular metabolisms, and it acts as a cofactor for more than two hundred enzymes [1]. Also, zinc has its crucial role during periods of growth and tissue proliferation (immune system, wound healing, skin, and GIT integrity). Furthermore, zinc has its own vital physiologic role in immune function, sexual maturation and normal growth [1].
Animal products including human milk are considered the best dietary source of zinc. In addition, whole grains and legumes contain moderate amount of zinc. Globally, poor bioavailability of zinc secondary to phytic acid, which inhibits zinc absorption from its sources, is considered more important factor than low intake in many cases of zinc deficiency [1].
A syndrome called Zinc Deficiency Dwarfism was first described in a group of children in the Middle East with low level of zinc in their hair, poor appetite, diminished taste acuity, hypogonadism, and short stature [1]. In developing countries, zinc supplementation can reduce morbidity and mortality among children from diarrhea and pneumonia, and zinc can enhance growth [1]. Prevalence of zinc deficiency is highly noticed in populations with high rates of stunting. Other contributing factors that can lead to zinc deficiency include inadequate zinc intake in complementary feeding or general diet, sever infection, lack of zinc in total parenteral nutrition and in premature infants fed human milk without fortification [1].
Clinical Manifestation of Zinc Insufficiency
In mild cases, poor appetite, growth faltering and immune impairment. Moderate-severe symptoms and signs include, delayed sexual maturation, rough skin and enlarged liver and spleen; whereas, manifestation as mood changes, growth and immune impairment, diarrhea, alopecia, photophobia, night blindness, scaling dermatitis, and acral and periorificial erythematous appear in severe zinc deficiency [1].
Diagnosis of zinc deficiency Is challenging and mainly clinical. In mild zinc deficiency, plasma zinc level is often normal although zinc level is frequently used; levels in moderate to severe deficiency are characteristically less than 60μg/dl. Nevertheless, the response to a trial of zinc supplementation with outcomes such as improved linear growth or weight gain, enhanced appetite, and improved immune function, is the principles for identification of zinc insufficiency [1]. As zinc has no pharmacologic effect on these functions, a positive response to supplementation is considered evidence of preexisting deficiency. Therefore, clinically an empirical trial of zinc supplementation (1μg/kg/day) is a safe and a sensible approach in situations in which zinc deficiency is considered probable.
A specific condition called acrodermatitis enteropathica occurs when there is sever zinc deficiency in which plasma zinc level is markedly reduced and serum alkaline phosphatase activity is low [1]. Clinically, this condition is characterized by acute oral and perianal dermatitis, alopecia, and failure to thrive as a result of lack of intestinal zinc absorption [1]. This is occurring 2-4 weeks after infants have been weaned from breast milk.
Such condition is an autosomal recessive disorder, and it is relatively uncommon due to lack in the secretion of zinc from the mammary gland resulting in abnormally low milk zinc concentration [1]. Hence, this occurs in breast fed infants especially premature present with standard signs of zinc deficiency including growth failure, diarrhea, and dermatitis. Treatment is with high dose of zinc with successful continuous breast feeding.
Zinc is nontoxic element, certain features such as nausea, vomiting, abdominal pain, headache, vertigo and seizures might appear as a result of excessive intake of zinc [1].
Moreover, the main dietary sources of zinc are meat, shellfish, wholegrain, legumes and cheese. In developing countries, due to malnutrition, zinc deficiency is quite common [1].
Zinc dwarfism, hypogonadism, dermatitis and T-cell immunodeficiency are signs of chronic zinc deficiency which often associated with lack of iron micronutrient [1].
People with certain conditions such as Crohn’s disease, short bowel syndrome, and sprue are more prone to zinc deficiency as a result of malabsorption of zinc and increased zinc losses in the urine [2,3]. In contrast, persons with mild human zinc deficiency states, the obvious features and laboratory/functional abnormalities of mild zinc deficiency are varied. Such variety is not altogether astonishing in view of the biochemistry of zinc and the ubiquity of this metal in biology with its contribution in an extraordinarily wide range of essential metabolic processes. Impaired growth velocity is a main clinical feature of mild zinc insufficiency and can be corrected with zinc supplementation [4,5]. Pregnancy outcome [6] and the function of the immune system [7] are evident cases that clinically respond to zinc supplementation.
Zinc is widely used in people with common cold. However, the clinical consequence of zinc lozenges in dropping the duration of flue is still unclear [8].
In patients fed intravenously with no addition of enough zinc to the infusates [9] and in cases of the autosomal recessively inherited disease such as acrodermatitis enteropathica, sever zinc deficiency is documented [10]. Because of association of zinc micronutrient in so many core areas of metabolism, the features of zinc deficiency are basic and nonspecific, including growth retardation, alopecia, diarrhea, delayed sexual maturation and impotence, eye and skin lesions, and impaired appetite. Therefore, clinical features and laboratory criteria are not always reliable. As a result, certain potentially useful laboratory indicators such as alkaline phosphatase activity can be used clinically to validate reliable, sensitive clinical or functional indicators of zinc status in the body.
Zinc and Neurodevelopment
In addition to studies on the clinical impacts of low-dose zinc supplements on individuals with pneumonia, diarrhea [11] and on nonspecific laboratory functional tests of zinc status such as tests of neuro- cognitive function [12] or immune status [13], research results including the effects of zinc supplementation on physical growth velocity in children are helpful to assess the dietary zinc intake for many causes. 1st, approved clinical evidence of zinc supplements on growth velocity (linear growth and weight) in children with variable degrees of growth delay has been documented [14,15]. 2nd, because an adequate figure of these studies has been done in North America, growth is appropriate as a functional/clinical indicator of zinc requirement in North American children [5,16-18]. Finally, database involved in these studies are adequate to use for cohort analysis.
Average Zinc Consumption from Human Milk During the First 12 Months of Feeding
In order to match the zinc intake of the infant in early weeks (Figure) the adequate intake is set at 2.0 mg/day (2.5mg/L × 0.78L/ day). Interestingly, similar clinical results have been obtained concerning weight gain and body length or height at ages foursix months when evaluated by zinc intake from human milk and complementary foods at this age [19]. Consequently, a helpful connection between zinc content of human milk at five months and improvement in the weight-for age Z scores for the five to seven months interval have been documented [20]. However, that growthlimiting zinc deficiency can occur in infants mainly fed human milk after the age of four months [21].
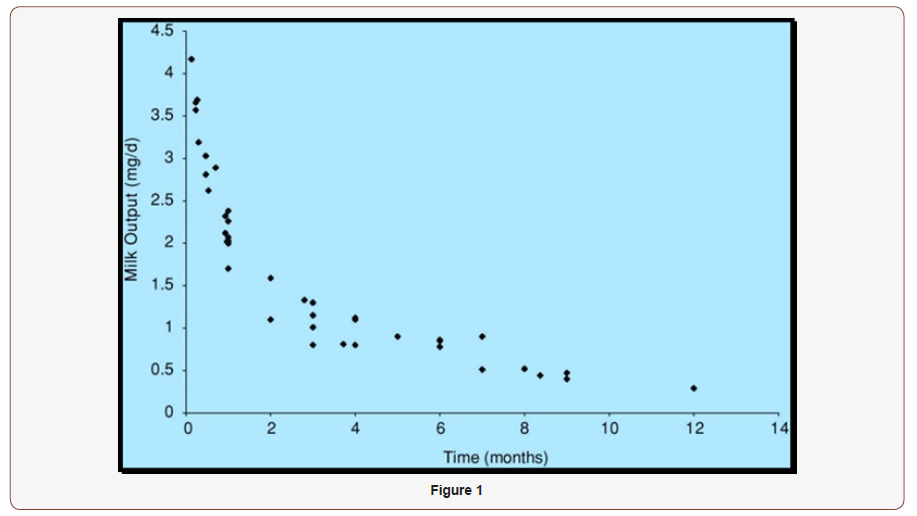
Factorial estimates of requirements (i.e., 2.1mg/day at one month and 1.54mg/day at five months) are consistent with this adequate intake for infants from birth to six months. Such evaluations are based on amount of zinc ingestion of infants fed human milk, fractional absorption, and endogenous losses [22]. Skin and urine losses are from available estimations [23]. In addition an earlier report that physical growth of male infants fed a zinc-fortified cow milk formula (5.8mg/L) was superior to that of infants receiving the same formula but with a zinc concentration of 1.8mg/L, which provided about 1.4mg/day of zinc [17].
Zinc intake from Human Milk. Zinc nourishment in afterward infancy varies quietly from that in the younger infant. It is likely that neonatal hepatic stores, which may contribute to metabolically usable zinc pools in early postnatal life, have been degenerated [24]. Human milk provides only 0.5mg/day of zinc by seven months postpartum [120], and the concentration drops even further by twelve months [25]. Consequently, human milk alone is an insufficient source of zinc after the first six months. Hence, extrapolation from human milk intake during the 1st six months after birth, which yields 2.4mg/day, does not reflect adequate zinc intake during the 2nd six months.
Zinc consumption from Human Milk and Complementary Foods. According to the Third National Health and Nutrition Examination Survey, the median intake of zinc from complementary foods is 1.48mg/day (n = 45) for older infants consuming human milk. So, the average zinc intake from human milk and complementary foods is estimated to be approximately 2mg/day (0.5 + 1.48).
Requirements of zinc for Growth. These requirements have been estimated from chemical analyses of infants and adults, which give an average concentration of 20μg/g wet weight of zinc [26]. It is assumed that each gram of new lean and adipose tissue requires this amount of zinc. The average amount of new tissue accreted for older infants and young children is thirteen and 6g/ day, correspondingly [27].
Therefore, the total amount of absorbed zinc required for infants ages seven to twelve months is 836μg/day (Table 1), while for children ages one to four years is 744μg/day (Table 2).
GIT (Gastrointestinal tract) losses vary directly with the amount of zinc absorbed (see “Adults Ages 19 Years and Older”). The average gut excretion of endogenous zinc in infants aged two-four months who receive human milk is approximately 50μg/kg/day [22]. This is risky in adults as the amount of absorbed zinc is equal to the total endogenous zinc losses. This critical level, derived from all available sets of data for adult men, yields an average emission of 34μg/kg/ day of zinc and is used for children beyond one year of age and adolescents. Consequently, 50μg/kg/day is used for older infants and 34μg/ kg/day for children aged one to three years.
Urinary system losses of zinc are concerning 7.5μg/kg/day for both sexes (see “Adults Ages 19 Years and Older”). After early infancy, excretion rates for children on a body weight basis seem to be different from adult values [23]. Yet, no evident database is approved on the skin losses in children, so estimates for children are resulting from statistics in adult men [28], which give an estimation of 14μg/kg/day of zinc. As a result, the estimated figure endogenous excretion of zinc is 64μg/kg/ day for older infants and 48μg/kg/day for children aged one to three years (Table 1 &2).
Table 1:Zinc consumption and need for infants aged seven-twelve months.
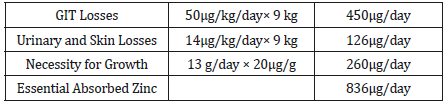
Table 2:Requirement for absorbed Zinc for children aged one-three years.

Children Ages 4-8 Years
For this age group, the average intestinal losses are 34μg/ kg/day of zinc and the amount of new tissue accreted is 7g/day [27]. Based on the summation of zinc losses and requirements for growth, the required amount of absorbed zinc for this age group is about 1.2mg/day (Table 3). With a fractional absorption of 0.3 based on studies in infants and young children [29,30], the EAR is 4.0mg/day of zinc (Table 3).
Table 3:Necessity for absorbed Zinc for kids aged 4-8 years.

Growth. Some dietary data are presented from children aged four-eight years whose growth percentiles were at the lower end of the normal range and who were subjects in placebo-controlled, randomized trials of dietary zinc supplementation. In each of two studies, one in Canada [31-62] and the other in the United States [63-98], zinc supplementation was associated with greater linear growth gain. Mean dietary intakes of the placebo-treated controls in the Canadian and U.S. studies were 6.4 and 4.6 mg/day of zinc, respectively. No growth response was observed with zinc supplementation of healthy children of either gender, unselected for growth, whose average calculated zinc intake was 6.3mg/day [99- 156]. The SDs were too large (likely attributable to methodological limitations) to use these data with any confidence in setting an EAR. Nevertheless, these data are consistent with the EAR derived from a factorial approach.
Children Ages 9-13 Years
With use of the same values as for younger children, an average accretion of 10g/day of new tissue [157-187], and a reference weight of 40 kg, the required amount of absorbed zinc is 2.1mg/ day (Table 4). Based on a fractional absorption of 0.3 observed in infants and young children [188-227], the EAR is 7mg/day (Table 4&5).
Table 4:Requirement for absorbed Zinc for children aged 9-13 years.
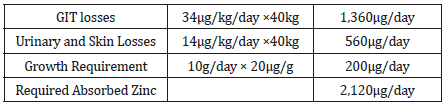
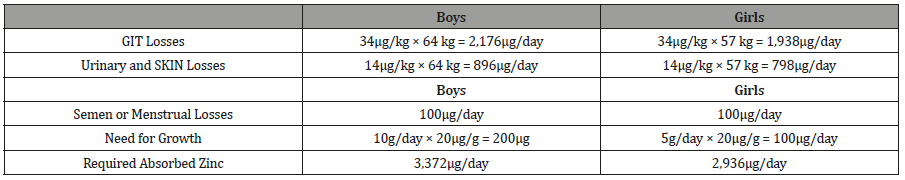
Table 5:Requirement for absorbed Zinc for adolescent boys and girls aged 14 through 18 years.
Acknowledgement
None.
Conflict of Interest
No conflicts of interest
References
- Abrams SA, Wen J, Stuff JE (1997) Absorption of calcium, zinc, and iron from breast milk by five- to seven-month-old infants. Pediatr Res 41(3): 384-390.
- Pironi L, Miglioli M, Cornia GL, Ursitti MA, Tolomelli M, et al. (1987) Urinary zinc excretion in Crohn's disease. Dig Dis Sci 32(4): 358-362.
- Valberg LS, Flanagan PR, Kertesz A, Bondy DC (1986) Zinc absorption in inflammatory bowel disease. Dig Dis Sci 31(7): 724-731.
- Hambidge KM, Chavez MN, Brown RM, Walravens PA (1979) a. Zinc nutritional status of young middle-income children and effects of consuming zinc-fortified breakfast cereals. Am J Clin Nutr 32(12): 2532-2539.
- Walravens PA, Hambidge KM, Koepfer DM (1989) Zinc supplementation in infants with a nutritional pattern of failure to thrive: A double-blind, controlled study. Pediatrics 83(4): 532-538.
- Goldenberg RL, Tamura T, Neggers Y, Copper RL, Johnston KE, et al. (1995) The effect of zinc supplementation on preganancy outcome. J Am Med Assoc 274(6): 463-468.
- Bogden JD, Oleske JM, Munves EM, Lavenhar MA, Bruening KS, et al. (1987) Zinc and immunocompetence in the elderly: Baseline data on zinc nutriture and immunity in supplemented subjects. Am J Clin Nutr 46(1): 101-109.
- Jackson JL, Lesho E, Peterson C (2000) Zinc and the common cold: A meta-analysis revisted. J Nutr 130(5): 1512S-1515S.
- Chen W, Chiang TP, Chen TC (1991) Serum zinc and copper during long-term total parenteral nutrition. J Formos Med Assoc 90(11): 1075-1080.
- Walling A, Householder M, Walling A (1989) Acrodermatitis enteropathica. Am Fam Physician 39(2): 151-154.
- Bhutta ZA, Nizami SQ, Isani Z (1999) Zinc supplementation in malnourished children with persistent diarrhea in Pakistan. Pediatrics 103(4): e42.
- Sandstead HH, Penland JG, Alcock NW, Dayal HH, Chen XC, et al. (1998) Effects of repletion with zinc and other micronutrients on neuropsychologic performance and growth of Chinese children. Am J Clin Nutr 68 (2 Suppl) : 470S-475S.
- Shankar AH, Prasad AS (1998) Zinc and immune function: The biological basis of altered resistance to infection. Am J Clin Nutr 68(2 Suppl): 447S-463S.
- Brown KH, Peerson JM, Allen LH (1998) Effect of zinc supplementation on children's growth: A meta-analysis of intervention trials. Bibl Nutr Dieta 54: 76-83
- Umeta M, West CE, Haidar J, Deurenberg P, Hautvast JGAJ (2000) Zinc supplementation and stunted infants in Ethiopia: A randomised controlled trial. Lancet 355(9220): 2021-2026.
- Gibson RS, Vanderkooy PD, MacDonald AC, Goldman A, Ryan BA, et al. (1989) A growth-limiting, mild zinc-deficiency syndrome in some southern Ontario boys with low height percentiles. Am J Clin Nutr 49(6): 1266-1273.
- Walravens PA, Hambidge KM (1976) Growth of infants fed a zinc supplemented formula. Am J Clin Nutr 29(10): 1114-1121.
- Walravens PA, Krebs NF, Hambidge KM (1983) Linear growth of low-income preschool children receiving a zinc supplement. Am J Clin Nutr 38(2): 195-201.
- Dewey KG, Cohen RJ, Brown KH, Rivera LL (1999) Age of introduction of complementary foods and growth of term, low-birth-weight, breast-fed infants: A randomized intervention study in Honduras. Am J Clin Nutr 69(4): 679-686.
- Krebs NF, Reidinger CJ, Robertson AD, Hambidge KM (1994) Growth and intakes of energy and zinc in infants fed human milk. J Pediatr 124(1): 32-39.
- Walravens PA, Chakar A, Mokni R, Denise J, Lemonnier D (1992) Zinc supplements in breastfed infants. Lancet 340(8821): 683–685.
- Krebs NF, Reidinger CJ, Miller LV, Hambidge KM (1996) Zinc homeostasis in breast-fed infants. Pediatr Res 39(4 Pt 1): 661-665.
- Krebs NF, Hambidge KM (1986) Zinc requirements and zinc intakes of breast-fed infants. Am J Clin Nutr 43(2): 288-292.
- Zlotkin SH, Cherian MG (1988) Hepatic metallothionein as a source of zinc and cysteine during the first year of life. Pediatr Res 24(3): 326-329.
- Casey CE, Neville MC, Hambidge KM (1989) Studies in human lactation: Secretion of zinc, copper, and manganese in human milk. Am J Clin Nutr 49(5): 773-785.
- Widdowson EM, Dickerson JWT (1964) Chemical composition of the body. In: Comar CL, editor, Bronner F, editor, eds. Mineral Metabolism. An Advanced Treatise, Vol II, The Elements, Part A, New York, Academic Press, pp. 1-247.
- Kuczmarski RJ, Ogden CL, Grummer-Strawn LM, Flegal KM, Guo SS, et al. (2000) CDC Growth Charts: United States. Adv Data 8(314): 1-27.
- Lee DY, Prasad AS, Hydrick-Adair C, Brewer G, Johnson PE (1993) Homeostasis of zinc in marginal human zinc deficiency: Role of absorption and endogenous excretion of zinc. J Lab Clin Med 122(5): 549-556.
- Davidsson L, Mackenzie J, Kastenmayer P, Aggett PJ, Hurrell RF (1996) Zinc and calcium apparent absorption from an infant cereal: A stable isotope study in healthy infants. Br J Nutr 75(2): 291-300.
- Fairweather Tait SJ, Wharf SG, Fox TE (1995) Zinc absorption in infants fed iron-fortified weaning food. Am J Clin Nutr 62(4): 785-789.
- Aggett PJ (1989) Severe zinc deficiency. In: Mills CF, Zinc in Human Biology. (edn) New York: Springer-Verlag. pp.259–279.
- Alexander D, Ball MJ, Mann J (1994) Nutrient intake and haematological status of vegetarians and age-sex matched omnivores. Eur J Clin Nutr 48(8): 538-546.
- Alexander FW, Clayton BE, Delves HT (1974) Mineral and trace-metal balances in children receiving normal and synthetic diets. Quart J Med 43(169): 89-111.
- Allen JC, Keller RP, Archer P, Neville MC (1991) Studies in human lactation: Milk composition and daily secretion rates of macronutrients in the first year of lactation. Am J Clin Nutr 54(1): 69-80.
- Anderson BM, Gibson RS, Sabry JH (1981) The iron and zinc status of long-term vegetarian women. Am J Clin Nutr 34(6): 1042-1048.
- Anderson RR (1993) Longitudinal changes of trace elements in human milk during the first 5 months of lactation. Nutr Res 13(5): 499-510.
- Aquilio E, Spagnoli R, Seri S, Bottone G, Spennati G (1996) Trace element content in human milk during lactation of preterm newborns. Biol Trace Elem Res 51(1):63-70.
- Artacho R, Ruiz-Lopez MD, Gamez C, Puerta A, Lopez MC (1997) Serum concentration and dietary intake of Zn in healthy institutionalized elderly subjects. Sci Total Environment 205(2-3): 159-165.
- August D, Janghorbani M, Young VR (1989) Determination of zinc and copper absorption at three dietary Zn-Cu ratios by using stable isotope methods in young adult and elderly subjects. Am J Clin Nutr 50(6):1457-1463.
- Baer MT, King JC (1984) Tissue zinc levels and zinc excretion during experimental zinc depletion in young men. Am J Clin Nutr 39(4): 556-570.
- Bales CW, Di Silvestro RA, Currie KL, Plaisted CS, Joung H, et al. (1994) Marginal zinc deficiency in older adults: Responsiveness of zinc status indicators. J Am Coll Nutr 13(5): 455-462.
- Beck FW, Kaplan J, Fine N, Handschu W, Prasad AS (1997) Decreased expression of CD73 (ecto-5′-nucleotidase) in the CD8+ subset is associated with zinc deficiency in human patients. J Lab Clin Med 130(2): 147-156.
- Beck FW, Prasad AS, Kaplan J, Fitzgerald JT, Brewer GJ (1997) Changes in cytokine production and T cell subpopulations in experimentally induced zinc-deficient humans. Am J Physiol 272(6 Pt 1): E1002-E1007.
- Behall KM, Scholfield DJ, Lee K, Powell AS, Moser PB (1987) Mineral balance in adult men: Effect of four refined fibers. Am J Clin Nutr 46(2):307-314.
- Berg JM, Shi Y (1996) The galvanization of biology: A growing appreciation for the roles of zinc. Science 271(5252):1081-1085.
- Berglund M, Akesson A, Nermell B, Vahter M (1994) Intestinal absorption of dietary cadmium in women depends on body iron stores and fiber intake. Environ Health Perspect 102(12): 1058-1066.
- Biego GH, Joyeux M, Hartemann P, Debry G (1998) Determination of mineral contents in different kinds of milk and estimation of dietary intake in infants. Food Addit Contam 15(7): 775-781.
- Black MR, Medeiros DM, Brunett E, Welke R (1988) Zinc supplements and serum lipids in young adult white males. Am J Clin Nutr 47(6): 970-975.
- Botash AS, Nasca J, Dubowy R, Weinberger HL, Oliphant M (1992) Zinc-induced copper deficiency in an infant. Am J Dis Child 146(6): 709-711.
- Boukaiba N, Flament C, Acher S, Chappuis P, Piau A, et al. (1993) A physiological amount of zinc supplementation: Effects on nutritional, lipid, and thymic status in an elderly population. Am J Clin Nutr 57(4): 566-572.
- Brants HA, Lowik MR, Westenbrink S, Hulshof KF, Kistemaker C (1990) Adequacy of a vegetarian diet at old age (Dutch Nutrition Surveillance System). J Am Coll Nutr 9(4): 292-302.
- Brewer GJ, Yuzbasiyan-Gurkan V, Johnson V, Dick RD, Wang Y (1993) Treatment of Wilson's Disease with zinc XII: Dose regimen requirements. Am J Med Sci 305(4): 199-202.
- Bunker VW, Lawson MS, Delves HT, Clayton BE (1982) Metabolic balance studies for zinc and nitrogen in healthy elderly subjects. Hum Nutr Clin Nutr 36(3): 213-221.
- Burke DM, DeMicco FJ, Taper LJ, Ritchey SJ (1981) Copper and zinc utilization in elderly adults. J Gerontol 36(5): 558-563.
- Butte NF, Garza C, Smith EO, Nichols BL (1984) Human milk intake and growth in exclusively breast-fed infants. J Pediatr 104(2): 187-195.
- Cakman I, Kirchner H, Rink L (1997) Zinc supplementation reconstitutes the production of interferon-α by leukocytes from elderly persons. J Interferon Cytokine Res 17(8): 469-472.
- Casey CE, Hambidge KM, Neville MC (1985) Studies in human lactation: Zinc, copper, manganese and chromium in human milk in the first month of lactation. Am J Clin Nutr 41(6): 1193-1200.
- Caulfield LE, Zavaleta N, Figueroa A (1999) Adding zinc to prenatal iron and folate supplements improves maternal and neonatal zinc status in a Peruvian population. Am J Clin Nutr 69(6): 1257-1263.
- Caulfield LE, Zavaleta N, Figueroa A, Leon Z (1999) Maternal zinc supplementation does not affect size at birth or pregnancy duration in Peru. J Nutr 129(8): 1563-1568.
- Chandra RK (1984) Excessive intake of zinc impairs immune responses. J Am Med Assoc 252(11): 1443-1446.
- Cheek DB, Reba RC, Woodward K (1968) Cell growth and the possible role of trace minerals. In: Cheek DB, editor, ed. Human Growth; Body Composition, Cell Growth, Energy, and Intelligence. Philadelphia: Lea and Febiger. pp.424-439.
- Chesters JK (1997) Zinc. In: O'Dell BL, editor, Sunde RA, editor, eds. Handbook of Nutritionally Essential Mineral Elements. New York: Marcel Dekker. pp.185-230.
- Cole TB, Wenzel HJ, Kafer KE, Schwartzkroin PA, Palmiter RD (1999) Elimination of zinc from synaptic vesicles in the intact mouse brain by disruption of the ZnT3 gene. Proc Natl Acad Sci USA 96(4): 1716-1721.
- Colin MA, Taper LJ, Ritchey SJ (1983) Effect of dietary zinc and protein levels on the utilization of zinc and copper by adult females. J Nutr 113(8): 1480-1488.
- Coudray C, Bellanger J, Castiglia-Delavaud C, Remesy C, Vermorel M, et al. (1997) Effect of soluble or partly soluble dietary fibres supplementation on absorption and balance of calcium, magnesium, iron and zinc in healthy young men. Eur J Clin Nutr 51(6): 375-380.
- Cousins RJ (1985) Absorption, transport, and hepatic metabolism of copper and zinc: Special reference to metallothionein and ceruloplasmin. Physiol Rev 65(2): 238-309.
- Cousins RJ (1989) a. Systemic transport of zinc. In: Mills CF, editor, ed. Zinc in Human Biology. New York: Springer-Verlag. pp.79-93.
- Cousins RJ (1989) b. Theoretical and practical aspects of zinc uptake and absorption. Adv Exp Med Biol 249: 3-12.
- Cousins RJ (1994) Metal elements and gene expression. Ann Rev Nutr 14: 449-469.
- Cousins RJ (1996) Zinc. In: Filer LJ, editor, Ziegler EE, editor, eds. Present Knowledge in Nutrition, 7th ed. Washington, DC: International Life Science Institute-Nutrition Foundation. pp.293-306.
- Couzy F, Kastenmayer P, Mansourian R, Guinchard S, Munoz-Box R, et al. (1993) Zinc absorption in healthy elderly humans and the effect of diet. Am J Clin Nutr 58(5): 690-694.
- Dalton TP, Bittel D, Andrews GK (1997) Reversible activation of mouse metal response element-binding transcription factor 1 DNA binding involves zinc interaction with the zinc finger domain. Molec Cell Biol 17(5): 2781-2789.
- Davis CD, Milne DB, Nielsen FH (2000) Changes in dietary zinc and copper affect zinc-status indicators of postmenopausal women, notably extracellular superoxide dismutase and amyloid precursor proteins. Am J Clin Nutr 71(3): 781-788.
- Devine A, Rosen C, Mohan S, Baylink D, Prince RL (1998) Effects of zinc and other nutritional factors on insulin-like growth factor I and insulin-like growth factor binding proteins in postmenopausal women. Am J Clin Nutr 68(1): 200-206.
- Donovan UM, Gibson RS (1995) Iron and zinc status of young women aged 14 to 19 years consuming vegetarian and omnivorous diets. J Am Coll Nutr 14(5): 463-472.
- Donovan UM, Gibson RS (1996) Dietary intakes of adolescent females consuming vegetarian, semi-vegetarian, and omnivorous diets. J Adolesc Health 18(4): 292-300.
- Duchateau J, Delepesse G, Vrijens R, Collet H (1981) Beneficial effects of oral zinc supplementation on the immune response of old people. Am J Med 70(5): 1001-1004.
- Ellis R, Kelsay JL, Reynolds RD, Morris ER, Moser PB, et al. (1987) Phytate:zinc and phytate x calcium:zinc millimolar ratios in self-selected diets of Americans, Asian Indians, and Nepalese. J Am Diet Assoc 87(8): 1043-1047.
- Faber M, Gouws E, Spinnler Benade AJ, Labadarios D (1986) Anthropometric measurements, dietary intake and biochemical data of South African lactoovovegetarians. S Afr Med J 69(12): 733-738.
- Failla ML (1999) Considerations for determining “optimal nutrition” for copper, zinc, manganese and molybdenum. Proc Nutr Soc 58(2): 497-505.
- Ferguson EL, Gibson RS, Opare-Obisaw C, Ounpuu S, Thompson LU, et al. (1993) The zinc nutriture of preschool children living in two African countries. J Nutr 123(9): 1487-1496.
- Festa MD, Anderson HL, Dowdy RP, Ellersieck MR (1985) Effect of zinc intake on copper excretion and retention in men. Am J Clin Nutr 41(2): 285-292.
- Fischer PWF, Giroux A, L Abbe MR (1984) Effect of zinc supplementation on copper status in adult man. Am J Clin Nutr 40(4): 743-746.
- Fortes C, Forastiere F, Agabiti N, Fano V, Pacifici R, et al. (1998) The effect of zinc and vitamin A supplementation on immune response in an older population. J Am Geriatr Soc 46(1): 19-26.
- Fosmire GJ (1990) Zinc toxicity. Am J Clin Nutr 51(2): 225-227.
- Fransson GB, Lonnerdal B (1982) Zinc, copper, calcium, and magnesium in human milk. J Pediatr 101(4): 504-508.
- Freeland-Graves JH, Bodzy PW, Eppright MA (1980) a. Zinc status of vegetarians. J Am Diet Assoc 77(6): 655-661.
- Freeland-Graves JH, Ebangit ML, Hendrikson PJ (1980) b. Alterations in zinc absorption and salivary sediment zinc after a lacto-ovo- vegetarian diet. Am J Clin Nutr 33(8): 1757-1766.
- Freeland-Graves JH, Friedman BJ, Han WH, Shorey RL, Young R (1982) Effect of zinc supplementation on plasma high-density lipoprotein cholesterol and zinc. Am J Clin Nutr 35(5): 988-992.
- Fung EB, Ritchie LD, Woodhouse LR, Roehl R, King JC (1997) Zinc absorption in women during pregnancy and lactation: A longitudinal study. Am J Clin Nutr 66(1): 80-88.
- Ganapathy SN, Booker LK, Craven R, Edwards CH (1981) Trace minerals, amino acids, and plasma proteins in adult men fed wheat diets. J Am Diet Assoc 78(5): 490-497.
- Gibson RS (1994) Content and bioavailability of trace elements in vegetarian diets. Am J Clin Nutr 59(5): 1223S-1232S.
- Gibson RS, Donovan UM, Heath AL (1997) Dietary strategies to improve the iron and zinc nutriture of young women following a vegetarian diet. Plant Foods Hum Nutr 51(1): 1-16.
- Gibson RS, Heath AL, Prosser N, Parnell W, Donovan UM, et al. (2000) Are young women with low iron stores at risk of zinc as well as iron deficiency? In: Roussel AM, editor; Anderson RA, editor, Favrier A, editor, eds. Trace Elements in Man and Animals 10. New York: Kluwer Academic. pp.323-328.
- Greger JL, Snedeker SM (1980) Effect of dietary protein and phosphorus levels on the utilization of zinc, copper and manganese by adult males. J Nutr 110(11): 2243-2253.
- Greger JL, Baligar P, Abernathy RP, Bennett OA, Peterson T (1978) Calcium, magnesium, phosphorus, copper, and manganese balance in adolescent females. Am J Clin Nutr 31(1): 117-121.
- Grider A, Bailey LB, Cousins RJ (1990) Erythrocyte metallothionein as an index of zinc status in humans. Proc Natl Acad Sci USA 87(4): 1259-1262.
- Günes C, Heuchel R, Georgiev O, Müller K-H, Lichtlen P, et al. (1998) Embryonic lethality and liver degeneration in mice lacking the metal-responsive transcriptional activator MTF-1. Embo J 17(10): 2846-2854.
- Gunshin H, Mackenzie B, Berger UV, Gunshin Y, Romero MF, et al. (1997) Cloning and characterization of a mammalian proton-coupled metal-ion transporter. Nature 388(6641): 482-488.
- Hallfrisch J, Powell A, Carafelli C, Reiser S, Prather ES (1987) Mineral balances of men and women consuming high fiber diets with complex or simple carbohydrate. J Nutr 117(1): 48-55.
- Hambidge KM (1989) Mild zinc deficiency in human subjects. In: Mills CF, editor., ed. Zinc in Human Biology. New York: Springer-Verlag. pp.281-296.
- Hambidge KM, Hambidge C, Jacobs M, Baum JD (1972) Low levels of zinc in hair, anorexia, poor growth, and hypogeusia in children. Pediatr Res 6(12): 868-874.
- Hambidge KM, Walravens PA, Casey CE, Brown RM, Bender C (1979) b. Plasma zinc concentrations of breast-fed infants. J Pediatr 94(4): 607-608.
- Hambidge KM, Krebs NF, Jacobs MA, Favier A, Guyette L, et al. (1983) Zinc nutritional status during pregnancy: A longitudinal study. Am J Clin Nutr 37(3): 429-442.
- Han O, Failla ML, Hill AD, Morris ER, Smith JC Jr (1994) Inositol phosphates inhibit uptake and transport of iron and zinc by a human intestinal cell line. J Nutr 124(4): 580-587.
- Harland BF, Peterson M (1978) Nutritional status of lacto-ovo vegetarian Trappist monks. J Am Diet Assoc 72(3): 259-264.
- Harland BF, Smith SA, Howard MP, Ellis R, Smith JC Jr (1988) Nutritional status and phytate:zinc and phytate x calcium:zinc dietary molar ratios of lacto-ovo vegetarian Trappist monks: 10 years later. J Am Diet Assoc 88(12): 1562-1566.
- Heinig MJ, Nommsen LA, Peerson JM, Lonnerdal B, Dewey KG (1993) Energy and protein intakes of breast-fed and formula-fed infants during the first year of life and their association with growth velocity: The DARLING Study. Am J Clin Nutr 58(2): 152-161.
- Hess FM, King JC, Margen S (1977) Zinc excretion in young women on low zinc intakes and oral contraceptive agents. J Nutr 107(9): 1610-1620.
- Holbrook JT, Smith JC Jr, Reiser S (1989) Dietary fructose or starch: Effects on copper, zinc, iron, manganese, calcium, and magnesium balances in humans. Am J Clin Nutr 49(6): 1290-1294.
- Hooper PL, Visconti L, Garry PJ, Johnson GE (1980) Zinc lowers high- density lipoprotein-cholesterol levels. J Am Med Assoc 244(17): 1960-1961.
- Hunt CD, Johnson PE, Herbel J, Mullen LK (1992) Effects of dietary zinc depletion on seminal volume and zinc loss, serum testosterone concentrations, and sperm morphology in young men. Am J Clin Nutr 56(1): 148-157.
- Hunt IF, Murphy NJ, Henderson C (1988) Food and nutrient intake of Seventh-day Adventist women. Am J Clin Nutr 48(3): 850-851.
- Hunt JR (1996) Bioavailability algorithms in setting recommended dietary allowances: Lessons from iron, applications to zinc. J Nutr 126(9): 2345S-2353S.
- Hunt JR, Mullen LK, Lykken GI (1992) Zinc retention from an experimental diet based on the US FDA Total Diet Study. Nutr Res 12(11): 1335-1344.
- Hunt JR, Gallagher SK, Johnson LK, Lykken GI (1995) High- versus low-meat diets: Effects on zinc absorption, iron status, and calcium, copper, iron, magnesium, manganese, nitrogen, phosphorus, and zinc balance in postmenopausal women. Am J Clin Nutr 62(3): 621-632.
- Hunt JR, Matthys LA, Johnson LK (1998) Zinc absorption, mineral balance, and blood lipids in women consuming controlled lactoovovegetarian and omnivorous diets for 8 weeks. Am J Clin Nutr 67(3): 421-430.
- Huse M, Eck MJ, Harrison SC (1998) A Zn2+ ion links the cytoplasmic tail of CD4 and the N-terminal region of Lck. J Biol Chem 273(30): 18729-18733.
- Jackson MJ, Jones DA, Edwards RH, Swainbank IG, Coleman ML (1984) Zinc homeostasis in man: Studies using a new stable isotope- dilution technique. Br J Nutr 51(2): 199-208.
- Jacob C, Maret W, Vallee BL (1998) Control of zinc transfer between thionein, metallothionein, and zinc proteins. Proc Natl Acad Sci USA 95(5): 3489-3494.
- Janelle KC, Barr SI (1995) Nutrient intakes and eating behavior scores of vegetarian and nonvegetarian women. J Am Diet Assoc 95: 180-186.
- Johansson G, Widerstrom L (1994) Change from mixed diet to lactovegetarian diet: Influence on IgA levels in blood and saliva. Scand J Dent Res 102(6): 350-354.
- Johnson MA, Baier MJ, Greger JL (1982) Effects of dietary tin on zinc, copper, iron, manganese, and magnesium metabolism of adult males. Am J Clin Nutr 35(6): 1332-1338.
- Johnson PE, Evans GW (1978) Relative zinc availability in human breast milk, infant formulas, and cow's milk. Am J Clin Nutr 31(3): 416-421.
- Johnson PE, Hunt CD, Milne DB, Mullen LK (1993) Homeostatic control of zinc metabolism in men: Zinc excretion and balance in men fed diets low in zinc. Am J Clin Nutr 57(4): 557-565.
- Kadrabova J, Madaric A, Kovacikova Z, Ginter E (1995) Selenium status, plasma zinc, copper, and magnesium in vegetarians. Biol Trace Elem Res 50(1): 13-24.
- Kaji M, Gotoh M, Takagi Y, Masuda H, Kimura Y, et al. (1998) Studies to determine the usefulness of the zinc clearance test to diagnose marginal zinc deficiency and the effects of oral zinc supplementation for short children. J Am Coll Nutr 17(4): 388-391.
- Kant AK, Moser Veillon PB, Reynolds RD (1989) Dietary intakes and plasma concentrations of zinc, copper, iron, magnesium, and selenium of young, middle aged, and older men. Nutr Res 9(7): 717-724.
- Kauwell GP, Bailey LB, Gregory JF 3rd, Bowling DW, Cousins RJ (1995) Zinc status is not adversely affected by folic acid supplementation and zinc intake does not impair folate utilization in human subjects. J Nutr 125(1): 66–72.
- Kelsay JL, Frazier CW, Prather ES, Canary JJ, Clark WM, et al (1988) Impact of variation in carbohydrate intake on mineral utilization by vegetarians. Am J Clin Nutr 48(3): 875-879.
- Kies CV (1988) Mineral utilization of vegetarians: Impact of variation in fat intake. Am J Clin Nutr 48(3): 884–887.
- King JC (1990) Assessment of zinc status. J Nutr 120(11): 1474-1479.
- King JC, Keen CL (1999) Zinc. In: Shils ME, editor, Olson JA, editor, Shike M, editor, Ross AC, editor, eds. Modern Nutrition in Health and Disease, 9th ed Baltimore, Williams & Wilkins, pp.223-239.
- King JC, Turnlund JR (1989) Human zinc requirements. In: Mills CF, editor, ed. Zinc in Human Biology. London: Springer-Verlag, pp.335-350.
- King JC, Stein T, Doyle M (1981) Effect of vegetarianism on the zinc status of pregnant women. Am J Clin Nutr 34(6): 1049-1055.
- King JC, Hambidge KM, Westcott JL, Kern DL, Marshall G (1994) Daily variation in plasma zinc concentrations in women fed meals at six- hour intervals. J Nutr 124(4): 508-516.
- Kirksey A, Ernst JA, Roepke JL, Tsai TL (1979) Influence of mineral intake and use of oral contraceptives before pregnancy on the mineral content of human colostrum and of more mature milk. Am J Clin Nutr 32(1): 30-39.
- Klug A, Schwabe JW (1995) Protein motifs 5. Zinc fingers. FASEB J 9(8): 597-604.
- Krajcovicova Kudlackova M, Simoncic R, Babinska K, Bederova A, Brtkova A, et al. (1995) Selected vitamins and trace elements in blood of vegetarians. Ann Nutr Metab 39(6): 334-339.
- Krebs NF, Hambidge KM, Jacobs MA, Rasbach JO (1985) The effects of a dietary zinc supplement during lactation on longitudinal changes in maternal zinc status and milk zinc concentrations. Am J Clin Nutr 41(3): 560-570.
- Krebs NF, Reidinger CJ, Hartley S, Robertson AD, Hambidge KM (1995) Zinc supplementation during lactation: Effects on maternal status and milk zinc concentrations. Am J Clin Nutr 61(5): 1030-1036.
- Kumar S (1976) Effect of zinc supplementation on rats during pregnancy. Nutr Rpts Intl 13: 33-36.
- Lee HH, Prasad AS, Brewer GJ, Owyang C (1989) Zinc absorption in human small intestine. Am J Physiol 256 (1 Pt 1): G87-G91.
- Levin N, Rattan J, Gilat T (1986) Mineral intake and blood levels in vegetarians. Isr J Med Sci 22(2): 105-108.
- Lin RS, Rodriguez C, Veillette A, Lodish HF (1998) Zinc is essential for binding of p56lck to CD4 and CD8α*. J Biol Chem 273(49): 32878-32882.
- Lonnerdal B (1989) Intestinal absorption of zinc. In: Mills CF (edt), Zinc in Human Biology. New York: Springer-Verlag. pp.33-55.
- Lonnerdal B, Keen CL, Hurley LS (1981) Iron, copper, zinc and manganese in milk. Ann Rev Nutr 1: 149-174.
- Lonnerdal B, Bell JG, Hendrickx AG, Burns RA, Keen CL (1988) Effect of phytate removal on zinc absorption from soy formula. Am J Clin Nutr 48(5): 1301-1306.
- Löwik MR, Schrijver J, Odink J, van den Berg H, Wedel M (1990) Long- term effects of a vegetarian diet on the nutritional status of elderly people (Dutch Nutrition Surveillance System). J Am Coll Nutr 9(6): 600-609.
- Mahalko JR, Sandstead HH, Johnson LK, Milne DB (1983) Effect of a moderate increase in dietary protein on the retention and excretion of Ca, Cu, Fe, Mg, P, and Zn by adult males. Am J Clin Nutr 37(1): 8-14.
- Mares-Perlman JA, Subar AF, Block G, Greger JL, Luby MH (1995) Zinc intake and sources in the US adult population: 1976–1980. J Am Coll Nutr 14(4): 349-357.
- McCabe MJ Jr, Jiang SA, Orrenius S (1993) Chelation of intracellular zinc triggers apoptosis in mature thymocytes. Lab Invest 69: 101-110.
- McKenna AA, Ilich JZ, Andon MB, Wang C, Matkovic V (1997) Zinc balance in adolescent females consuming a low- or high-calcium diet. Am J Clin Nutr 65(5): 1460-1464.
- McMahon RJ, Cousins RJ (1998) Mammalian zinc transporters. J Nutr 128(4): 667-670.
- Merialdi M, Caulfield LE, Zavaleta N, Figueroa A, DiPietro JA (1998) Adding zinc to prenatal iron and folate tablets improves fetal neurobehavioral development. Am J Obstet Gynecol 180 (2 Pt 1): 483-490.
- Miller LV, Hambidge KM, Naake VL, Hong Z, Westcott JL, et al. (1994) Size of the zinc pools that exchange rapidly with plasma zinc in humans: Alternative techniques for measuring and relation to dietary zinc intake. J Nutr 124(2): 268-276.
- Miller LV, Krebs NF, Hambidge KM (1998) Human zinc metabolism: Advances in the modeling of stable isotope data. Adv Exp Med Biol 445: 253–269.
- Milne DB, Canfield WK, Mahalko JR, Sandstead HH (1983) Effect of dietary zinc on whole body surface loss of zinc: Impact on estimation of zinc retention by balance method. Am J Clin Nutr 38(2): 181-186.
- Milne DB, Canfield WK, Mahalko JR, Sandstead HH (1984) Effect of oral folic acid supplements on zinc, copper, and iron absorption and excretion. Am J Clin Nutr 39(4): 535-539.
- Milne DB, Canfield WK, Gallagher SK, Hunt JR, Klevay LM (1987) Ethanol metabolism in postmenopausal women fed a diet marginal in zinc. Am J Clin Nutr 46(4): 688-693.
- Moser PB, Reynolds RD (1983) Dietary zinc intake and zinc concentrations of plasma, erythrocytes, and breast milk in antepartum and postpartum lactating and nonlactating women: A longitudinal study. Am J Clin Nutr 38(1): 101-108.
- Moser-Veillon PB, Reynolds RD (1990) A longitudinal study of pyridoxine and zinc supplementation of lactating women. Am J Clin Nutr 52(1): 135-141.
- Moss AJ, Levy AS, Kim I, Park YK (1989) Use of Vitamin and Mineral Supplements in the United States: Current Users, Types of Products, and Nutrients. Advance Data, Vital and Health Statistics of the National Center for Health Statistics, Number 174. Hyattsville, MD: National Center for Health Statistics.
- Nakamura T, Nishiyama S, Futagoishi-Suginohara Y, Matsuda I, Higashi A (1993) Mild to moderate zinc deficiency in short children: Effect of zinc supplementation on linear growth velocity. J Pediatr 123(1): 65-69.
- Neggers YH, Goldenberg RL, Tamura T, Johnston KE, Copper RL, DuBard M (1997) Plasma and erythrocyte zinc concentrations and their relationship to dietary zinc intake and zinc supplementation during pregnancy in low-income African-American women. J Am Diet Assoc 97(11): 1269-1274.
- Ninh NX, Thissen JP, Collette L, Gerard G, Khoi HH, et al. (1996) Zinc supplementation increases growth and circulating insulin- like growth factor I (IGF-I) in growth-retarded Vietnamese children. Am J Clin Nutr 63(4): 514-519.
- Oberleas D, Muhrer ME, O'Dell BL (1966) Dietary metal-complexing agents and zinc availability in the rat. J Nutr 90(1): 56-62.
- O'Brien KO, Zavaleta N, Caulfield LE, Wen J, Abrams SA (2000) Prenatal iron supplements impair zinc absorption in pregnant Peruvian women. J Nutr 130(9): 2251-2255.
- Ortega RM, Andres P, Martinez RM, Lopez-Sobaler AM, Quintas ME (1997) Zinc levels in maternal milk: The influence of nutritional status with respect to zinc during the third trimester of pregnancy. Eur J Clin Nutr 51(4): 253-258.
- Paik HY, Joung H, Lee JY, Lee HK, King JC, et al. (1999) Serum extracellular superoxide dismutase activity as an indicator of zinc status in humans. Biol Trace Elem Res 69(1): 45-57.
- Payette H, Gray-Donald K (1991) Dietary intake and biochemical indices of nutritional status in an elderly population, with estimates of the precision of the 7-d food record. Am J Clin Nutr 54(3): 478-488.
- Picciano MF, Guthrie HA (1976) Copper, iron, and zinc contents of mature human milk. Am J Clin Nutr 29(3): 242-254.
- Prasad AS (1976) Deficiency of zinc in man and its toxicity. In: Prasad AS, editor, Oberleas D (edt) Trace Elements in Human Health and Disease, Volume 1. Zinc and Copper. New York: Academic Press. pp.1-20.
- Prasad AS (1991) Discovery of human zinc deficiency and studies in an experimental human model. Am J Clin Nutr 53(2): 403-412.
- Prasad AS, Brewer GJ, Schoomaker EB, Rabbani P (1978) Hypocupremia induced by zinc therapy in adults. J Am Med Assoc 240(20): 2166-2168.
- Prasad AS, Fitzgerald JT, Hess JW, Kaplan J, Pelen F, et al. (1993) Zinc deficiency in elderly patients. Nutrition 9(3): 218-224.
- Prasad AS, Mantzoros CS, Beck FW, Hess JW, Brewer GJ (1996) Zinc status and serum testosterone levels of healthy adults. Nutrition 12(5): 344-348.
- Roesijadi G, Bogumil R, Vasak M, Kagi JH (1998) Modulation of DNA binding of a tramtrack zinc finger peptide by the metallothionein- thionein conjugate pair. J Biol Chem 273(28): 17425-17432.
- Rossander-Hulten L, Brune M, Sandstrom B, Lonnerdal B, Hallberg L (1991) Competitive inhibition of iron absorption by manganese and zinc in humans. Am J Clin Nutr 54(1): 152-156.
- Roth HP, Kirchgessner M (1985) Utilization of zinc from picolinic or citric acid complexes in relation to dietary protein source in rats. J Nutr 115(12): 1641-1649.
- Ruz M, Cavan KR, Bettger WJ, Gibson RS (1992) Erythrocytes, erythrocyte membranes, neutrophils and platelets as biopsy materials for the assessment of zinc status in humans. Br J Nutr 68(2): 515-527.
- Samman S, Roberts DC (1987) The effect of zinc supplements on plasma zinc and copper levels and the reported symptoms in healthy volunteers. Med J Aust 146(5): 246-249.
- Samman S, Roberts DC (1988) The effect of zinc supplements on lipoproteins and copper status. Atherosclerosis 70(3): 247-252.
- Samman S, Soto S, Cooke L, Ahmad Z, Farmakalidis E (1996) Is erythrocyte alkaline phosphatase activity a marker of zinc status in humans? Biol Trace Elem Res 51(3): 285-291.
- Sandstrom B, Lonnerdal B (1989) Promoters and antagonists of zinc absorption. In: Mills CF (edt) Zinc in Human Biology. New York: Springer-Verlag. pp.57-78.
- Sandström B, Cederblad A, Lonnerdal B (1983) Zinc absorption from human milk, cow's milk, and infant formulas. Am J Dis Child 137(8): 726-729.
- Scholl TO, Hediger ML, Schall JI, Fischer RL, Khoo CS (1993) Low zinc intake during pregnancy: Its association with preterm and very preterm delivery. Am J Epidemiol 137(10): 1115-1124.
- Seal CJ, Heaton FW (1985) Effect of dietary picolinic acid on the metabolism of exogenous and endogenous zinc in the rat. J Nutr 115(8): 986-993. Sian L, Mingyan X, Miller LV, Tong L, Krebs NF, et al. (1996) Zinc absorption and intestinal losses of endogenous zinc in young Chinese women with marginal zinc intakes. Am J Clin Nutr 63(3): 348-353.
- Sievers E, Oldigs HD, Dorner K, Schaub J (1992) Longitudinal zinc balances in breast-fed and formula-fed infants. Acta Paediatr 81(1): 1-6.
- Singh H, Flynn A, Fox PF (1989) Zinc binding in bovine milk. J Dairy Res 56(2): 249-263.
- Smit Vanderkooy PD, Gibson RS (1987) Food consumption patterns of Canadian preschool children in relation to zinc and growth status. Am J Clin Nutr 45(3): 609-616.
- Snedeker SM, Smith SA, Greger JL (1982) Effect of dietary calcium and phosphorus levels on the utilization of iron, copper, and zinc by adult males. J Nutr 112(1): 136-143.
- Solomons NW, Jacob RA (1981) Studies on the bioavailability of zinc in humans: Effects of heme and nonheme iron on the absorption of zinc. Am J Clin Nutr 34(4): 475-482.
- Spencer H, Asmussen CR, Holtzman RB, Kramer L (1979) Metabolic balances of cadmium, copper, manganese, and zinc in man. Am J Clin Nutr 32(9): 1867-1875.
- Spencer H, Kramer L, Norris C, Osis D (1984) Effect of calcium and phosphorus on zinc metabolism in man. Am J Clin Nutr 40(6): 1213-1218.
- Srikumar TS, Johansson GK, Ockerman PA, Gustafsson JA, Akesson B (1992) Trace element status in healthy subjects switching from a mixed to a lactovegetarian diet for 12 months. Am J Clin Nutr 55(4): 885-890.
- Sullivan VK, Burnett FR, Cousins RJ (1998 Metallothionein expression is increased in monocytes and erythrocytes of young men during zinc supplementation. J Nutr 128(4): 707-713.
- Swanson CA, King JC (1982) Zinc utilization in pregnant and nonpregnant women fed controlled diets providing the zinc RDA. J Nutr 112(4): 697-707.
- Swanson CA, King JC (1987) Zinc and pregnancy outcome. Am J Clin Nutr 46(5): 763-771.
- Swanson CA, Mansourian R, Dirren H, Rapin CH (1988) Zinc status of healthy elderly adults: Response to supplementation. Am J Clin Nutr 48(2): 343-349.
- Taper LJ, Hinners ML, Ritchey SJ (1980) Effects of zinc intake on copper balance in adult females. Am J Clin Nutr 33(5):1077-1082.
- Taylor CM, Bacon JR, Aggett PJ, Bremner I (1991) Homeostatic regulation of zinc absorption and endogenous losses in zinc-deprived men. Am J Clin Nutr 53(3): 755-763.
- Telford WG, Fraker PJ (1995) Preferential induction of apoptosis in mouse CD4+CD8+αβTCRIoCD3εIothymocytes by zinc. J Cell Physiol 164(2): 259-270.
- Thomas AJ, Bunker VW, Hinks LJ, Sodha N, Mullee MA, et al. (1988) Energy, protein, zinc and copper status of twenty-one elderly inpatients: Analysed dietary intake and biochemical indices. Br J Nutr 59(2): 181-191.
- Thomas EA, Bailey LB, Kauwell GA, Lee DY, Cousins RJ (1992) Erythrocyte metallothionein response to dietary zinc in humans. J Nutr 122(12): 2408-2414.
- Turnlund JR, Michel MC, Keyes WR, King JC, Margen S (1982) Use of enriched stable isotopes to determine zinc and iron absorption in elderly men. Am J Clin Nutr 35(5): 1033-1040.
- Turnlund JR, King JC, Keyes WR, Gong B, Michel MC (1984) A stable isotope study of zinc absorption in young men: Effects of phytate and alpha-cellulose. Am J Clin Nutr 40(5): 1071-1077.
- Turnlund JR, Durkin N, Costa F, Margen S (1986) Stable isotope studies of zinc absorption and retention in young and elderly men. J Nutr 116(7): 1239-1247.
- Turnlund JR, Keyes WR, Hudson CA, Betschart AA, Kretsch MJ, Sauberlich HE (1991) A stable-isotope study of zinc, copper, and iron absorption and retention by young women fed vitamin B-6-deficient diets. Am J Clin Nutr 54(6): 1059-1064.
- Udomkesmalee E, Dhanamitta S, Yhoung Aree J, Rojroongwasinkul N, Smith JC Jr (1990) Biochemical evidence suggestive of suboptimal zinc and vitamin A status in schoolchildren in northeast Thailand. Am J Clin Nutr 52(3): 564-567.
- Valberg LS, Flanagan PR, Chamberlain MJ (1984) Effects of iron, tin, and copper on zinc absorption in humans. Am J Clin Nutr 40(3): 536-541.
- Vallee BL, Galdes A (1984) The metallobiochemistry of zinc enzymes. Adv Enzymol 56: 283-429.
- Vuori E, Makinen SM, Kara R, Kuitunen P (1980) The effects of the dietary intakes of copper, iron, manganese, and zinc on the trace element content of human milk. Am J Clin Nutr 33(2): 227-231.
- Wada L, King JC (1986) Effect of low zinc intakes on basal metabolic rate, thyroid hormones and protein utilization in adult men. J Nutr 116(6): 1045-1053.
- Wada L, Turnlund JR, King JC (1985) Zinc utilization in young men fed adequate and low zinc intakes. J Nutr 115(10): 1345-1354.
- Wastney ME, Aamodt RL, Rumble WF, Henkin RI (1986) Kinetic analysis of zinc metabolism and its regulation in normal humans. Am J Physiol 251(2 Pt 2): R398-R408.
- Wastney ME, Aamodt RL, Rumble WF, Henkin RI (1986) Kinetic analysis of zinc metabolism and its regulation in normal humans. Am J Physiol 251(2 Pt 2): R398-R408.
- Whittaker P (1998) Iron and zinc interactions in humans. Am J Clin Nutr 68(2): 442S-446S.
- WHO (World Health Organization) (1996) Trace Elements in Human Nutrition and Health. Geneva: WHO, pp.72-104.
- Williams AW, Erdman JW Jr (1999) Food processing: Nutrition, safety, and quality balances. In: Shils ME, editor, Olson JA, editor, Shike M, editor, Ross AC, editor, eds. Modern Nutrition in Health and Disease, 9th ed. Baltimore: Williams & Wilkins. pp.1813-1821.
- Wisker E, Nagel R, Tanudjaja TK, Feldheim W (1991) Calcium, magnesium, zinc, and iron balances in young women: Effects of a low-phytate barley-fiber concentrate. Am J Clin Nutr 54(3): 553-559.
- Wood RJ, Zheng JJ (1997) High dietary calcium intakes reduce zinc absorption and balance in humans. Am J Clin Nutr 65(6): 1803-1809.
- Yadrick MK, Kenney MA, Winterfeldt EA (1989) Iron, copper, and zinc status: Response to supplementation with zinc or zinc and iron in adult females. Am J Clin Nutr 49(1): 145-150.
- Yokoi K, Alcock NW, Sandstead HH (1994) Iron and zinc nutriture of premenopausal women: Associations of diet with serum ferritin and plasma zinc disappearance and of serum ferritin with plasma zinc and plasma zinc disappearance. J Lab Clin Med 124(6): 852-861.
- Yuzbasiyan-Gurkan V, Grider A, Nostrant T, Cousins RJ, Brewer GJ (1992) Treatment of Wilson's disease with zinc: X. Intestinal metallothionein induction. J Lab Clin Med 120(3): 380-386.
- Zalewski PD, Forbes IJ, Seamark RF, Borlinghaus R, Betts WH, et al. (1994) Flux of intracellular labile zinc during apoptosis (gene-directed cell death) revealed by a specific chemical probe, Zinquin. Chem Biol 1(3): 153-161.
- Ziegler EE, Edwards BB, Jensen RL, Filer LJ, Fomon SJ (1978) Zinc balance studies in normal infants. In: Kirchgessner M, editor, ed. Trace Element Metabolism in Man and Animals-3. Freising-Weihenstephan: Arbeitskreis fier Tierernahrungsforschung. pp.292-295.
-
Khajik Sirob Yaqob. ‘’ZINC ‘’ The Friendly and Forgotten Micronutrient. Glob J of Ped & Neonatol Car. 1(3): 2019. GJPNC.MS.ID.000512.
Congenital CMV Infection, Brain Injury, Pseudocyst, SEPC, CMV infection, viral infections, pseudocysts
-

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.






