 Research Article
Research Article
Neurodevelopmental Outcomes of Very, Moderately and Late Preterm Infants and Infants with Hypoxic Ischemic Encephalopathy between 8 to 36 Months of Age: A Retrospective Study
Eileen R McGrath1*, Mark Borgstrom2, Cody Melcher2 and Jennie Jacob3
1Steele Children’s Research Center, College of Medicine, The University of Arizona, Tucson, AZ, USA
2University Information Technology Services, The University of Arizona, Tucson, AZ, USA
3Diamond Children’s Banner Medical Center, Tucson, AZ, USA
Eileen R McGrath, Steele Children’s Research Center, College of Medicine, The University of Arizona, Tucson, AZ, USA.
Received Date: September 10, 2024; Published Date: October 14, 2024
Abstract
The purpose of this retrospective study is to identify medical conditions impacting neurodevelopmental outcomes of very preterm, moderately
preterm, and late preterm infants including infants born with hypoxic ischemic encephalopathy from eight months through three years of age.
Infants born in Banner Diamond Children’s University Medical Center, receiving services in the Newborn Intensive Care Unit, and attending
Neonatal Developmental Follow-Up Clinic were identified. Participants received developmental assessment and follow-up from August 2012
through December 2019. Relevant clinical conditions during initial hospital stay and up to three years of age were obtained by reviewing medical
and developmental records. Bayley Scales of Infant Toddler Development (Bayley III) was used to evaluate skill development at 8 to 12 months,15
to 18 months, and 24 to 36 months.
This retrospective study reports significant association between head circumference (HC) and Hispanic origin with fine motor deficits at 8 to 12
months corrected age; Hispanic origin, and bronchopulmonary dysplasia (BPD) with oxygen at 36 weeks postmenstrual age (PMA) with receptive
language deficits at 8 to 12 months corrected age; and weight at the 10th percentile at the first developmental evaluation and cognitive delays at
8 to 12 months corrected age. Significant associations between maternal chorioamnionitis, isolated cerebellar hemorrhage, and BPD with oxygen
at 36 weeks PMA and expressive language deficits; PDA with NSAID and Caffeine with fine motor deficits; and isolated cerebellar hemorrhage and
nitric oxide with cognitive deficits at 15 to 18 months corrected age. During the 24 to 36-month evaluation period, when age was no longer corrected
for prematurity, many more variables were found significant for deficits in cognition, receptive and expressive language, and fine and gross motor
deficits.
Keywords: Very Preterm; Moderately Preterm; Late Preterm; Hypoxic Ischemic Encephalopathy; Neurodevelopmental Outcomes
Abbreviations: BPD: Bronchopulmonary Dysphagia; BSID-III: Bayley Scales of Infant and Toddler Development (3rd Edition); BW: Birth Weight; CPAP: Continuous Positive Airway Pressure; CP: Cerebral Palsy; DEX: Dexamethasone; EPT: Extremely Preterm; FiO2: Fraction of Inspired Oxygen; GA: Gestational Age; HC: Head Circumference; HIE: Hypoxic Ischemic Encephalopathy; iNO: Nitric Oxide; IVH: Intraventricular Hemorrhage; LPT: Late Preterm; MPT: Moderate Preterm; NDD: Neurodevelopmental Disorder; NICU: Neonatal Intensive Care Unit; PDA: Patent Ductus Arteriosus PMA: Post Menstrual Age; PVL: Periventricular Leukomalacia; RDS: Respiratory Distress Syndrome; SD: Standard Deviation; VLBW: Very Low Birth Weight; VPT: Very Preterm
Introduction
This purpose of this retrospective study is to identify medical conditions impacting neurodevelopmental outcomes and evaluate contributing factors leading to long-term suboptimal neurodevelopmental outcomes in very preterm (VPT) infants born between 28 and 32 weeks gestation, moderately preterm (MPT) infants born between 32 and 34 weeks gestation, late preterm infants (LPT) born between 34 and 36 weeks gestation and those infants born with hypoxic ischemic encephalopathy (HIE) born between 34 and 41 weeks gestation. Pertinent clinical details (head circumference (HC), birthweight (BW), Apgar scores, intraventricular hemorrhage (IVH), bronchopulmonary dysplasia (BPD), hypoxic ischemic encephalopathy (HIE)) infants experienced during their initial hospital stay and through three years of age were acquired through a review of developmental, diagnostic, and medical records.
Very preterm infants are those born from 28 weeks gestation to 31 weeks gestation and very low birth weight (VLBW) infants weigh less than 3.3 pounds or 1500 grams. A meta-analytic review conducted in 2018 by Pascal, Oostra, Naulaers, Ortibus and Van Den Broeck, of the neurodevelopmental outcomes of VPT and VLBW infants born in the past decade reported that preterm birth occur in 11.1% of all worldwide births and that 10% of those deliveries are VPT infants [1-4]. According to Liang, Rogers, and Smyser (2019)5 severe cognitive impairment occurs in 10% of VPT infants and mild to moderate impairment in 30 to 50% of VPT infants. The incidence of severe cerebral palsy (CP) has been found to affect 5 to 15% of VPT infants and mild to moderate CP occurs in 30% to 50%. Mild to moderate visual impairment occurs in 30% to 50% of VPT infants and hearing impairment in up to 14% of VPT infants. Twenty percent to thirty-five percent are diagnosed with a language impairment. These infants are more likely to repeat one or more grades, to require special education services and are more likely to end their education early. Seventy-four to eighty percent graduate from high school and only 30 to 32% matriculate to college [4]. Very preterm infants are less likely to be able to establish an independent adult life and show increased risk-taking behaviors. These infants continue to demonstrate increased rates of neurodevelopmental disability across domains, including cognitive, motor, language, hearing, vision, behavioral, and psychiatric deficits throughout the lifespan [5].
According to Liang et al, [5] eight percent of all live births or 75% of preterm births are infants born between 32 and 36 weeks gestation. These infants are identified as moderate or moderately preterm. While much of the research literature focuses on extremely preterm (EPT) and VPT infants, there is growing evidence of increased risk for adverse neurodevelopmental outcomes in the moderate to late preterm population. Risk levels vary; however, there seems to be a greater proportion of moderate to late preterm infants facing milder or more subtle symptoms. Research shows that premature birth at this gestational age range can lead to adverse developmental outcomes leading to difficulties throughout life [4,6-10]. For example, the risk for cognitive, language and social impairments is up to two times greater for the moderate-to-late preterm infant when compared to their full term counterparts. The risk for cerebral palsy and/or motor delays is two to three times greater. Moderate-to-late preterm infants have been found to have impaired school performance commonly with reading and writing. Research shows that 71% of moderate preterm infants achieve below their expected grade level and 36% of late preterm infants achieve below expected grade level. Regarding behavioral and psychiatric problems, moderate to late preterm infants can have an increased risk for psychiatric disorders (such as anxiety) during childhood [5,8,11-18].
According to epidemiologists Martin and Osterman (2024), [19] preterm birth rates in the United States have increased from 2014 to 2022. Late preterm and early preterm births (less than 34 weeks gestation) declined from 2007 to 2014; however, they have been on the rise in recent years. From 2014 to 2022, all preterm births increased by 12% and early preterm increased by 20% during the same time period. Additionally, full-term births declined by 6% and post-term births declined by 28%.
Hypoxic-ischemic encephalopathy (HIE) is a brain injury that can occur generally in moderate-to-late preterm infants when they don’t receive enough oxygen or blood flow to the brain before, during, or shortly after birth. HIE can affect the central nervous system and cause neurological or developmental problems. Infants born prematurely have a high incidence of neonatal brain injury, with detrimental effects on motor, cognitive, behavioral, social, attentional, and sensory outcomes. Increased survival in lower gestational ages is accompanied by increased suboptimal neurodevelopmental outcomes [20-26]. Between 24 and 40 weeks gestation, the human brain undergoes rapid changes that expose the developing brain to injury from hypoxic-ischemia, inflammation, free radical and excitotoxic damage. Even without exacerbating factors, preterm birth is associated with subtle white matter pathology [27]. According to Gopagondanahalli et al, [20] there are few well-designed studies that are able to provide definitive answers on the neurological outcomes following preterm HIE. In a 2012 study conducted by Chalak, Rollins, Morriss et al, all preterm infants with Stage 1 and Stage 2 HIE had normal neurological examinations and met their developmental milestones at 12 months of age [28]. They found that all the surviving infants in their study with Stage 3 HIE had poor neurological outcomes; two of the nine infants in their study died. Preterm HIE produces a complex, heterogeneous, and characteristic pattern of injury to the developing brain with a wide spectrum of clinical manifestations. It poses a great challenge for the treating physician to recognize, evaluate, and predict outcomes [20].
Methods
This retrospective study was approved by Institutional Review Board (IRB, Project Number: 151017061) to evaluate contributing factors leading to long-term suboptimal neurodevelopmental outcomes (motor, sensorineural or other developmental disorders) in extremely preterm infants born between 23 to 27 and 6/7 weeks gestation, very preterm infants born 28 to 33 and 6/7 weeks gestation and those with hypoxic ischemic encephalopathy (HIE) born between 34 to 41 weeks gestation. Neurodevelopmental outcomes of extremely preterm infants born between 23 to 27 and 6/7 weeks gestation have been published. This article will report on those very preterm infants born between 28 to 32 weeks gestation, moderately preterm infants born between 32- and 34-weeks’ gestation, late preterm infants born from 34 to 36 weeks gestation and those infants born with hypoxic ischemic encephalopathy (HIE) born at from 34 weeks to 41 weeks gestation. Pertinent clinical and demographic details (HC, BW, Apgar scores, IVH, BPD, HIE, male/ female, maternal age, Hispanic, maternal smoking) were acquired through a review of developmental, diagnostic, and medical records.
One hundred and forty-four (144) infants born from 2011 through 2015 who attended the Neonatal Developmental Followup Clinic from August 2012 through December of 2019, were evaluated using two standardized assessments. Prior to or at four months corrected age, the infants were evaluated using the Test of Infant Motor Performance (TIMP). The Bayley Scales of Infant Toddler Development (BSID-III) was then used to evaluate infants at approximately 6, 9, 12, 18, 24, and 36 months. Ages were corrected for each child until two years of age. During the study period, twenty-two (15%) infants were lost to follow-up and one child passed away. This cohort included eight sets of twins and four sets of triplets. Fifty-eight percent were non-Hispanic and 42% were Hispanic. Seventy-one of these infants were born between 28 and 41 weeks gestation. This article reports on the longitudinal data obtained on the very preterm, moderately preterm, and late preterm infants and on those infants born with HIE.
Bayley-III Descriptive Classifications for Composite Scores
The Bayley Scales of Infant and Toddler Development, 3rd Edition (BSID-III) is a standardized and norm-referenced neurobehavioral assessment developed to facilitate the identification of young children with neurodevelopmental disabilities, delays or those requiring intervention services [29,30]. The BSID-III evaluates cognitive, language and motor skill development in infants and toddlers from 16 days through 42 months and is widely used in research studies of the developmental outcomes of this age group and allows for correction of prematurity to be factored into developmental scoring. The BSID-III also includes adaptive behavior and social-emotional development questionnaires; however, these were excluded from this study.
Composite Scores derived from the BSID-III are a transformation of a distribution of scores that have a standard deviation (SD) and a given mean. The transformation allows for a measurement in units of SD of how far an individuals’ score is from the mean. Composite Scores are based on various sums of sub-test scaled scores for Language and Motor each of which have two subdomains and composite equivalents for scaled scores from the Cognitive domain, which has a single domain.
The composite scores are scaled to a metric with a range of 40 to 160, a mean of 100 and SD of 15. For example, a score of 100 on any of the composites identifies “Average” performance of a given age group. Scores of 85 and 115 are one SD below and one SD above the mean, respectively. Levels of Performance are as follows: Very Superior = 130 and above; Superior = 120-129; High Average = 110-119; Average = 90-109; Low Average = 80-89; Borderline = 70- 79; and Extremely Low = 69 and below.
Inclusion and Exclusion Criteria
Medically compromised and preterm infants born in and hospitalized at the University of Arizona Medical Center (now Banner Diamond Children’s University Medical Center) Newborn Intensive Care Unit (NICU) from January 1, 2012, through December 30, 2015, and followed through the Neonatal Developmental Follow-Up Clinic through three years of age were included in this retrospective study. Data collection concluded and participants completed follow-up visits by December 31, 2019. Two infants born at 28 weeks gestation outside of and transferred to this hospital are included.
Fifteen percent of those born during the study period were lost to follow-up and were excluded from data analysis. Additionally, those whose BSID-III evaluations were not completed between 8 to 12 months, 15 to 18 months, or 24 to 36 months were excluded. Infants or toddlers who missed one of these assessment points in time were not included. The exclusion criteria and loss to follow-up reduced the study size of this study to 73 infants and toddlers.
Data Abstraction
Sixty-one independent variables were identified and collected along with Composite Scores for Cognitive, Receptive and Expressive Language, Total Language, Gross and Fine Motor and Total Motor from the BSID-III. An Excel spreadsheet was developed to record relevant medical and diagnostic records and BSID-III scores at 8 to 12 months, 15 to 18 months and 24 to 36 for each participating child. Variables were marked as Yes/No for “existing” or “nonexistent” on the Excel spreadsheet after all medical data were collected. The list of study variables can be found in Appendix A.
Appendix A:Medical and Demographic Data Collected
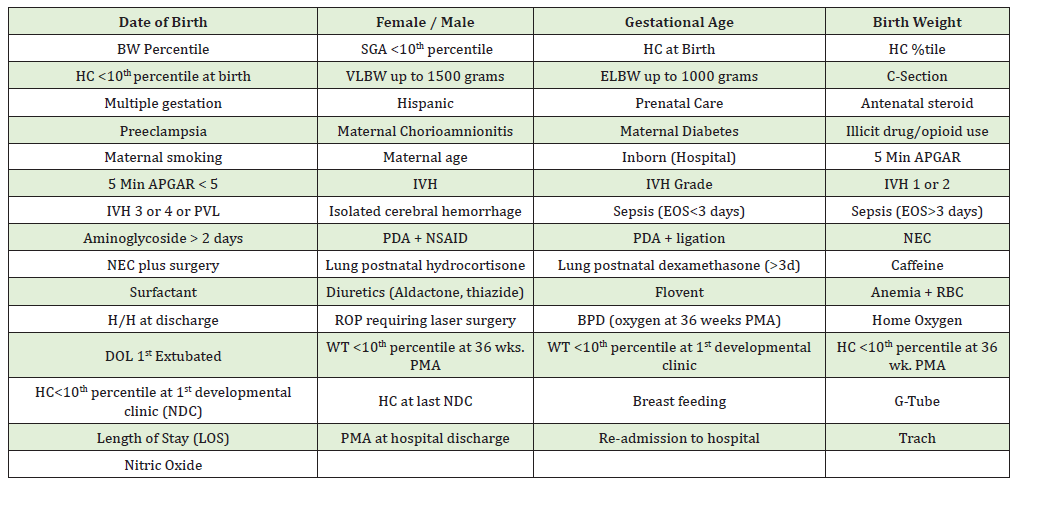
Statistical Analysis
A Pearson Chi-Square test was used to analyze the significance between “Yes/No” Variables (Appendix B) against the 8 to 12-month, 15 to 18 month and 24-to-36-month BSID-III Scaled Scores for Cognitive Delay, Receptive and Expressive Language Delay and Fine and Gross Motor Delay.
Appendix B:“YES” _ “NO” Variables.
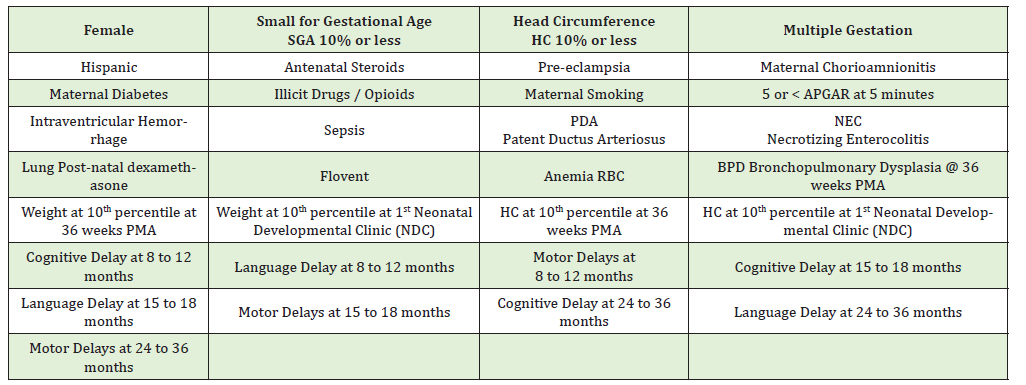
In a Mixed Model Analysis, the different measures of the BSIDIII were collapsed into one variable to use as a dependent variable that measures how well the infants are faring. The first analysis used the non-composite variables. Along with the longitudinal variable of the three time periods, a “scale” variable was created to keep track of the different scales of the Bayley. The base model of this analysis has time, scale, and their interactions as fixed effects. Then each of the other covariates were added one at a time to the base model along with their interactions with scale and time. This created a series of models to help filter out the variables that do not have a significant effect on Bayley scores, either directly or as an interaction with the other two variables. “Type III Sums of Squares Tests of Fixed Effects” were used to determine significance. All statistical analyses were carried out with SPSS 28, [31] commonly used statistical analytic software.
Results
Chi-Square Analysis
The Pearson Chi-Square test did reveal a statistically significant relationship between some of the 60 independent variables at three different time periods (8 to 12 months, 15 to 18 months, 24 to 36 months) when evaluating the Cognitive, Receptive and Expressive Language, Fine and Gross Motor Developmental Domains of the BSID-III.
Table 1:Delay in Milestones at 8 to 12 Months Corrected Age.
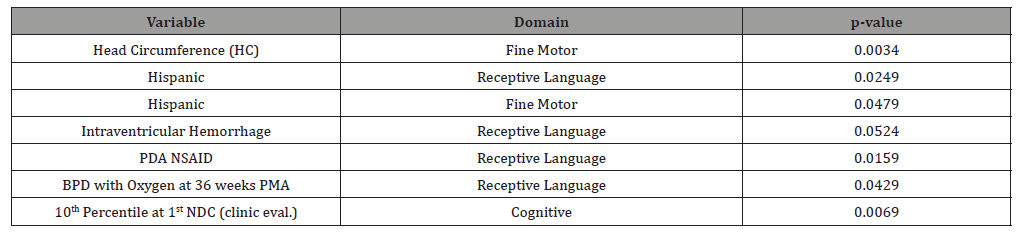
During the 8 to 12 month evaluation period, relationships were found at the p <0.01 significance between Head Circumference and Fine Motor and 10th percentile for weight at the first neonatal developmental follow up clinic and the Cognitive domain. Relationships were found at the p <0.05 significance between receptive language and IVH, PDA NSAID, and BPD with oxygen at 36 weeks PMA.
Table 2:Delay in Milestones at 15 to 18 Months Corrected Age.
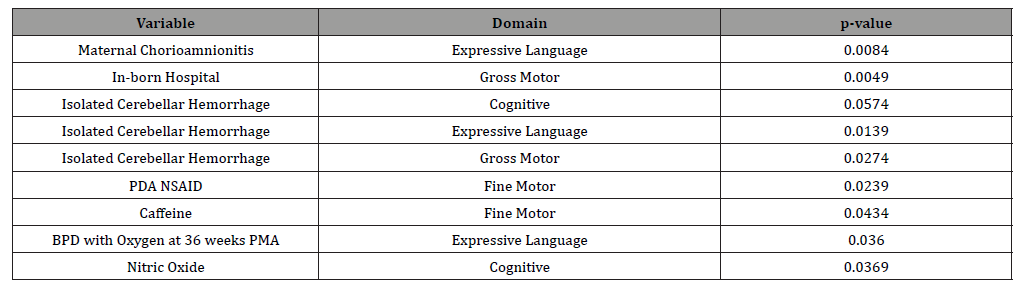
At the 15 to 18 month evaluation period, Nitric Oxide and Isolated Cerebellar Hemorrhage had a significant impact on Cognitive development with correlations found at the p <0.05 level.
Maternal Chorioamnionitis, Isolated Cerebellar Hemorrhage, and BPD with oxygen at 36 weeks were significant at the p <0.05 level for Expressive Language delay. Isolated Cerebellar Hemorrhage was significant at the p <0.05 level for Gross Motor delays, and PDA with NSAID and caffeine were significant at the p <0.05 level for Fine Motor delays.
Table 3:Delay in milestones at 24 to 36 months.
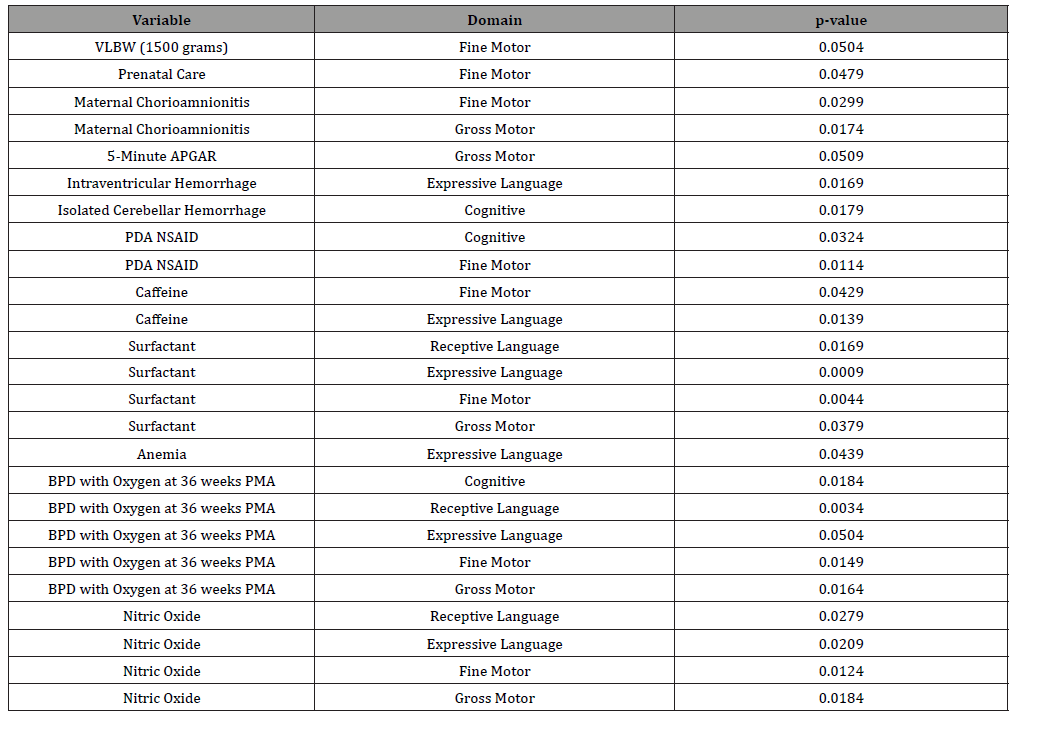
Each child’s age was no longer corrected for prematurity during the 24 to 36 month evaluation period. Delays are minimal during the first two testing age ranges when adjusted for prematurity; however, we see a spike in delays across all domains after 24 months and beyond when age is no longer adjusted for prematurity and true age expectations are in place.
Prenatal Care, Maternal Chorioamnionitis, PDA NSAID, Caffeine, Surfactant, BPD with oxygen at 36 weeks PMA and Nitric Oxide were all significant at the p <0.05 level for Fine Motor delays. Maternal Chorioamnionitis, Surfactant, BPD with oxygen at 36 weeks PMA and Nitric Oxide were all significant at the p <0.05 level for Gross Motor delays. Isolated Cerebellar Hemorrhage, PDA NSAID, and BPD with oxygen at 36 weeks PMA were all significant at the p <0.05 level for Cognitive delays. IVH, Caffeine, and Nitric Oxide were all significant at the p <0.05 level for Expressive Language delays. Additionally, Surfactant, BPD with oxygen at 36 weeks PMA and Nitric Oxide were all significant at the p <0.05 level for Receptive Language delays.
Variables with marginal significance (very low birthweight (VLBW) & Fine Motor; 5-Minute APGAR & Gross Motor; BPD with Oxygen at 36 weeks PMA & Expressive Language) were included in Table 3 so as to highlight important variables that other research has shown to be significant for motor and cognitive delays [1,32]. The data presented in this current research indicate that these premature infants did not “catch up by two-years of age” or develop the skill sets expected for the 24 to 36-month age range.
Mixed Model Analysis
Nine measurements were conducted for each child resulting in 783 data points in this analysis. Attrition did occur; however, it is unknown as to whether this biased the study. Table 4 provides the number of data points for each age range evaluated and each scale that was measured.
Table 4:Data Points for Time and Scale.
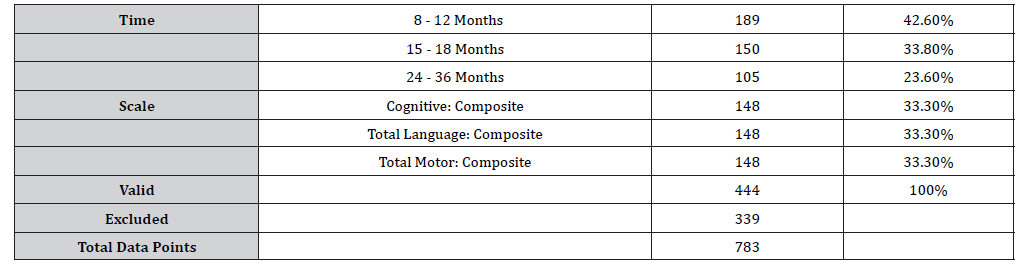
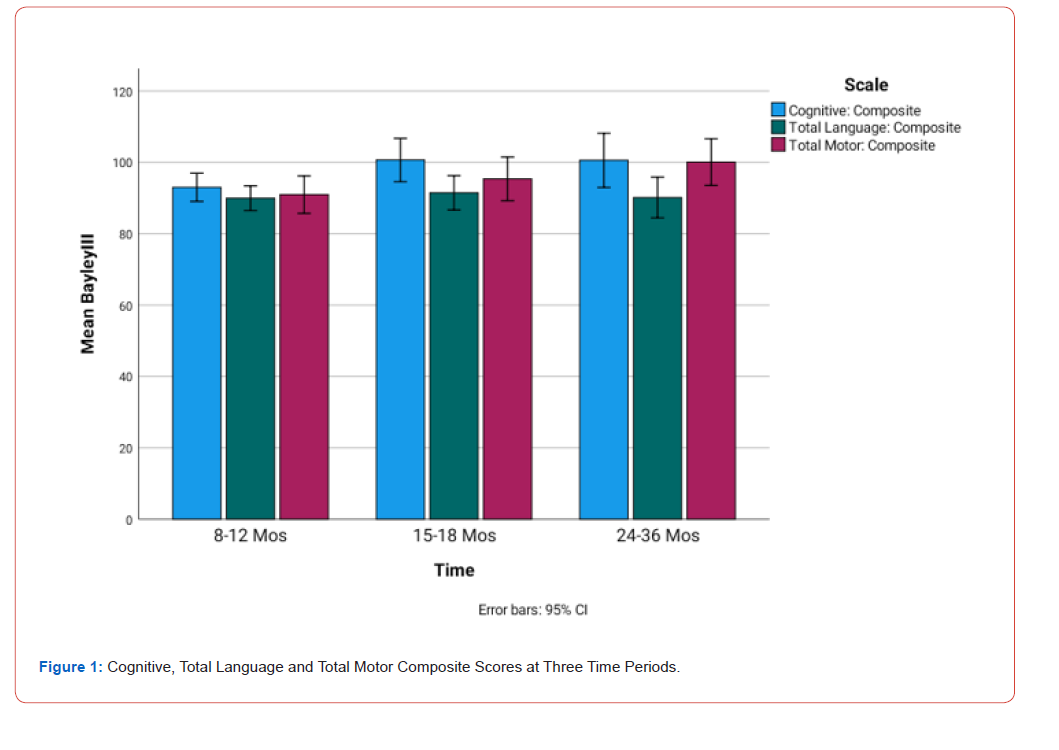
Figure 1 highlights Composite Scores at the 95% Confidence Interval for Cognitive, Total Language and Total Motor developmental domains across all three time periods. During the 8 to 12-month evaluation period, all developmental domains were below the mean. Cognitive skill development showed improvement at the 15 to 18-month evaluation period with Total Language Composite and Total Motor Composite remaining below the mean. In the final evaluation period, only the Total Language Composite remained below the mean with Language Composite scores remaining below the mean for all three time periods.
Type III Tests of Fixed Effects
The Type III Tests of Fixed Effects provides similar outcomes as the Pearson Chi-Square Test. These tests both found significance between time and specific variables. Table 5 highlights these results.
Table 5:Type III Sums of Squares Tests of Fixed Effects: Significance between Time and Variable.
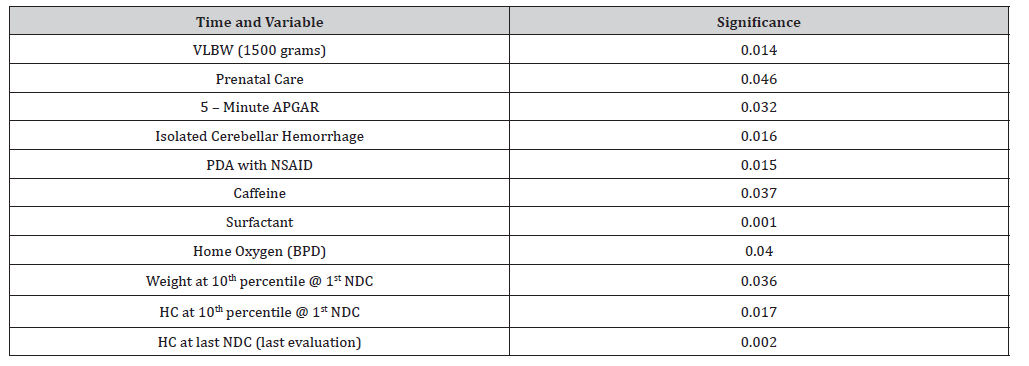
The Pearson Chi-Square test found Maternal Chorioamnionitis to be significant for delays in Expressive Language at 15 to 18 months and from 24 to 36 months. This was not validated by the Type III Tests which looked at variables across all timeframes (8 to 12 months, 15 to 18 months, and 24 to 36 months). Additional medical conditions found to be significant on the Pearson Chi- Square but not on the Type III tests include: IVH, Anemia, Nitric Oxide, and born in hospital.
Discussion
Very Preterm, moderately preterm, and late preterm infants are challenging to study and their outcomes are difficult to generalize due to medical, educational, and environmental complexities and the diversity of these populations throughout childhood. This also holds true for those infants born with hypoxic ischemic encephalopathy. Variables are difficult to control and complexities with fragile infants can compound effects across developmental domains. Major findings for this study were observed in the following areas.
Prenatal Care
According to a study conducted in 2002 by Vintzileos, Ananth, Smulian, Scorza, and Knuppel, [33] preterm birth rates were higher for African American women than white women regardless of prenatal care from 1995 through 1998. The absence of prenatal care increased the risk of a preterm birth 2.8-fold for both African American and white women. Compared with infants born to mothers who received prenatal care, infants whose mothers did not receive prenatal care are three times more likely to have a low birth weight (less than 2500 grams or 5.5 pounds) and five times more likely to die during infancy [33]. Researchers at Yale School of Public Health have found that group prenatal care for expecting mothers reduces the risks for preterm birth and low birth weight. Women with five or more group prenatal care visits had a 68 percent lower risk of having a preterm birth and a 66 percent lower risk of having a low birthweight baby than their peers receiving traditional care [34].
In this retrospective study, a relationship was found at the p <0.05 significance between lack of Prenatal Care and Fine Motor delay during the 24 to 36 month evaluation.
Maternal Chorioamnionitis
Chorioamnionitis is a serious bacterial infection of the placenta and amniotic fluid that causes inflammation for both the mother and the fetus. Chorioamnionitis is a dangerous form of infection-related inflammation that appears to directly cause brain injury among infants born between 22 and 32 weeks gestation. Chorioamnionitis occurs in 25% to 40% of preterm deliveries, but only 4% of fullterm deliveries [35]. Viral, et al discovered that moderate to severe acute histologic chorioamnionitis was associated with increased risk for brain injury and developmental abnormalities by termequivalent age.
In this retrospective study, a relationship was found at the p <0.05 significance between Maternal Chorioamnionitis and Fine and Gross Motor delay during the 24 to 36 month evaluation.
Intraventricular Hemorrhage (IVH)
Intraventricular hemorrhage (IVH) is the most acute complication of a premature birth. Chorioamnionitis, acidosis, birth asphyxia, shock, blood pressure fluctuations and respiratory distress associated with hypercapnia, hypoxemia or mechanical ventilation are common related problems [36]. Gomella, et al conclude that prematurity and respiratory distress syndrome have remained the most closely related clinical conditions to IVH. Longterm major neurologic sequelae of IVH depends primarily on the degree of associated parenchymal injury and any effects of shortterm complications. The incidence of major neurologic sequelae (spastic motor deficits, major cognitive deficits) after minor degrees of hemorrhage is slightly higher than that in infants without hemorrhage and increases to approximately 50% in infants with severe hemorrhage. A higher incidence occurs in infants with IVH complicated by periventricular hemorrhagic infarction or cystic Periventricular Leukomalacia (PVL) or both. The rate of disability rises as gestational age decreases. In the multicenter EPIPAGE study that enrolled 1,954 infants, it was reported that in infants born at <32 weeks’ gestation, cerebral palsy rates with isolated grade I to III IVH increased from 5% in infants born at 31 to 32 weeks to 10% to 15% in those born at 27 to 30 weeks and 33% in those born at 24 to 26 weeks. In a recent study, the mortality rate was 8% for infants with the IVH grade III. Among the survivors, 87% exhibited major motor deficits, and 68% had cognitive function deficits [36].
In this retrospective study, relationships were found at the p <0.05 significance between IVH and Receptive Language skill development during the 8 to 12 month evaluation and Expressive Language delays during the 24 to 36 month evaluation. Contrary to what other studies report, we found no relationship between IVH and cognitive delays in any evaluation period for the population studied.
Isolated Cerebellar Hemorrhage
Gestational age, BW, acidosis, emergency caesarean delivery, and PDA during the first days of life are risk factors for an isolated cerebellar hemorrhage in preterm infants [37-39]. Neurodevelopmental sequelae of neonatal cerebellar hemorrhage are still a subject of debate, however, a review of literature revealed that outcomes after cerebellar hemorrhage and cerebellar infarction indicated an association with behavioral, cognitive, motor, and language impairment. After extensive review of the literature, Hortensius, et al concluded that 43% to 75% of infants with isolated cerebellar hemorrhage were severely delayed in cognition, motor, language and/or behavioral development [37,40]. In the assessment of cognitive disorders in preterm infants, Hammad, Ahmed, Rashwan and Fahem (2018) found that cognitive delays without major motor deficits are the predominant sequela in these infants. They found that the impairments in all developmental domains were more evident as the infants age, but that cognition improved at 12 months [41].
In this retrospective study, relationships were found at the p <0.05 significance between Isolated Cerebellar Hemorrhage in Cognitive Expressive Language and Gross Motor delays during the 15 to 18 month evaluation period but only Cognitive delays at the 24 to 36 month evaluation period. Unlike the results discussed by Hammad et al [41] the cognitive delays continued beyond evaluation periods.
Patent Ductus Arteriosus (PDA) with NSAID
A PDA is a persistent opening between two major blood vessels leading from the heart which allows oxygen-rich blood from the aorta to mix with blood that is low in oxygen in the pulmonary artery. This causes too much blood to flow into the lungs, putting a strain on the heart and increasing blood pressure in the pulmonary arteries. PDA occurs in 40-60% of VLBW infants during the first few weeks of life and more than 60% of extremely low birthweight infants at < 29 weeks GA [42,43]. A study conducted in Seoul Korea found that VLBW infants who underwent a PDA ligation exhibited significantly lower cognitive scores on the BSID-II at 18 to 24 months corrected age compared to those who did not have a ligation [42]. Hamrick and Hansmann (2010) stressed the need for pharmacologic intervention, such as use of an NSAID, over surgical ligation to reduce adverse outcomes in VLBW and ELBW infants [41]. NSAIDs include NeoProfen, and other non-steroidal antiinflammatory medications.
In this retrospective study, we found that PDA with NSAID is significant for cognitive and fine motor delays at 24 to 36 months.
Caffeine
Caffeine is effective in treating apnea of prematurity and intermittent hypoxemia and is helpful in reducing the incidence of chronic lung disease. A number of studies suggest that caffeine intake in preterm infants may have a neuroprotective effect [45- 48]. Prolonged treatment with caffeine has been shown to reduce hypoxemia events in premature infants which can lead to adverse neurodevelopmental outcomes due to severity and duration [49,50]. Generally, neonatal caffeine therapy appears to be safe into middle-school age with no adverse effects on general motor function, intelligence, attention, and behavior [48]. Although studies indicate little to no effect on neurodevelopmental outcomes, this retrospective study found that utilization of Caffeine was significant for fine motor deficits at the 15 to 18 month evaluation and significant for Fine Motor and Expressive Language deficits at the 24 to 36 month evaluation.
Surfactant
Surfactant is a mixture of fat, and proteins made in the lungs that coats the alveoli or air sacs in the lungs where oxygen enters the body. In premature infants, the lack of surfactant can lead to poor lung function which can lead to respiratory distress syndrome (RDS). Treatment with exogenous surfactant has saved the lives of thousands of premature babies in the past few decades. It has been shown to reduce pneumothorax and increase survival rates significantly [51-53]. In a follow-up of a randomized clinical trial of 486 infants at 25 to 28 weeks’ gestation, minimally invasive surfactant therapy did not lead to a reduction in the composite outcome of death or neurodevelopmental disability at 2 years of age [54]. In another randomized clinical trial comparing surfactant delivery via a thin catheter with ongoing respiratory support with continuous positive airway pressure (CPAP) researchers did not detect a difference in incidence of BPD, nor was there a discernible effect on the rate of NDD at 2 years of age [55,56]. Surfactant has been shown to increase survival rates, but the method of administration has not seemed to positively impact NDD outcomes.
In this retrospective study, relationships were found at the p <0.05 significance between Surfactant in Receptive Language, Expressive Language (Total Language) and Fine and Gross Motor (Total Motor) delays during the 24 to 36 month evaluation period.
Anemia
Anemia of prematurity is caused by early birth occurring prior to the completion of placental iron transport and fetal erythropoiesis (stem cell becomes a mature red blood cell), by blood loss taken for testing by low plasma levels, and by disorders causing red blood cell (RBC) losses due to bleeding and/or rupture or destruction of red blood cells (hemolysis) [57]. Preterm infants weighing 1000 grams or less and <29 weeks gestation will nearly all require RBC transfusions shortly after birth. In 2010, Strauss reported a controversy, and outcomes related to the prescribing of RBC transfusions using restrictive guidelines (i.e., permitting relatively low hematocrit values before RBC transfusion) versus liberal guidelines (i.e., relatively high pretransfusion hematocrit values). Increases in apnea, severe intraventricular bleeding, and PVL were found in the infants who were transfused using the restrictive guidelines [58]. In a study conducted by Kirpalani, White and Andersen (2006), no differences were found in serious outcomes between infants receiving transfusions based on liberal guidelines versus restrictive guidelines; however, serious outcomes were high in both groups in the Kirpalani study due to the extreme prematurity of the infants [59].
In this retrospective study, a relationship was found at the p <0.05 significance between Anemia and Expressive Language skill development at the 24 to 36 month evaluation. No relationship of significance was found during the 8 to 12 month or 15 to 18 month evaluation periods.
Bronchopulmonary Dysplasia (BPD)
The diagnostic criteria for a diagnosis of BPD is based on an infants’ supplemental oxygen needs for at least 28 days after birth. The severity of BPD is then graded according to the respiratory support needed at 36 weeks PMA and a GA < 32 weeks or at 56 days after birth. Grades of severity are as follows: 1) mild, breathing room air; 2) moderate, FiO2 <0.3; and 3) severe, FiO2 ≥ 0.3 with mechanical ventilation [60,61]. A retrospective study conducted at the Division of Neonatology, the Second Xiangya Hospital, Central South University in China investigated bronchopulmonary dysplasia (BPD) and its treatment with dexamethasone (DEX) in premature infants with BW <1500 g. In this study from China, researchers identified neonatal respiratory distress syndrome, neonatal pneumonia, positive sputum culture, pulmonary hemorrhage, and respiratory failure as risk factors for BPD. A multivariate logistic regression revealed that neonatal respiratory distress syndrome and GA were independent risk factors for BPD. Based on the results of a two-week treatment with DEX of infants diagnosed with BPD, these researchers suggest that low GA was the most important risk factor for BPD. DEX reduced oxygen dependency but decreased weight gain [32]. Most researchers now agree with the hypothesis that BPD is a developmental disorder that results from interference with the process of pulmonary maturation, leading to developmental maturation abnormalities of the structures and functions of the distal lung parenchyma [32,62-64]. Consequently, lung injuries in BPD are attributed to barotrauma, hyperoxia, stress and pulmonary immaturity, infections, intrauterine hypoxia, and PDA [65-67].
In our retrospective study, relationships were found at the p <0.05 significance between Receptive Language and BPD with oxygen at 36 weeks PMA during the 8 to 12 month evaluation period, and, with Expressive Language during the 15 to 18 month evaluation. During the 24 to 36 month evaluation period, relationships were found at the p<0.05 significance between Cognitive, Receptive Language, Fine Motor and Gross Motor. No relationship was found between BPD and GA during any or the three evaluation periods.
Nitric Oxide
Nitric oxide (iNO) is used to treat newborns with persistent pulmonary hypertension (PPHN). PPHN is a dangerous condition that occurs when the newborn’s blood vessels to the lungs do not open after birth. When diagnosed, infants tend to have significant hypoxemia and are in respiratory failure. Meconium aspiration, respiratory distress syndrome, pneumonia and sepsis can contribute to PPHN in newborns. Research shows that nitric oxide acutely improves oxygenation and decreases the need for ECMO (extracorporeal membrane oxygenation) support in newborns with PPHN [68]. Feng, Wu, Xu, and Wu (2024) conducted a study to assess the efficacy of iNO treatment in preterm infants ≤ 34 weeks gestation. Their research included a total of 17 studies involving 4,080 neonates and 7 follow-up studies. Feng et al found that in neonates, iNO treatment reduced the incidence of BPD, decreasing mortalities without increasing the risk of short-term (e.g., IVH or PVL) or long-term (e.g., neurodevelopmental impairment or cerebral palsy) neurological outcomes. Meta-analyses showed that iNO did not lead to poor neurodevelopmental outcomes including a Bayley mental developmental index (MDI) <70, cerebral palsy or other neurodevelopmental impairment [69].
In our retrospective study, relationships were found at the p <0.05 significance between Nitric Oxide (iNO) Cognitive delays at the 15 to 18 month evaluation and Receptive and Expressive Language delays and Fine and Gross Motor delays at the 24 to 36 month evaluation.
Conclusion
Care of preterm infants managed in any NICU should include evidence-based clinical practices to prevent the common forms of brain injury and mitigate the resulting adverse neurodevelopmental outcomes. Motor, language, cognitive and social and emotional development are interrelated and hinge upon the basic senses of vision, hearing, and touch. Interventions such as multidisciplinary or interdisciplinary developmental follow-up and therapies should be implemented beyond three years of age. Parents of high-risk infants should receive education on developmental stages and ways to enhance developmental progress and on the potential challenges that the child might experience while their infant receives care in the NICU. Liang, Rogers, & Smyser (2019) stress that prior to discharge, infants at risk for impaired neurodevelopmental outcome should be identified so that indicated supportive services and early intervention programs can be initiated and caregivers can receive appropriate education [5].
Acknowledgement
Neonatologists and Pediatricians who cared for these infants in the NICU and evaluated them in the Neonatal Developmental Follow-Up Clinic from 2011 through 2019 include: Drs. Alan Bedrick, Mohammed Bader, Ranjit Kylathu, Joseph Livingston, Karen Davenport, and Graciela Wilcox. NNP, Cynthia Garcia and Neonatal Physical Therapists Nichole Lensing and Mary Beth Hoffmeyer along with Authors Jacob and McGrath conducted diagnostic evaluations, provided parent education, referral, and support services.
Conflicts of Interest
The authors declare no conflicts of interest regarding the publication of this paper.
References
- Pascal A, Govaert P, Oostra A, Naulaers G, Ortibus E, et al. (2018) Neurodevelopmental Outcome in Very Preterm and Very-Low-Birthweight Infants Born over the Past Decade: A Meta-Analytic Review. Developmental Medi- cine & Child Neurology 60(4): 342-355.
- Blencowe H, Cousens S, Chou D, Oestergaard M, Say L, et al. (2013) Born Too Soon: The Global Epidemiology of 15 Million Preterm Births. Reproductive Health 10: S2.
- Harrison MS, Goldenberg RL (2016) Global Burden of Prematurity. Seminars in Fetal and Neonatal Medicine 21(2): 74-79.
- McGrath ER, Borgstrom M, Ramirez Moreno EA, Jacob J (2023) Neurodevelopmental Outcomes of Extremely Premature Infants with Extremely Low and Very Low Birth Weight at Three Years of Age: A Retrospective Study. Open Journal of Pediatrics 13: 333-346.
- Liang SN, Rogers CE, Smyser CD (2019) Long-term neurodevelopmental impairment. Jain L, Suresh GK (Eds.), Clinical Guidelines in Neonatology. McGraw-Hill Education.
- Bhutta AT, Cleves MA, Casey PH, Mary M Cradock, K J S Anand, et al. (2002) Cognitive and behavioral outcomes of school-aged children who were born preterm: a meta-analysis. JAMA 288(6): 728-737.
- Arpino C, Compagnone E, Montanaro ML, Denise Cacciatore, Angela De Luca, et al. (2010) Preterm birth, and neurodevelopmental outcome: a review. Childs Nerv Syst 26(9): 1139-1149.
- Johnson S, Evans TA, Draper ES, David J Field, Bradley N Manktelow, et al. (2015) Neurodevelopmental outcomes following late and moderate prematurity: a population-based cohort study. Arch Dis Child Fetal Neonatal Ed 100(4): F301-308.
- McGowan JE, Alderdice FA, Holmes VA, Linda Johnston (2011) Early childhood development in late-preterm infants: a systematic review. Pediatrics 127(6): 1111-1124.
- Sansavini A, Guarini A, Justice LM, Silvia Savini, Serena Broccoli, et al. (2010) Does preterm birth increase a child’s risk for language impairment? Early Hum Devel 86(12): 765-772.
- Futagi Y, Toribe Y, Ogawa K, Yasuhiro Suzuki (2006) Neurodevelopmental outcome in children with intraventricular hemorrhage. Pediatr Neurol 34(3): 219-224.
- Hack M (2009) Adult outcomes of preterm children. Journal of Developmental & Behavioral Pediatrics 30(5): 460-470.
- Kerstjens JM, de Winter AF, Bocca Tjeertes IF, Elisabeth M J ten Vergert, Sijmen A Reijneveld, et al. (2011) Developmental delay in moderately preterm-born children at school entry. J Pediatr 159(1): 92-98.
- Limperopoulos C, Bassan H, Gauvreau K, Richard L Robertson Jr, Nancy R Sullivan, et al. (2007) Does cerebellar injury in premature infants contribute to the high prevalence of long-term cognitive, learning, and behavioral disability in survivors? Pediatrics 120(3): 584-593.
- Petrini JR, Dias T, McCormick MC, Maria L Massolo, Nancy S Green, et al. (2009) Increased risk of adverse neurological development for late preterm infants. J Pediatr 154(2): 169-176.
- Stephens BE, Vohr (2009) Neurodevelopmental outcome of the premature infant. Pediatr Clin North Am 56: 631-646.
- Woodward LJ, Moor S, Hood KM, P R Champion, S Foster-Cohen, et al. (2009) Very preterm children show impairments across multiple neurodevelopmental domains by age 4 years. Arch Dis Child Fetal Neonatal Ed 94(5): F339-344.
- Brady JM, DeMauro SB (2018) Developmental outcomes of high-risk infants. Kline M.W.(Ed.), Rudolph's Pediatrics, 23e. McGraw-Hill Education.
- Martin JA, Osterman MJK (2024) Natl Vital Stat Rep 73(1): 1-11.
- Gopagondanahalli KR, Li J, Fahey MC, Hunt RW, Jenkin G, et al. (2016) Preterm Hypoxic-Ischemic Encephalopathy. Front Pediatr 4: 114.
- Moore T, Hennessy EM, Myles J, Johnson SJ, Draper ES, et al. (2012) Neurological and developmental outcome in extremely preterm children born in England in 1995 and 2006: the EPICure studies. BMJ 345: e7961.
- Hintz SR, Kendrick DE, Vohr BR, Poole WK, Higgins RD, et al. (2005) National Institute of Child Health, and Human Development Neonatal Research Network. Changes in neurodevelopmental outcomes at 18 to 22 months’ corrected age among infants of less than 25 weeks’ gestational age born in 1993-1999. Pediatrics 115(6): 1645-1651.
- Mikkola K, Ritari N, Tommiska V, Salokorpi T, Lehtonen L, et al. (2005) Neurodevelopmental outcome at 5 years of age of a national cohort of extremely low birth weight infants who were born in 1996-1997. Pediatrics 116(6): 1391-1400.
- Allen MC (2008) Neurodevelopmental outcomes of preterm infants. Curr Opin Neurol 21(2): 123-128.
- Woodward LJ, Edgin JO, Thompson D, Inder TE (2005) Object working memory deficits predicted by early brain injury and development in the preterm infant. Brain 128(11): 2578-2587.
- Bayless S, Stevenson J (2007) Executive functions in school-age children born very prematurely. Early Hum Dev 83(4): 247-254.
- Rees S, Inder T (2005) Fetal and neonatal origins of altered brain development. Early Human Development 81(9): 753-776.
- Chalak LF, Rollins N, Morriss MC, Brion LP, Heyne R, et al. (2012) Perinatal acidosis and hypoxic-ischemic encephalopathy in preterm infants of 33 to 35 weeks’ gestation. J Pediatr 160(3): 388-394.
- Bayley N (2006) Bayley Scales of Infant and Toddler Development. 3rd Edition. Harcourt Assessment Inc, San Antonio, TX.
- Bayley N (2006) Technical Manual: The Bayley Scales of Infant and Toddler Development-Third Edition. The Psychological Corporation, San Antonio, TX.
- SPSS, IBM Corp. Released 2021. IBM SPSS Statistics for Windows, Version 28.0. Armonk, NY: IBM Corp.
- Shen L, Bo T, Luo SL, Zhang RI, Li J, et al. (2016) Bronchopulmonary Dysplasia in Premature Infants with Very Low Birth Weight: A Single Centre Retrospective Study in China. Open Journal of Pediatrics 6: 295-307.
- Vintzileos AM, Ananth CV, Smulian JC, Scorza WE, Knuppel RA, et al. (2002) The impact of prenatal care in the United States on preterm births in the presence and absence of antenatal high-risk conditions. Am J Obstet Gynecol 187(5): 1254-7.
- Cunningham SD, Lewis JB, Shebl FM, Boyd LM, Robinson MA, et al. (2019) Group Prenatal Care Reduces Risk of Preterm Birth and Low Birth Weight: A Matched Cohort Study. Journal of Women’s Health 28(1): 17-22.
- Viral G J, Kline JE, He l, Kline-Fath BM, Altaye M, et al. (2022) Acute histologic chorioamnionitis independently and directly increases the risk for brain abnormalities seen on magnetic resonance imaging in very preterm infants. American Journal of Obstetrics and Gynecology 227(4): e1-623.e13.
- Gomella T, Eyal FG, Bany-Mohammed F (2024) (Eds.) Gomella's Neonatology: Management, Procedures, On-Call Problems, Diseases, and Drugs, 8e. McGraw-Hill Education.
- Hortensius LM, Dijkshoorn ABC, Ecury-Goosen GM, et al. (2018) Neurodevelopmental Consequences of Preterm Isolated Cerebellar Hemorrhage: A Systematic Review. Pediatrics 142(5): e20180609.
- Limperopoulos C, Benson CB, Bassan H, Donald N Disalvo, Daniel D Kinnamon, et al. (2005) Cerebellar hemorrhage in the preterm infant: ultrasonographic findings and risk factors. Pediatrics 116(3): 717-724.
- Dyet LE, Kennea N, Counsell SJ, Elia F Maalouf, Morenike Ajayi-Obe, et al. (2006) Natural history of brain lesions in extremely preterm infants studied with serial magnetic resonance imaging from birth and neurodevelopmental assessment. Pediatrics 118(2): 536-548.
- Brossard-Racine M, du Plessis AJ, Limperopoulos C (2015) Developmental cerebellar cognitive affective syndrome in ex-preterm survivors following cerebellar injury. Cerebellum 14(2): 151-164.
- Hammad EM, Ahmed EA, Rashwan IR, Fahem MG. Assessment of Some Cognitive Disorders in Preterm Infants. Res Pediatr Neonatology 1(5): 000522.018.
- Kim S, Kim SY, Shin JA (2024) Risk factors for PDA ligation and neurodevelopmental outcomes at corrected 18–24 months in very low birth weight infants. BMC Pediatr 24(1): 376.
- Costeloe K, Hennessy E (2000) Gibson AT. Pediatrics 106: 659-671
- Hamrick EG, Hansmann G (2010) Patent Ductus Arteriosus of the Preterm Infant. Pediatrics 125: 5.
- Back SA, Craig A, Luo NL, Jennifer Ren, Ravi Shankar Akundi, et al. (2006) Protective effects of caffeine on chronic hypoxia-induced perinatal white matter injury. Ann Neurol 60(6): 696-705.
- Connolly S, Kingsbury TJ (2010) Caffeine modulates CREB-dependent gene expression in developing cortical neurons. Biochem Biophys Res Commun 397(2): 152-156.
- Silva CG, Métin C, Fazeli W, Nuno J Machado, Sanja Darmopil, et al. (2013) Adenosine receptor antagonists including caffeine alter fetal brain development in mice. Sci Transl Med 5(197):197ra104.
- Moschino L, Zivanovic S, Hartley C, Trevisanuto D, Baraldi E, et al. (2020) Caffeine in preterm infants: where are we in 2020? ERJ Open Res 6(1): 00330-2019.
- Rhein LM, Dobson NR, Darnall RA, Michael J Corwin, Tim C Heeren, et al. (2014) Effects of caffeine on intermittent hypoxia in infants born prematurely: a randomized clinical trial. JAMA Pediatr 168(3): 250-257.
- Poets CF, Roberts RS, Schmidt B, Robin K Whyte 4, Elizabeth V Asztalos, et al. (2015) Association between intermittent hypoxemia or bradycardia and late death or disability in extremely preterm infants. JAMA 314(6): 595-603.
- Polin RA, Carlo WA, Committee on Fetus and Newborn (2014) American Academy of Pediatrics Surfactant replacement therapy for preterm and term neonates with respiratory distress. Pediatrics 133(1):156-63.
- Nouraeyan N, Lambrinakos-Raymond A, Leone M, Sant'Anna G (2014) Surfactant administration in neonates: A review of delivery methods. Can J Respir Ther 50(3): 91-5.
- Ramanathan R (2021) Surfactant Treatment—A National, Population-Based Study of Adherence to Best Practice, Off-Label Use, and Associations With Outcomes. JAMA Netw Open 4(5): e217848.
- Dargaville PA, Kamlin COF, Orsini F, Wang X, De Paoli AG, et al. (2023) Two-Year Outcomes After Minimally Invasive Surfactant Therapy in Preterm Infants: Follow-Up of the OPTIMIST-A Randomized Clinical Trial. JAMA 330(11): 1054-1063.
- Göpel W, Kribs A, Ziegler A, Reinhard Laux, Thomas Hoehn, et al. (2011) German Neonatal Network. Avoidance of mechanical ventilation by surfactant treatment of spontaneously breathing preterm infants (AMV): an open label, randomised, controlled trial. Lancet 378(9803): 1627-1634.
- Herting E, Kribs A, Härtel C, Axel von der Wense, Ursula Weller, et al. (2020) Two-year outcome data suggest that less invasive surfactant administration (LISA) is safe: results from the follow-up of the randomized controlled AMV (avoid mechanical ventilation) study. Eur J Pediatr 179(8): 1309-1313.
- Strauss RG (2010) Anaemia of prematurity: pathophysiology and treatment. Blood Rev 24(6):221-5.
- Bell EF, Strauss RG, Widness JA, Mahoney LT, Mock DM, et al. (2005) Randomized trial of liberal versus restrictive guidelines for red blood cell transfusions in preterm infants. Pediatrics 115(6): 1685-1691.
- Kirpalani H, Whyte RK, Andersen C, Elizabeth V Asztalos, Nancy Heddle, et al. (2006) The Premature Infants in Need of Transfusion (PINT) study: a randomized, controlled trial of a restrictive (low) versus liberal (high) transfusion threshold for extremely low birth weight infants. J Pediatr 149(3): 301-307.
- McGrath ER (2017) Neurodevelopmental Outcomes of a 25-Week Premature Infant at Three Years of Age. Pediatric Dimensions 2: 1-4.
- Joeb AH, Bancalari E (2001) Bronchopulmonary Dysplasia. American Journal of Respiratory Critical Care Medicine 163(7): 1723-1729.
- Fiaturi N, Castellot JJ, Nielsen HC (2014) Neuregulin-ErbB4 Signaling in the Developing Lung Alveolus: A Brief Review. Journal of Cell Communication and Signaling 8(2): 105-111.
- Shaw G M, O’Brodovich, HM (2013) Progress in Understanding the Genetics of Bronchopulmonary Dysplasia. Seminars in Perinatology 37(2): 85-93.
- Tang JR, Karumanchi SA, Seedorf G, Markham N, Abman SH, et al. (2012) Excess Soluble Vascular Endothelial Growth Factor Receptor-1 in Amniotic Fluid Impairs Lung Growth in Rats: Linking Preeclampsia with Bronchopulmonary Dysplasia. American Journal of Physiology-Lung Cellular and Molecular Physiology 302(1): L36-L46.
- Kallapur SG, Kramer BW, Jobe AH (2013) Ureaplasma and BPD. Seminars in Perinatology 37: 94-101.
- Been JV, Debeer A, van Iwaarden JF, Kloosterboer N, Passos VL, et al. (2010) Early Alterations of Growth Factor Patterns in Bronchoalveolar Lavage Fluid from Preterm Infants Developing Bronchopulmonary Dysplasia. Pediatric Research 67(1): 83-89.
- Clyman RI (2013) The Role of Patent Ductus Arteriosus and its Treatment in the Development of Bronchopulmonary Dysplasia. Seminars in Perinatology 37(2): 102-107.
- Nandula PS, Shah SD (2024) Persistent Pulmonary Hypertension of the Newborn. [Updated 2023 Jul 31]. In: StatPearls Publishing.
- Feng Z, Wu X, Xu X, Cui Q, Wu F, et al. (2024) Efficacy of inhaled nitric oxide in preterm infants ≤ 34 weeks: a systematic review and meta-analysis of randomized controlled trials. Frontiers in Pharmacology 14: 1268795.
-
Eileen R McGrath*, Mark Borgstrom, Cody Melcher and Jennie Jacob. Neurodevelopmental Outcomes of Very, Moderately and Late Preterm Infants and Infants with Hypoxic Ischemic Encephalopathy between 8 to 36 Months of Age: A Retrospective Study. Glob J of Ped & Neonatol Car. 5(2): 2024. GJPNC.MS.ID.000607.
Very Preterm, Moderately Preterm, Late Preterm, Hypoxic Ischemic Encephalopathy, Neurodevelopmental Outcomes, Heterogeneous, Clinical manifestations, Diagnostic
-

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.






