 Case Report
Case Report
Nonimmune Hydrops Fetalis secondary to Primary Superior Vena Cava Obstruction
Tara N Shepherd MD, MS1,2* and Luke Viehl MD1,2
1 Department of Pediatrics, Washington University in St. Louis School of Medicine, St. Louis, MO, USA
2 St. Louis Children’s Hospital, St. Louis, MO, USA
Tara N Shepherd, Department of Pediatrics, Emory University School of Medicine, Atlanta, GA, USA.
Received Date:November 04, 2025; Published Date:November 10, 2025
Abstract
Hydrops fetalis, characterized by two or more abnormal fetal fluid collections, can be effectively identified during antenatal ultrasound. Historically, red cell alloimmunization has been the primary cause of hydrops fetalis infection. However, currently, the majority of cases are attributed to nonimmune hydrops. The underlying etiologies for nonimmune hydrops fall into four major categories: increased hydrostatic capillary pressure, reduction in intravascular osmotic pressure, obstruction of lymphatic flow, and damage to peripheral capillary integrity. Obstruction of lymphatic flow and subsequent development of hydrops fetalis are rare and serious clinical problems. This case report highlights the clinical presentation and diagnostic challenges in a case of nonimmune hydrops fetalis secondary to superior vena cava obstruction.
Keywords: Superior vena cava syndrome; Nonimmune hydrops fetalis; Chylothorax; Case report
Abbreviations: NIHF: Nonimmune hydrops fetalis; SVC: Superior vena cava; RPR: Rapid plasma regain, CPAP: Continuous positive airway pressure; NICU: Neonatal intensive care unit; MCT: Medium-chain triglyceride; CTA: Computed tomography angiography
Introduction
Hydrops fetalis is characterized by disrupted management of the flow and absorption of fetal fluid. It is diagnosed by two or more abnormal fetal fluid collections. These include ascites, pleural effusion, pericardial effusion, and generalized skin edema (defined as a skin thickness >5 mm) [1]. Classically, most cases of hydrops fetalis have been attributed to red blood cell alloimmunization. However, currently, most cases are classified as nonimmune hydrops fetalis (NIHF). The fluid accumulation in non-immune hydrops fetalis can be explained by four main mechanisms: (one) increased hydrostatic capillary pressure, (two) reduction of intravascular osmotic pressure, (three) obstruction of lymphatic flow, and (four) damage to peripheral capillary integrity [2]. A range of fetal pathologies, including congenital heart disease, genetic abnormalities, lymphatic malformations, hematologic disorders, infections, and idiopathic factors can influence these mechanisms. Identifying and understanding the underlying conditions play a crucial role in understanding the etiology of NIHF.
Chylothorax occurs when the lymphatic fluid accumulates in the pleural space. Chylothoraces can be either acquired or congenital. Acquired chylothoraces are often secondary to thoracic surgery trauma, or thrombosis. Congenital chylothorax is often associated with abnormal lymphatic pathway development. Although rare, congenital chylothorax remains the most common cause of neonatal pleural effusions [3]. Chylous fluid contains >1000 white blood cells per microliter, with more than 70-80% lymphocytes. It has a similar protein content to plasma and a triglyceride concentration of >1000 mg/dL in feeding patients [4].
Superior vena cava (SVC) obstruction, particularly due to venous thrombosis, is a rare but documented cause of significant complications in pediatric patients [5]. SVC plays a critical role in returning blood from the upper body to the heart, and any obstruction can lead to increased venous pressure. The thoracic duct, which drains lymphatic fluid into the venous system at the junction of the subclavian and internal jugular veins, is particularly vulnerable to elevated pressure in cases of SVC obstruction. This pressure can disrupt lymphatic drainage, leading to accumulation of chyle in the pleural cavity, known as chylothorax. In this report, we describe a patient with congenital SVC obstruction that progressed to bilateral chylothoraces, causing widespread fluid accumulation, ultimately leading to hydrops fetalis.
Case Presentation
A 3.66 kg female infant born at 35 weeks via cesarean section due to preterm labor was referred to a fetal care center at 32 weeks for evolving hydrops. Ultrasonography showed polyhydramnios and bilateral pleural effusions (Figure 1). Follow-up ultrasound at 33 weeks revealed bilateral pleural effusions, ascites, and scalp edema. Amniotic fluid tests for cytomegalovirus DNA, Toxoplasmosis gondii PCR, and Parvovirus B19 PCR were negative, as was a nonimmune hydrops panel. A chromosomal microarray analysis confirmed a normal female karyotype. Maternal tests were negative for HIV, rapid plasma regain (RPR), and Group B Streptococcus. The maternal blood type was positive and indirect Coombs negative. The patient was diagnosed with Nonimmune hydrops (NIHF).
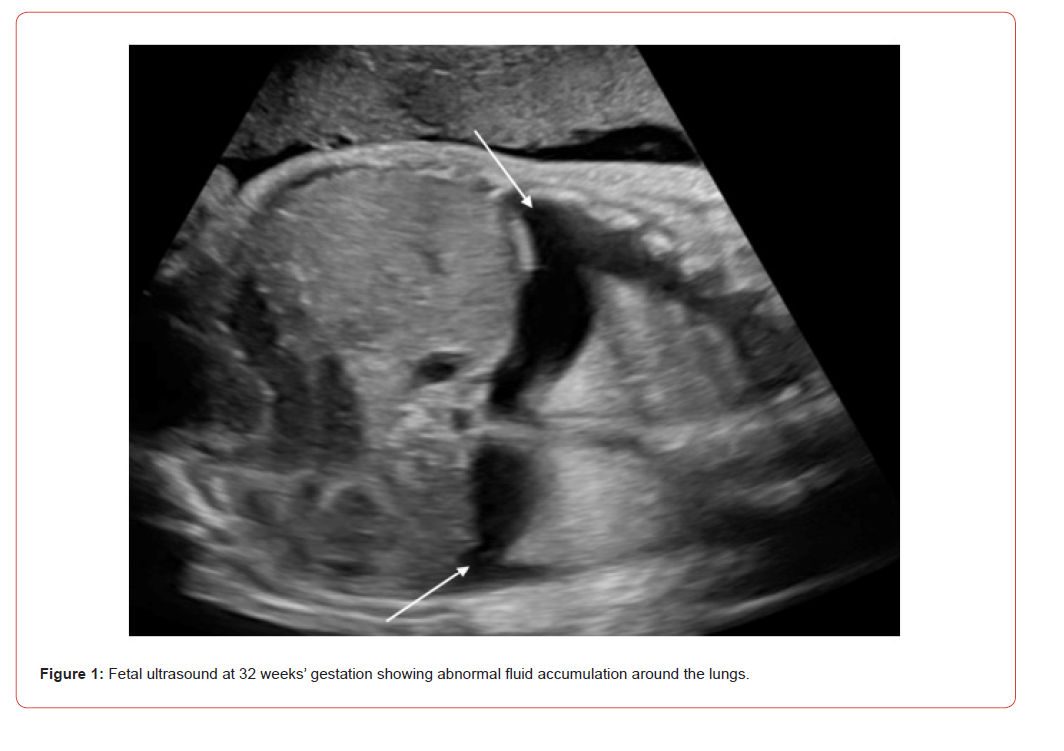
The infant had Apgar scores of 4 and 7 at one and five minutes, respectively. Owing to respiratory distress, she was placed on continuous positive airway pressure (CPAP) and transferred to the neonatal intensive care unit (NICU). Chest radiography confirmed bilateral pleural effusions, and intubation was required until day 9. A unilateral chest tube was placed until day 10, and the contralateral effusion was drained via thoracentesis. The pleural fluid was cloudy yellow, with 94% lymphocytes and 4% neutrophils, and was chylous, based on biochemical analysis (pH 7.60, protein > 600 mg/dL, glucose 82 mg/dL). After respiratory stabilization, she was treated with a traditional formula, but the effusions reaccumulated, and she was subsequently placed on a medium-chain triglyceride (MCT) formula.
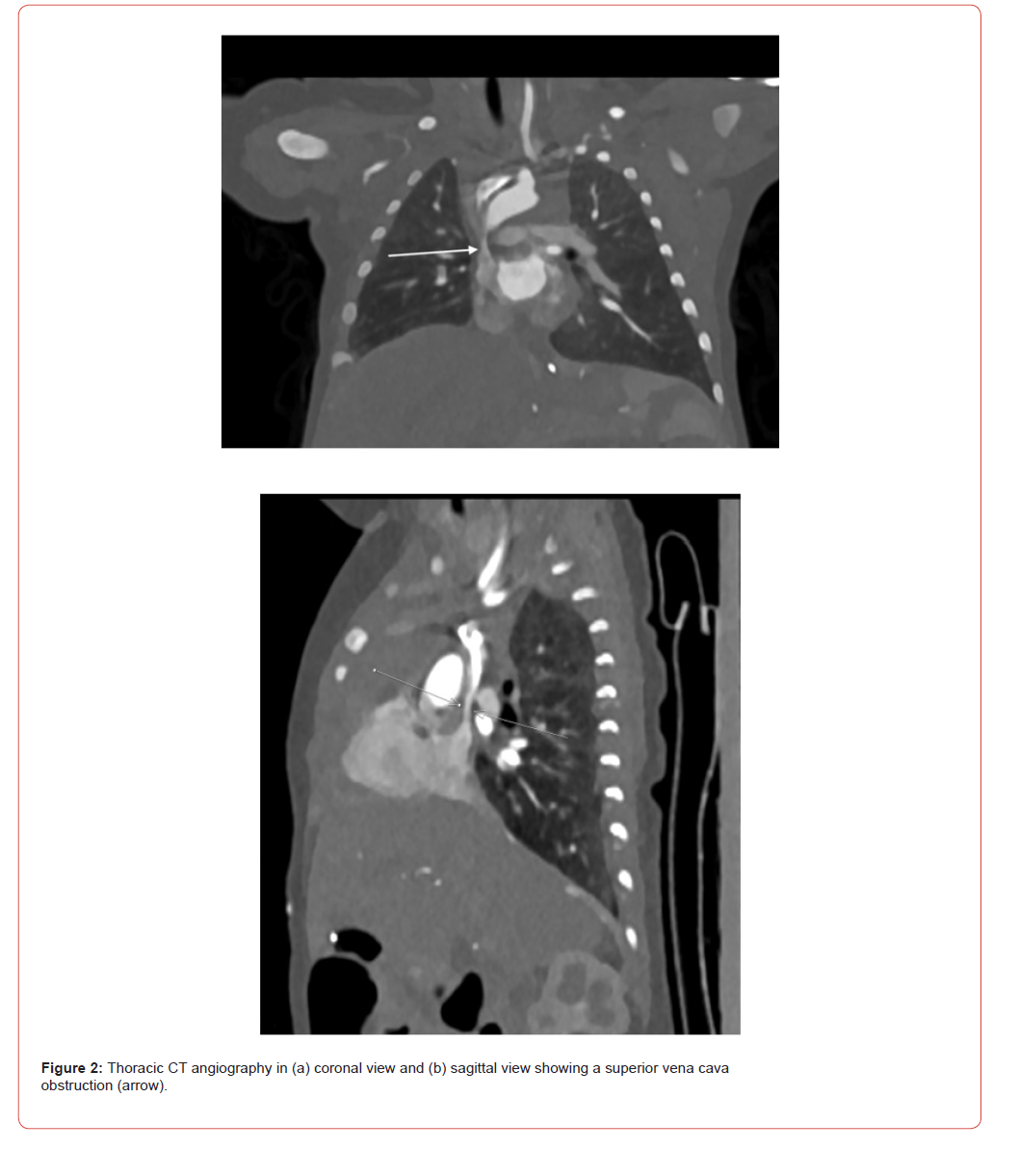
Her evaluation for nonimmune hydrops included a normal head and abdominal ultrasound. Rapid genomic and mitochondrial DNA sequencing revealed no pathogenic variants. On the first day, echocardiogram showed normal anatomy and chamber function but detected flow acceleration in the SVC. Follow-up echocardiograms on days four and twenty-five confirmed abnormal SVC flow with both acceleration and continuous flow patterns. Computed tomography angiography (CTA) further examined this finding, revealing that flow acceleration began near the right pulmonary artery (Figure 2). Three months after discharge, SVC flow remained abnormal, leading to the decision to perform SVC stenting.
Discussion
The development of fetal hydrops occurs because of an imbalance between the production of interstitial fluid and its proper drainage through the lymphatic system. The features of hydrops fetalis are detected by ultrasound and defined as the presence of two or more abnormal fluid collections in the fetus. These include ascites, pleural effusion, pericardial effusion, and generalized skin edema (defined as a skin thickness >5 mm) [1]. Immune-mediated hydrops was the predominant cause before the introduction of immunoglobulin prophylaxis for at-risk mothers. Currently, most reported cases involve nonimmune hydrops.
Four mechanisms explain the abnormal accumulation of fluid in NIHF: increased hydrostatic capillary pressure, reduction in intravascular osmotic pressure, obstruction of lymphatic flow, and damage to peripheral capillary integrity [2]. In our patient, obstruction of lymphatic flow secondary to SVC obstruction appeared to be the primary mechanism for the development of fetal hydrops.
Thrombosis of the superior vena cava or a postoperative cardiac surgery complication resulting in the development of chylothorax has been described in the literature [5,6]. However, congenital superior vena cava obstruction causing hydrops fetalis is rare, as we found only one other case report in a PubMed and Medline search [7]. When there is an obstruction in the SVC, it can cause a significant increase in venous pressure. This elevated pressure can disrupt the normal circulation of the blood and lymphatic fluid throughout the body.
Consequently, changes in fluid dynamics can lead to the development of chylothorax, scalp edema, and ascites.
The management of nonimmune hydrops often involves providing comprehensive care to address the symptoms and complications associated with the condition. This involves close monitoring of the fetal and neonatal status, ultrasound evaluations, and laboratory investigations. In addition to supportive care, targeted interventions may be required based on the specific etiology identified [8].
Conclusion
Non-immune hydrops fetalis, marked by abnormal fluid accumulation in the fetus, poses significant challenges in neonatal care owing to its high mortality rate. Early diagnosis and understanding of underlying causes are critical for effective management and improved outcomes. Our infant presented with congenital superior vena cava obstruction and bilateral pleural effusions that required pleural fluid drainage and medium-chain triglyceride formula. After the initial conservative treatment, outpatient monitoring with sequential echocardiograms revealed the need for SVC stenting, prompted by increasing head circumference and concerns for potential development of hydrocephalus and SVC syndrome. This case highlights the clinical progression and management strategies for this rare condition and offers insights into its complexities and therapeutic approaches.
Conflict of Interest
We have no financial interest or any conflict of interest.
References
- Society for Maternal-Fetal Medicine (SMFM), Norton ME, Chauhan SP, Dashe JS (2015) Society for Maternal-Fetal Medicine (SMFM) clinical guideline #7: nonimmune hydrops fetalis. Am J Obstet Gynecol 212(2): 127-139.
- Swearingen C, Colvin ZA, Leuthner SR (2020) Nonimmune Hydrops Fetalis. Clinical Perinatology. Clin Perinatol 47(1): 105-121.
- Brock WW, Bradshaw WT (2016) Congenital Chylothorax: A Unique Presentation of Nonimmune Hydrops Fetalis in a Preterm Infant. Adv Neonatal Care 16(2): 114-123.
- Attar MA, Donn SM (2017) Congenital chylothorax. Semin Fetal Neonatal Med 22(4): 234-239.
- Beghetti M, La Scala G, Belli D, Bugmann P, Kalangos A, et al. (2000) Etiology and management of pediatric chylothorax. J Pediatr 136(5): 653-658.
- Barracano R, Scognamiglio G, Palma M, Giacomo S, Merola A, et al. (2021). Chylothorax Due to Superior Vena Cava Obstruction in a Patient with Complex Congenital Heart Disease. JACC Case Rep 3(5): 736-739.
- Adiotomre PNA, Burns JE, McIntosh N (1994) Hydrops foetalis and chylothorax associated with superior caval vein obstruction and resolution following balloon dilatation. Acta Paediatr 83(9): 983-985.
- Murphy JH (2004) Nonimmune Hydrops Fetalis. Neoreviews 5(1): e5-e15.
-
Tara N Shepherd MD, MS* and Luke Viehl MD. Nonimmune Hydrops Fetalis secondary to Primary Superior Vena Cava Obstruction. Glob J of Ped & Neonatol Car. 5(5): 2025. GJPNC.MS.ID.000623.
Superior vena cava syndrome, Nonimmune hydrops fetalis, Chylothorax, Case report, NIHF, Pediatric patients, Neonatal care
-

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.






