 Research article
Research article
Evidence That No One Needs a Tracheotomy for Only Being Too Weak to Breathe
John R Bach1*, Antonio Huerta Armijo2 and Julia Dorta Díez de la Lastra3
1 Professor of Physical Medicine and Rehabilitation, Professor of Neurology, Department of Physical Medicine and Rehabilitation, Rutgers University – New Jersey Medical School, Medical Director of the Center for Ventilator Management Alternatives, University Hospital of Newark, New Jersey, USA, Grupo Iberoamericano de Cuidados Respiratorios en Enfermedades Neuromusculares (GICREN), ORCID: 0000-0002-4964-665X.
2 Unidad de hospitalización domiciliaria NANEAS, Hospital clínico San Borja Arriarán, Santiago, Chile, Grupo Iberoamericano de cuidados respiratorios en enfermedades neuromusculares (GICREN), ORCID: 0000-0002-4308-6161
3 Servicio de Neumología, Hospital Universitario del Henares, Madrid, España, Grupo Iberoamericano de cuidados respiratorios en enfermedades neuromusculares (GICREN), ORCID: 0000-0001-5802-7167
John R Bach, Department of Physical Medicine and Rehabilitation, Rutgers University – New Jersey Medical School, Medical Director of the Center for Ventilator Management Alternatives, University Hospital of Newark, New Jersey, USA, Grupo Iberoamericano de Cuidados Respiratorios en Enfermedades Neuromusculares (GICREN), ORCID: 0000-0002-4964- 665X.
Received Date: April 30, 2024; Published Date: May 10, 2024
Abstract
Introduction: In 1957 61 Iron Lung dependent pediatric post-poliomyelitis survivors (PPSs), and 117 others, left Iron Lungs for sleep plus
daytime, eventually continuous, dependence on noninvasive intermittent positive pressure ventilatory support (CNVS) via oral interfaces. The 61
had little to no vital capacity (VC) and depended on CNVS with no autonomous ability to breathe for 1560 patient years through 1993. Besides the
PPSs, 228 others with ventilatory pump failure also left Iron Lungs to depend on NVS/CNVS for 2350 patient-years through 1993. Fifty-two of the 61
PPSs, including 8 with no measurable VC, continued to be followed after 1993. Intercurrent intubations were managed by extubation to CNVS and
mechanical in-exsufflation (MIE).
Methods: Use of NVS via oral, nasal, and oronasal interfaces, abdominal pressure ventilator, MIE, and protocol maintaining normal oxyhemoglobin
saturation (O2sat) using NVS and MIE. NVS/CNVS utilization and survival outcomes are reviewed.
Results: The 52 PPSs continued to be dependent on CNVS for at least 529 additional patient-years after 1993 in addition to the 1560 previously.
None were known to undergo tracheotomies despite 16 being intubated for acute respiratory failure, ventilator unweanable, but extubated back to
CNVS. At least four PPSs have been CNVS dependent for 70 years. Thirty-two presented with but were decannulated of tracheostomy tubes.
Conclusion: The facts that at least 61 post-polio patients had no measurable VC or ventilator free breathing ability for over 30 years other
than by glossopharyngeal (“frog”) breathing, that the same center has 41 dependent patients with spinal muscular atrophy (SMA) type 1, including
19 between 20 and 30 years of age, all with 0 ml of VC and only residual eye movements, and that 385 with Duchenne muscular dystrophy (DMD)
have been described to be CNVS dependent, now for up to 30 years and all without tracheostomy tubes, implies that no one apparently needs a
tracheotomy for only being too weak to breathe.
Introduction
When the ventilatory pump fails because of severely weakened or overworked respiratory muscles and ineffective cough flows, patients are susceptible to acute on chronic respiratory failure. This typically occurs for hypercapnic patients during respiratory tract infections when oxygen is administrated instead of up to continuous noninvasive intermittent positive pressure ventilatory support (CNVS) and mechanical insufflation exsufflation (MIE) used to clear the airways [1,2]. Noninvasive ventilation, or “NIV,” which has become synonymous with continuous positive airway pressure (CPAP) or bi-level PAP, and supplemental oxygen typically do not prevent episodes of CO2 narcosis, intubation, then urging for tracheotomy for mechanical ventilation (TMV) for those remaining unweanable from ventilatory support when intubated [1-3]. The rationale for urging tracheotomy is often to avoid laryngeal damage from prolonged intubation. However, about 10% of intubated patients with neuromuscular conditions require gastrostomy tubes for more than 2 months whether intubated longer than or shorter than 3 weeks and no prolonged untoward effects on speech have been reported [4]. We explore definitive noninvasive management for pure ventilatory pump failure.
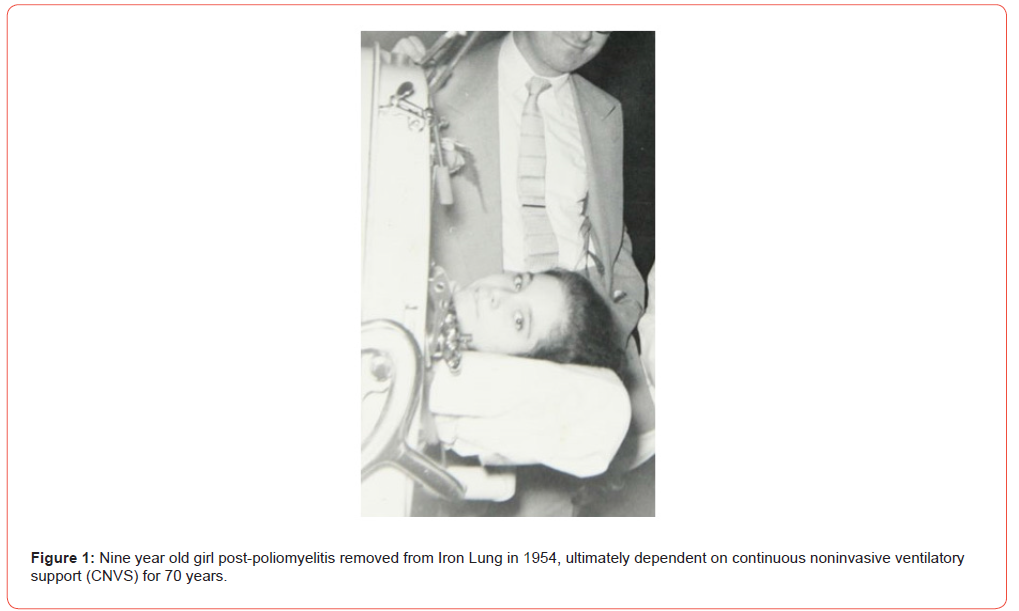
Home mechanical ventilation became possible since electricity became widely available around 1930. However, the predominant body ventilator available from 1930 through 1952 was the 400 kg Iron Lung. Its absence in Denmark in 1952 led to tracheostomies being placed for TMV. This spread to the United States and permitted polio patients to leave Iron Lungs for wheelchairs. Then, in 1953, Dr. John Affeldt described NVS via TygonTM “straws” (Saint-Gobain Performance Plastics, Wayne, N.J.), scuba, and other mouthpiece/ oral interfaces, instead of tracheostomies in Rancho Los Amigos Hospital in Los Angeles [5]. Rancho was one of 16 rehabilitation centers for Iron Lung users in the United States [6]. In 1957 Dr. Augusta Alba switched almost 200 patients post-poliomyelitis (PPSs) and patients with muscular dystrophies, high level spinal cord injuries, and other causes of ventilatory pump failure, from Iron Lungs to mouthpiece CNVS (Figure 1) [7]. Ultimately, lip cover phalanges (LipsealsTM, Life Care Inc., Colorado) were used for support during sleep, for some, now, for 70 years (Figure 2).

Tracheostomy tubes, on the other hand, typically increase ventilator dependence [1,2,5,7]. Many not using any ventilatory assistance before hospitalization for acute on chronic ventilatory failure remain continuously dependent on (C)TMV until they are decannulated [8], at which point they wean back to their pre-hospitalization regimens of ventilator use, typically sleep mouthpiece/nasal NVS. In 1955 it was pointed out that “every knows” that an Iron Lung user with 500 ml of VC can breathe all day on their own but once trached can no longer breathe at all [5]. The tubes and TMV directly cause the deaths of the 50 to 90% of ventilatory pump failure patients dependent on TMV [1,7,9-11]. We present the use of up to CNVS, instead of CTMV, for long term ventilatory support to avoid the morbidity and mortality caused by the latter.
Methods
The noninvasive interventions include: NVS administered at 650 to 1800 ml volumes or pressures of 17 or more cm H2O via mouthpieces, nasal, and/or oronasal interfaces, intermittent abdominal pressure ventilators (IAPVs) that deliver air under the user’s clothing to compress the abdomen and move the diaphragm to ventilate the lungs, MIE used at 50 to 60 cm H2O pressures via mouthpieces and oronasal interfaces and at 60 to 70 cm H2O via airway tubes, and an oximetry feedback protocol to maintain, or return, oxyhemoglobin saturation (O2sat) over 94%. This is used to prevent respiratory infections turning into pneumonias and for extubating and decannulating ventilator unweanable patients.
Results
Of the patients leaving body ventilators for NVS/CNVS at Goldwater Memorial Hospital in 1957 [6], 101 pediatric PPSs used mouthpiece NVS for at least 8 hours per night with 61 requiring CNVS, that is, over 20 hours per day of NVS. These 61 PPSs had been CNVS dependent for 1560 patient-years, 26 years each, up until 1993 [7]. At least 52 of them continued CNVS for at least 529 additional patient-years until death or being lost to follow up. None are known to have undergone tracheostomies despite 16 in acute respiratory failure being extubated back to CNVS and MIE despite being ventilator unweanable. At least four of these have now been CNVS dependent for 70 years.
Another 52 PPSs who had no access to MIE underwent tracheotomies before 1993 during episodes of acute respiratory failure. Fifty to 90% of these patients died directly from TMV complications within 4 years [7]. On the other hand, all 16 PPS CNVS users who got intubated but refused tracheostomies to be transferred to our critical care units were successfully extubated back to CNVS and MIE despite VCs as low as unmeasurable. None of our PPSs with access to MIE have undergone tracheotomy since MIE came on the market in February 1993.
Since 1993, 16 of the 52 PPS CNVS users died but all from clearly non-pulmonary causes and 27 were lost to follow-up. Thirty-two additional PPSs who presented dependent on CTMV were decannulated to CNVS and MIE prior to 1993 and 5 more subsequently [7].
Discussion
In 2010, 19 centers from 16 countries presented 700 DMD pediatric NVS users, 385 of whom dependent on CNVS for a mean 5.4±1.6 years after 4 years of sleep-only NVS [12], as an alternative to tracheostomy. Ventilator dependent patients with DMD had been managed by TMV at a Japanese center until 1991 and they eventually died at the mean age of 29.1 years. Subsequently, all with DMD were managed only by NVS/CNVS and MIE and 88 had a 50% survival to age 39.6 years of age (p<0.001) [13]. Today, the same Japanese center has 98 DMD patients from 9 to 53 years of age with 6 over 50 years of age and using CNVS for over 25 years (verbal communication, Dr. Yuka Ishikawa). While most if not almost all such patients who undergo tracheostomies also undergo gastrostomies, since the trach tubes make it more difficult to swallow food, only 8% of the Japanese patients required them. Indeed, none of the patients from Japanese and Portuguese centers, with over 200 DMD CNVS users, have undergone tracheostomies with all who required intercurrent intubations being extubated to CNVS and MIE [14].
Considering the U.S. center, with one exception, all 39 DMD CNVS users who needed to be intubated were successfully extubated back to CNVS and MIE [1,2]. One with DMD underwent tracheotomy for severe bronchiectasis. No deaths of DMD CNVS users have been ascribed to respiratory causes as opposed to 14 of 27 who presented using CTMV to the U.S. center and died as a result [1,2]. While four with DMD from the U.S. center, and 6 from the Japanese center, have been over 50 years of age and CNVS dependent for 25 to 30 years, five have never been hospitalized for respiratory failure despite eventually having little to no measurable VC. Eight DMD patients who presented using CTMV were decannulated to CNVS and MIE and six then weaned to less than CNVS with their VCs increasing only after decannulation.
In 2010 the centers also presented 41 children with SMA type 1 dependent on CNVS instead of CTMV for a mean 5.9±1.8 years [12]. Today, there are over 80 CNVS dependent with SMA1, 22 between 20 and 30 years old. All are CNVS dependent since as young as 3 months of age with none having any functional muscle activity other than eye movements and most with 0 ml of VC.
In 1996, despite managing hundreds of CNVS users with ventilatory pump failure but all of whom with at least some bulbar innervated muscle function and believing that the latter was necessary to protect the airways, Bach told the parents of two boys with SMA type 1 that they would not survive 1 year without tracheostomy tubes. The two boys are now 30 and 29 years old, CNVS dependent since 8 months and 14 months old. The mother of the latter boy has another with SMA1, who is now 27 years old, also CNVS dependent since 4 months of age with 0 ml of VC and only trace eye movement.
The U.S. center also reported 354 ALS NVS users of whom 113 eventually required CNVS. They had required part-time NIV/NVS for 10.9±10.5 months before requiring CNVS for 12.8±16.2 months with an outlier CNVS dependent for 12 years [15]. MIE-exsufflation flows (MIE-EF) correlate with patency of the upper airways and successful use of MIE to clear the airways to avoid pneumonias and respiratory failure. Whereas adult patients with DMD and SMA type 1 typically have 250 to 370 L/min of MIE-EF, for ALS patients these flows can decrease below 150 L/min and necessitate tracheostomies [16]. However, only five of our 43 ALS patients with ventilatory pump failure have failed extubation to CNVS and MIE and undergone tracheostomies (Figure 3) even though many have very poor bulbar innervated muscle function. We have also now decannulated 10 CTMV dependent ALS patients to CNVS and MIE.
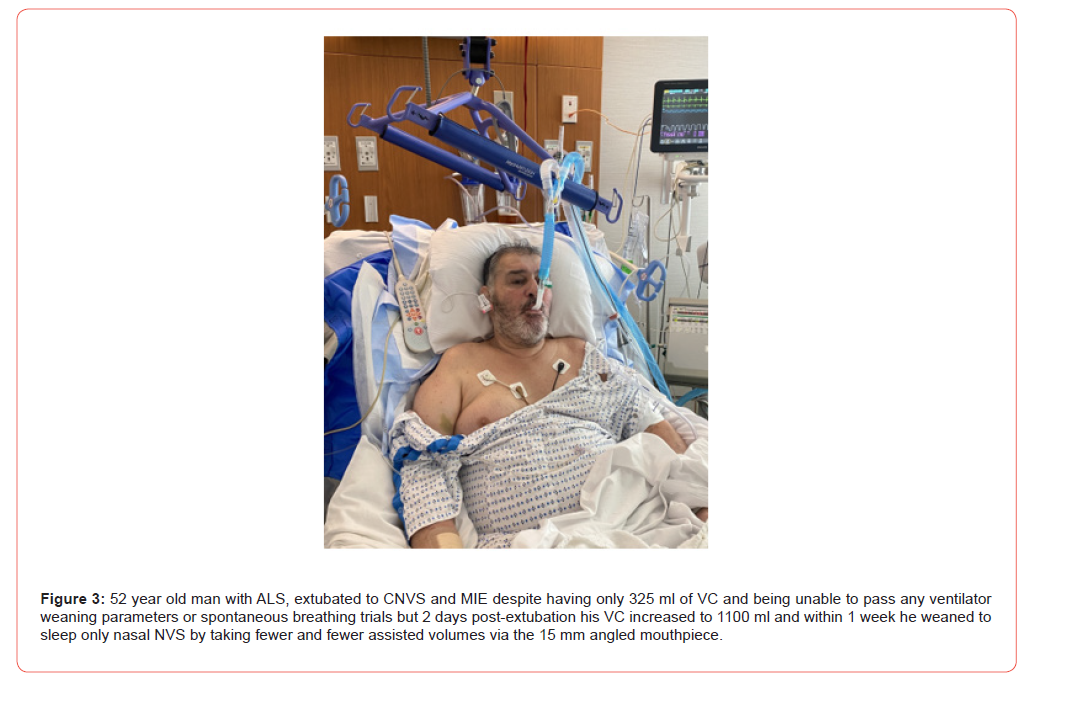
In 1988, extubation criteria were developed for patients who can not pass ventilator weaning parameters or breathing trials, to be extubated to CNVS and MIE. They include being fully alert, having no visceral organ failure, fever, or signs of infection, low to normal blood CO2 levels on ventilatory support, and having O2sat normal in ambient air MIE being used via the tube every hour or so until this is the case. One can then be extubated CNVS and MIE at NVS settings and physiologic back-up rates and always without supplemental oxygen. Upon mastering nasal ventilation, they are then trained in mouthpiece ventilation [1,2]. Using this approach, none of our myopathic, dystrophic, or lower motor neuron disease patients at any age have required tracheostomies, many even with 0 ml of VC, and some presenting using CTMV even decannulated since 1969 [17]. Following tube removal, patients often wean from the ventilator by taking fewer and fewer positive pressure ventilations (Figure 3) and, because the tubes are removed, their VCs increase. With VC over 250 ml they at least partially wean CNVS. Although there is no more risk of harm to speech or swallowing by being intubated over 3 weeks as there is under 3 weeks [4], if postextubation paroral feeding is unself, gastrostomies are performed under local anesthesia.
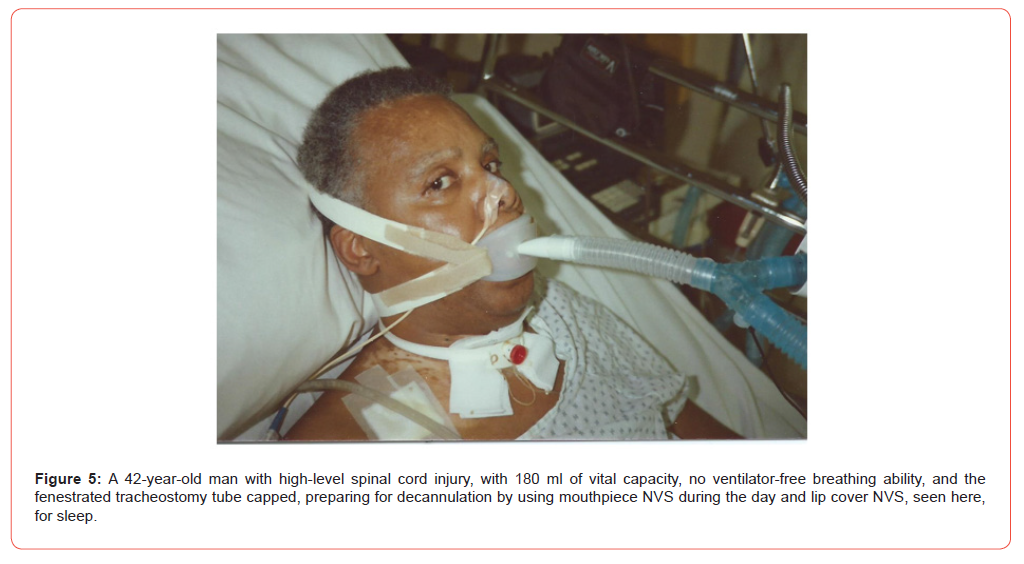
In 1996, we decannulated 50 ventilator unweanable spinal cord injured patients to CNVS and MIE [17,18], with some having poor bulbar innervated muscle function. We recently decannulated a 26 year old with SMA 1 who had been dependent on CNVS from 8 months of age until an emergency tracheotomy was needed. He has had 0 ml of VC for over 15 years, only trace eye movements almost since infancy, but kept the tube for 6 months then requested that it be removed and we accommodated him [19]. His MIE-EF via the upper airway were over 350 L/min which is why airway clearance was effective and tracheostomy was unnecessary. MIE was used up to every 20 to 30 min until the ostomy closed (Figure 4, 5).

Patients without upper MND causing glottis dysfunction and MIE-EF greater than 150 L/min can be good candidates for decannulation [16,19]. Whereas TMV typically increases ventilator dependence due to the secretions it causes, the constant tidal volumes, and deconditioning, decannulation makes it easier to breathe, speak, and swallow. Decannulated patients, even lacking any ability to breathe but with VC greater than 250 ml, at least partially wean from CNVS.
Oximetry Feedback Protocol
This is the use of oximetry for feedback using up to CNVS and MIE to maintain O2sat normal, or return it to normal, in ambient air during intercurrent respiratory tract infections and after extubations and decannulations. Patients typically use MIE for all desaturations and extend sleep-only NVS to CNVS when weakening and as needed. Tracheostomies should be avoided, and reversed when present, to permit weaning and off ventilator time by glossopharyngeal breathing [1,2,20,21].
Conclusion and Recommendations
A recent survey of diurnal mouthpiece NVS use had 198 responses with 1 or more patients using sleep plus daytime mouthpiece NVS in each center. There were 116 in Europe, 19 in South Europe, 19 in South America and in the United States, 10 in Australia, 9 in Canada, 9 in Asia, and 16 elsewhere including the Middle-East and Africa. Only 8 centers had over 50 mouthpiece NVS users with six of them cited in www.breatheNVS.com. However, very suboptimal settings of 200 to 400 ml and low pressure support levels are typically being used in most centers [22]. Many are also extending sleep bi-level PAP ventilation into daytime hours at low settings via mouthpieces despite the facts that bi-level, like any pressure preset ventilation and low volume settings, preclude the deep breaths needed for lung expansion and increasing cough flows and voice volumes. Bi-level devices also uncomfortably gush air at the patient’s throat to compensate for leak, do not have internal batteries for practical daytime use, and the EPAP is counterproductive and can not be turned off [23]. Yet, pressures of 17 cm H2O or more have been used optimally for hundreds of patients for rest and support since 1957 as have NVS volumes of 650 to 1800 ml since volume ventilation became available in 1976 [6,7]. Aiding and fully compensating for respiratory muscle failure by using NVS settings and MIE can not only sustain life and preserve its quality, but can also save up to $440,000 per year in nursing care per person as well as literally millions of dollars in charges for tracheostomies and prolonged futile critical care weaning attempts. With over $5 trillion the cost of medical care in the U.S. this year, what better time for a paradigm shift to more humane noninvasive management.
Acknowledgement
None.
Conflict of Interest
None.
References
- Bach JR, Goncalves MR, Hamdani I, Winck JC (2010) Extubation of patients with neuromuscular weakness: a new management paradigm. Chest 137(5): 1033-39.
- Bach JR, Sinquee DM, Saporito LR, Botticello AL (2015) Efficacy of mechanical insufflation-exsufflation in extubating unweanable subjects with restrictive pulmonary disorders. Respir Care 60(4): 477-83.
- Chiou M, Bach JR, Saporito LR, Albert O (2016) Quantitation of Oxygen induced hypercapnia in respiratory pump failure. Revista Portuguesa de Pneumologia, Portuguese Journal of Pulmonology 22(5): 262-65.
- Bach JR, Nair A. Comparison of the consequences of short vs. long-term intubation on speech and swallowing. Am J Phys Med Rehabil (in press).
- Hodes HL (1955) Treatment of respiratory difficulty in poliomyelitis. In Poliomyelitis. papers and discussions presented at the third international poliomyelitis conference, Lippincott, Philadelphia pp. 91-113.
- Bach JR, Goncalves MR (2023) A brief history of noninvasive mechanical ventilation. In: Bach JR, Gonçalves MR. A Compendium of Interventions for the Noninvasive Management of Ventilatory Pump Failure: for Humane Management of Neuromuscular Diseases, Spinal Cord Injury, Morbid Obesity, Chest Wall Deformity, Critical Care Neuromyopathy, and Other Neurological and Pulmonary Disorders, First Edition, Second Issue, ventilamed.com pp. 49-78.
- Bach JR, Alba AS, Saporito LR (1993) Intermittent positive pressure ventilation via the mouth as an alternative to tracheostomy for 257 ventilator users. Chest 103(1): 174-82.
- Bach JR, Saporito LR, Shah HR, Sinquee D (2014) Decanulation of patients with severe respiratory muscle insufficiency: efficacy of mechanical insufflation-exsufflation. J Rehabil Med 46(10): 1037-41.
- Carter RE, Donovan WH, Halstead L, Wilkerson MA (1987) Comparative study of electrophrenic nerve and mechanical ventilatory support in traumatic spinal cord injury. Paraplegia 25(2): 86-91.
- Bach JR (1993) Amyotrophic lateral sclerosis: communication status and survival with ventilatory support. Am J Phys Med Rehabil 72(6): 343-349.
- Patel S, Cuenant L, Bach JR (2019) Respiratory management of spinal muscular atrophy. Journal of Clinical Neurology, Neurosurgery and Spine 2(1): 119-127.
- Goncalves MR, Bach JR, Ishikawa Y, Saporito L, Winck JC, et al. (2021) Continuous noninvasive ventilatory support outcomes for neuromuscular disease: a multicenter data collaboration. Pulmonol 27(6): 509-517.
- Ishikawa Y, Miura T, Ishikawa Y, Aoyagi T, Ogata H, et al. (2011) Duchenne muscular dystrophy: survival by cardio-respiratory interventions. Neuromuscular Disorders 21(1): 47-51.
- Bach JR, Goncalves MR (2023) International Views, Patient Care Networks, and Paradigm Paralysis In: Bach JR, Gonçalves MR. A Compendium of Interventions for the Noninvasive Management of Ventilatory Pump Failure: for Humane Management of Neuromuscular Diseases, Spinal Cord Injury, Morbid Obesity, Chest Wall Deformity, Critical Care Neuromyopathy, and Other Neurological and Pulmonary Disorders 1(2): 617-31.
- Mahajan KR, Bach JR, Saporito LR, Perez N (2012) Diaphragm pacing and noninvasive respiratory management of amyotrophic lateral sclerosis/motor neuron disease. Muscle Nerve 46(6): 851-855.
- Bach JR, Upadhyaya N (2018) Association of need for tracheotomy with decreasing mechanical in-exsufflation flows in amyotrophic lateral sclerosis: a case report. Am J Phys Med Rehabil 97(4): e20-e22.
- Bach JR, Alba AS (1990) Noninvasive options for ventilatory support of the traumatic high level quadriplegic. Chest 98(3): 613-619.
- Bach JR (1991) New approaches in the rehabilitation of the traumatic high level quadriplegic. Am J Phys Med Rehabil 70(1): 13-20.
- Bach JR, Saporito LR, Schepis M (2023) Spinal muscular atrophy (SMA) type 1: case study of decannulation. Acta Scientific Medical Sciences 7(8): 182-186.
- Bach JR, Rajaraman R, Ballanger F, Tzeng AC, Ishikawa Y, et al. (1998) Neuromuscular ventilatory insufficiency: effect of home mechanical ventilator use v oxygen therapy on pneumonia and hospitalization rates. Am J Phys Med Rehabil 77(1): 8-19.
- Bach JR, Bianchi C, Vidigal-Lopes M, Turi S, Felisari G, et al. (2007) Lung inflation by glossopharyngeal breathing and “air stacking” in Duchenne muscular dystrophy. Am J Phys Med Rehabil 86(4): 295-300.
- Chatwin M, Gonçalves M, Gonzalez-Bermejoc J, Toussaint M, for the ENMC Respiratory Therapy Consortium (2020) 252nd ENMC international workshop: Developing best practice guidelines for management of mouthpiece ventilation in neuromuscular disorders. March 6th to 8th 2020, Amsterdam, the Netherlands. Neuromuscular disorders 30(9): 772-81.
- Crescimanno G, Greco F, Arrisicato S, Morana N, Marrone O (2016) Effects of positive end expiratory pressure administration during noninvasive ventilation in patients affected by amyotrophic lateral sclerosis: a randomized crossover study. Respirology 21(7): 1307-13.
-
John R Bach*, Antonio Huerta Armijo and Julia Dorta Díez de la Lastra. Evidence That No One Needs a Tracheotomy for Only Being Too Weak to Breathe. Glob J of Ped & Neonatol Car. 4(5): 2024. GJPNC.MS.ID.000598.
Breathing, Respiratory, Spinal cord injuries, Ventilation, Oxyhemoglobin, Extubations, Pathophysiology, Myopathies, Glossopharyngeal Breathing
-

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.






