 Research Article
Research Article
Corticosteroids in Spine Surgery
Juan Ulises Villanueva Morales1*, Ricardo Erick Robles Ortíz2, Alejandro Fabián Rodríguez Messina2 and Gabriel Herrera Armendáriz1
1 Neurosurgery, Hospital Regional Puebla, ISSSTE
2 Spine Surgery, Hospital Regional Puebla, ISSSTE
Corresponding AuthorDr. Juan Ulises Villanueva Morales, Hospital Regional Puebla, Instituto de Seguridad y Servicios Sociales de los Trabajadores del Estado (ISSSTE).
Received Date:February 02, 2026; Published Date:February 17, 2026
Abstract
Corticosteroids have a diversity of therapeutic indications in different fields of medicine; spinal pathology is no exception. Corticosteroids have a great anti-inflammatory and immunomodulatory potency, this due to the mechanism of action of this group of drugs. This review briefly discusses basic aspects of corticosteroids such as physiology and pharmacology as well as recent evidence-based recommendations in various spinal conditions such as: acute spinal cord injury, spinal tumors, cervical surgery (discectomy and anterior cervical fusion), low back pain, and spinal steroid infiltration. The use of corticosteroids is associated with side effects that affect different organs and systems, so they should be administered with caution and strategically and above all based on the evidence of their real benefit on patients.
Keywords:Corticosteroids; Spinal cord injury; Spinal cord compression; Low back pain; Cervical surgery; Spinal infiltration
Introduction
Corticosteroids are a group of drugs with great antiinflammatory and immunomodulatory power, these therapeutic properties make their indications broad, knowing the mechanism of action helps us to understand the broad therapeutic spectrum as an anti-inflammatory and its role in modulating the immune system and pain. In relation to pathological processes affecting the spine, corticosteroids have been indicated as part of conservative management, as well as postoperative complications. One of the main drawbacks of the use of corticosteroids is their side effects that increase morbidity and mortality, and these adverse effects are associated with high doses or prolonged treatments. There is still some controversy in the use of this group of drugs in certain pathological processes, while in others there is already wellestablished evidence on the therapeutic guideline such as: acute spinal cord injury, spinal tumors, low back pain, among others. This review briefly discusses basic aspects of corticosteroids such as physiology and pharmacology and recent evidence-based recommendations in various spinal conditions.
Corticosteroid physiology: hypothalamic-pituitaryadrenal axis
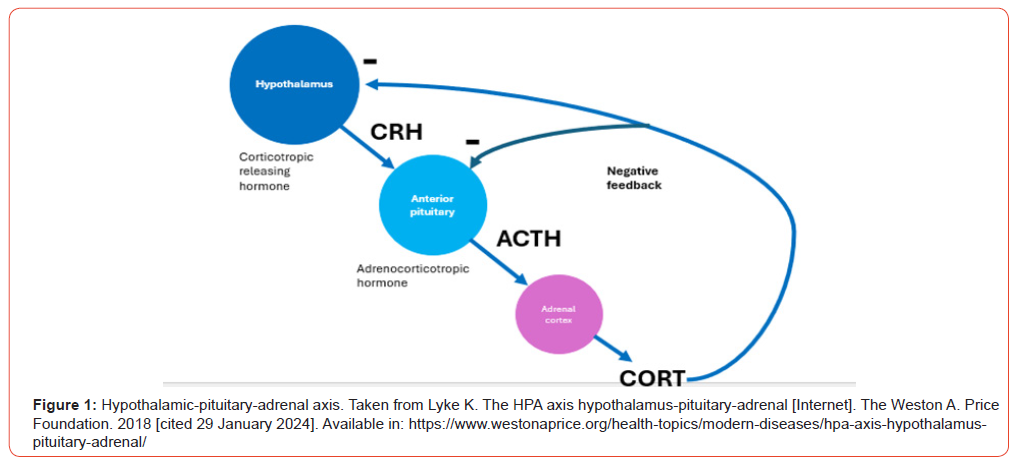
Endogenous cortisol production by the adrenal gland is controlled by the hypothalamic-pituitary-adrenal axis and occurs in a diurnal and circadian pattern every 24 h. Corticotropin-releasing hormone (CRH) is released from the hypothalamus and acts on the anterior pituitary gland to release adrenocorticotropic hormone (ACTH), which stimulates the production and release of cortisol from the adrenal gland (Figure1) [1]. Plasma cortisol concentrations are usually highest in the morning (e.g., 10 to 15 g/mL from 6:00 to 8:00 a.m.) and lowest at night while sleeping. Circulating cortisol exerts negative feedback on the production of ACTH and CRH [1- 2]. Normal function of the hypothalamic-pituitary-adrenal axis is important for overall health and well-being. Under stress-free conditions, cortisol production is about 20 mg daily in adults [2].
Pharmacology of Steroids
Steroids have played a prominent role in the treatment of many disease states since their identification nearly 80 years ago. Many of the clinical functions of steroids are related to their potent antiinflammatory and immunomodulatory properties [1]. Clinically relevant side effects of steroids are common and problematic, ranging from a minor case of acne to Cushing’s syndrome that can result in diabetes mellitus and life-threatening heart disease if left untreated. Side effects can occur at a wide range of doses and vary depending on the route of administration [1, 3].
Mechanism Of Action
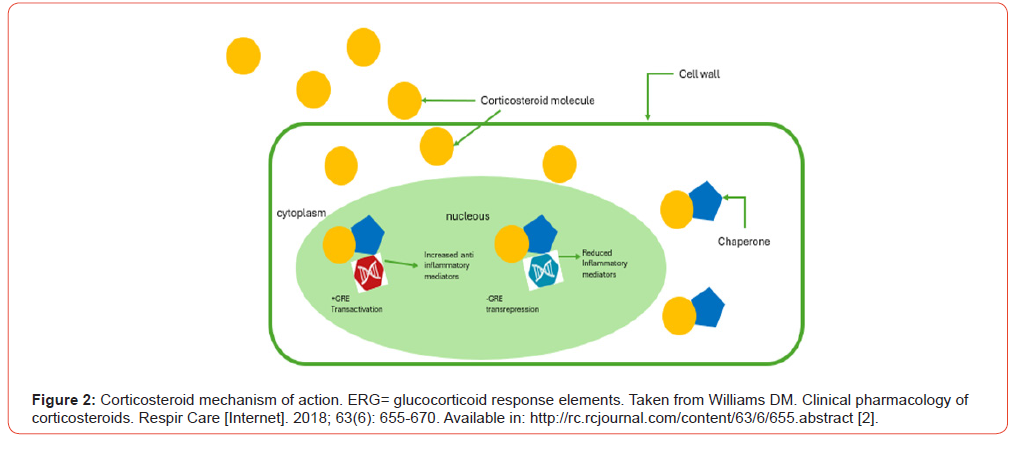
Corticosteroids affect numerous steps in the inflammatory pathway, increasing their usefulness. To exert an effect, the steroid molecule diffuses across cell membranes and binds to glucocorticoid receptors, causing a conformational change in the receptor. The receptor-glucocorticoid complex can move into the cell nucleus and binds to glucocorticoid response elements (Figure 2). Glucocorticoid response elements are associated with genes that suppress (trans repression) or stimulate (transactivation) transcription, resulting in ribonucleic acid and protein synthesis; Finally, these agents inhibit transcription factors that control the synthesis of pro-inflammatory mediators [4]. Another important effect is the inhibition of phospholipase A2, which is responsible for the production of numerous inflammatory mediators.2 Corticosteroids inhibit genes responsible for the expression of cyclooxygenase-2, inducible nitric oxide synthase, and proinflammatory cytokines, including tumor necrosis factor-alpha and several interleukins. In contrast, corticosteroids initiate upregulation of lipocortin and annexin A1, a protein that reduces the synthesis of prostaglandins and leukotrienes and also inhibits cyclooxygenase-2 activity and reduces neutrophil migration to inflammatory sites. Because the action of corticosteroids occurs intracellularly, the effects persist, even when detection in plasma is absent [2, 4].
Pharmacokinetics and Pharmacodynamics
Corticosteroids used systemically are classified according to potency, mineralocorticoid effects, and duration of suppression of the hypothalamic-pituitary-adrenal axis (Table 1) [5]. Potency is expressed in relation to hydrocortisone and is useful for determining comparable doses. The activity of mineralocorticoids is also described in relation to hydrocortisone, and structural modifications of the steroid molecule are designed to increase the potency and minimize the effects of mineralocorticoids when these agents are used in pharmacological doses to prevent or treat allergic, inflammatory, or immune reactions [1, 5].
Table 1: Systemic properties of corticosteroids.
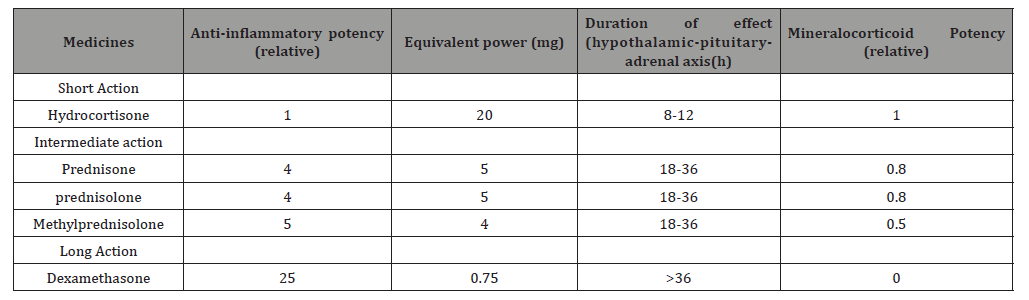
Although more potent agents exhibit fewer mineralocorticoid effects, at commonly used doses, side effects are common with all available agents. Finally, these agents are classified as short-acting, medium-acting, or long-acting based on the duration of suppression of the hypothalamic-pituitary-adrenal axis [2]. The duration of action is not well correlated with the duration of the effect, possibly due to the intracellular mechanism. Because the mechanism of corticosteroids is intracellular, the effects persist even after corticosteroids have disappeared from circulation [2]. Therefore, the duration of action of specific corticosteroids is often based on the duration of effects on the hypothalamic-pituitary-adrenal axis, while the actual therapeutic action is more prolonged. The onset of effect of systemic corticosteroids is often delayed by 3 to 8 h, regardless of the route of administration [5]. This may be related to the intracellular and nuclear effect of these therapies. When you look at the anti-inflammatory effect, it’s often dramatic. For life-threatening conditions, acute use of systemic corticosteroids should not be delayed [2, 5].
Acute Spinal Cord Injury
Acute spinal cord injury (AML) is a devastating event that can have a profound impact on the lives of patients and their families [6]. Currently, pharmacological interventions for spinal cord injuries revolve around the use of corticosteroids and the decision whether or not to use them. While there is no definitive medical treatment, the role of methylprednisolone in the treatment of LAME has been thoroughly investigated; however, its use remains controversial. While methylprednisolone initially showed promise in the efficacy of LAME treatment, more recent studies have questioned its use citing numerous systemic adverse effects. Pharmacological treatments in this area are poorly understood due to the scarcity of knowledge about the pathophysiology and heterogeneity of patients presenting with these conditions [7].
A randomized phase 3 trial by Bracken et al. [8] demonstrated that methylprednisolone sodium succinate improves sustained neurologic recovery, and has been replicated in a second trial [9]. Therapy should be initiated within eight hours of injury using an initial bolus of 30 mg/kg intravenously over 15 minutes, followed 45 minutes later by a continuous infusion of 5.4 mg/kg/hour for 24 hours. Further improvement in recovery of motor function has been shown to occur when maintenance therapy is extended over 48 hours. This is particularly evident when the initial bolus dose could only be administered three to eight hours after injury [8, 9].
More recent studies have explored the effectiveness of corticosteroids in LAME more extensively. In 2017, Sunshine et al, [10] retrospectively reviewed 160 patients with LAME who received methylprednisolone in an effort to determine their motor function. At discharge, they found no improvement in ASIA score or motor score among methylprednisolone recipients. Interestingly, the authors found no ill effects after adjusting for age, sex, race, weight, level of injury, and receipt of surgery [10]. The authors concluded that methylprednisolone should not be administered in patients with traumatic spinal cord injuries due to negligible improvement in motor scores [10]. A meta-analysis by Liu et al, [11] evaluated 1863 patients and concluded that treatment with methylprednisolone was not significantly associated with better outcomes. Patients were not found to have an increase in motor score, sensory retrieval or ASIA grade [11]. Similar results were published by Sultan et al, [12] in 2020 where they reported that methylprednisolone was not associated with significant short- or long-term improvement in motor or sensory scores [12].
Adverse Reactions
In patients with LAME, often a particularly vulnerable patient population, the adverse effects of steroids have the potential to be devastating. Sultan et al, [12] in a review of 12 studies (5 randomized controlled trials and 7 observational studies), reported on the systemic effects of methylprednisolone and noted an increased risk of pneumonia and hyperglycaemia compared to controls, ultimately concluding in a recommendation of “careful consideration” for steroid use in patients with LAME [12]. Liu et al revealed that methylprednisolone treatment demonstrated numerous adverse effects, including a significantly higher incidence of gastrointestinal bleeding and respiratory tract infection, ultimately leading to their recommendation against methylprednisolone therapy [11]. Although MP remains a common treatment option, mixed evidence of its efficacy, reports of unfavorable clinical effects, and safety considerations prevent improving the controversy surrounding its use. Current evidence in the literature does not support the routine use of systemic corticosteroids in patients with acute AML [7].
Spinal Tumors
Spinal cord compression is a well-known complication in cancer patients, with a huge potential impact on quality of life. The role of corticosteroids is also controversial in metastatic spinal cord compression (MSMC) [7]. Primary spine cancer is rare, accounting for 5% of tumors [7]. Consequently, most cancerous lesions are the result of metastatic disease (up to 30%) and it is estimated that between 10% and 20% of them develop spinal cord compression [13]. Spinal cord metastases can be classified into three types according to the location of the tumor deposits: extradural (epidural), intradural, extramedullary, and intramedullary [13]. The most frequent location is extradural (> 90%), usually as a result of the direct extension of a vertebral metastasis. The posterior half of the vertebral body is the most frequently affected, followed by the posterior elements (lamina, pedicles) [14]. The most common site of CMME is the thoracic spine (60% 80%), followed by the lumbosacral spine (15% 30%) and the cervical spine (<15%) [13]. Sorensen et al, [15] demonstrated that initial treatment of CMME included high-dose dexamethasone (96 mg intravenous bolus followed by 96 mg orally for 3 days and tapered over 10 days), showing that patients receiving dexamethasone treatment had significantly better gait function compared to those who did not. To date, this study represents the only randomized trial comparing the outcomes of steroid therapy in CMME with a control group [15].
As with LAME, there are no clear guidelines for how corticosteroids should be administered for patients with MSMC.7 Current practices cite the optimal balance between the efficacy of dexamethasone treatment and the management of side effects using a low-dose bolus of 10 mg IV and a low-dose protocol of 16 mg daily divided into 4 doses of 4 mg administered orally or intravenously and decreased over the course of 7 to 14 days [14]. This regimen is most effective when started immediately after confirmation of CMME [14]. Patients who received dexamethasone treatment within 12 hours of symptom onset had significantly better chances of preserving or regaining ambulatory status, an outcome that has been linked to decreased mortality and improved quality of life [16]. In a recent systematic review evaluating CMME and steroid therapy, Kumar et al. [14] concluded that corticosteroid therapy followed by definitive treatment with radiation therapy or surgery may increase the number of patients able to maintain ambulatory capacity 1 year after therapy, but did not demonstrate an apparent effect on survival. however, treatment of CMME with dexamethasone reduces vasogenic edema [14].
Adverse Reactions
More recent studies have shown that treatment with highdose dexamethasone does not significantly improve ambulatory status, bladder function, or pain and instead results in higher rates of gastrointestinal bleeding complications with perforation, pneumonia, wound infections, and persistent hyperglycemia compared to lower doses [15].
Spinal Cord Compression Without Neurological Involvement
For patients who have CMME without neurological symptoms, steroids may not be necessary. A clinical trial conducted by Maranzano et al, [17] followed 20 patients with metastatic disease in the spine as seen on MRI or CT scan, with no neurological deficits or radiculopathy, who received radiation treatment without systemic steroids. Their results were promising: 85% reported an improvement in pain, no patients experienced motor impairment, and patients requiring support became ambulatory with no new motor symptoms [17]. The findings of their study suggest that nonsteroidal radiation may be a more ideal treatment regimen in patients without motor impairment to avoid the systemic adverse effects of steroids [17]. However, if the patient’s neurological status worsens, re-evaluation of the overall treatment regimen and the possible role of steroid treatment may be warranted [17].
Cervical Surgery: Discectomy and Anterior Cervical Fusion
The anterior cervical approach is commonly used in the surgical management of cervical spine pathology, although it is considered safe to prevent the development of the cervical spine [18]. Postoperative dysphagia has been reported to have an incidence of up to 79% [19]. Symptoms occur primarily during the early postoperative period, decreasing significantly within 6 months after surgery and ultimately stabilizing at 1 year in most patients. The etiology of postoperative dysphagia is multifactorial [20]. Contributing factors include prevertebral soft tissue edema, vocal cord paresis, scar tissue formation, equipment complication, and cervical graft material. Multiple risk factors for undesirable complications have been identified, such as smoking (particularly for dysphonia), prior cervical surgery, multi-level or higher-level fusion, preoperative opioid use, revision surgery, and female gender [21].
A promising therapeutic intervention to decrease the incidence of dysphagia is the use of perioperative steroids; by reducing the inflammatory response, steroids should decrease dysphagia as well as pain in the postoperative period [22]. Steroid use has led to a decrease in the incidence and severity of dysphagia, as well as airway edema, in some studies [23]. In addition to these results inconsistent with dysphagia, there are concerns about adverse effects of steroids, such as delayed melting time and increased infection rates [24]. The use of systemic and local steroids has been reported in previous studies. The main justification for local steroids over systemic steroids has been the presumed lower rate of systemic complications with local [24]. Shari Cui et al conducted a randomized trial with patients who underwent elective anterior cervical procedures, and the steroid group received 0.3 mg/kg intravenous (IV) dexamethasone preoperatively and 2 doses of 0.15 mg/kg IV dexamethasone every 8 hours for 2 postoperative doses [25]. They found that this regimen was associated with a significant decrease in postoperative dysphagia, with no effect on rates of medical or surgical complications [25]. They cited several additional studies with different duration times and specific medications, including one study evaluating 24 hours of postoperative methylprednisolone and another study administering dexamethasone once intraoperatively and four doses in the first 24 hours postoperatively [25]. Again, the results demonstrated a significant decrease in postoperative dysphagia. These studies demonstrate the potential for future research to standardize the recommended dose, type, and duration of administration [25].
For the severity of dysphagia/odynophagia in the short term, Adenikinju et al. [24] in their systematic review report that there was no significant difference between the groups (steroids versus placebo) at any time during the follow-up period [24]. Regarding long-term dysphagia, Jeyamohan et al, [26] evaluated 112 patients after previous cervical spine surgery of several levels, which were divided into 2 groups (dexamethasone vs. placebo) reported a decrease in the severity of dysphagia, in the steroid group at 1 month postoperatively, however, this difference decreased [2]. In relation to prevertebral soft tissue edema (PTBTE), 2 studies were evaluated [20]. Nam et al, [27] evaluated the ETBP by measuring the area (cm2) from the lower border of C1 to the upper endplate of C7 for patients who received high-dose dexamethasone, low-dose dexamethasone, and placebo, respectively [27]. The ETBP peaked on day 3 postoperatively and continued to decline until day 5, but no significant difference was observed between the groups.27 Song et al, [23] found similar trends when measuring the length of the ETBP (mm) at each level from C2 to C7 using lateral radiographs. During postoperative days 2-6, the ETBP increased significantly more in the control group than in those who received steroids [23].
Fusion time was assessed by Jeyamohan et al, [26] in a randomized study where they found a significantly lower fusion rate at 6 months follow-up in patients who had received steroids; however, there was no significant difference in fusion rate at 12 months of follow-up [26]. Their findings suggest that while steroids may delay fusion in the short term, they ultimately do not affect fusion in the long term [26]. The mean hospital stay was shorter in the group receiving systemic steroids compared to the control group in two separate studies [23, 27]. The complication rate was 2% [24]. Complications in the studies included in this cohort were: vocal cord motility disorders; and tonsillar laceration secondary to intubation. There were no reports of intraoperative esophageal perforation, vascular involvement, or cerebrospinal fluid leakage [24].
Topical Steroid Use
There is recent literature suggesting the benefits of topical corticosteroid preparation, with the potential advantage of reducing systemic adverse effects found with intravenous corticosteroids [20]. Regarding the incidence Koreckij et al, found no statistically significant difference at 6 weeks postoperatively; however, at 3 months there was a significant decrease in the incidence of dysphagia in the steroid group compared to the control group [28]. A randomized controlled trial conducted by Kim et al demonstrated a significant improvement in dysphagia severity at day 2 and 1 month postoperatively [29]. Another randomized controlled trial by Jenkins et al, [30] compared local triamcinolone and intravenous dexamethasone administered intraoperatively, both routes of administration showing better patient-reported outcomes compared to controls who did not receive steroids at 6 weeks, 3 months, and 1 year postoperatively [30]. However, in the early postoperative period within the first 2 weeks after ACDF, patients who received local steroids had a significant decrease in dysphagia symptoms compared to systemic steroids and controls.30 A systematic review and meta-analysis by Yu et al, [31] which included eight randomized trials, further supported topical steroid administration. They found a significant reduction in dysphagia, neck pain, and PTBD postoperatively compared to placebo, again most noticeable in the first 2 weeks postoperatively [31]. There were no apparent differences between local and systemic steroids in terms of infections, other complications, and hospital readmission, as reported by Adenikinju et al. [24] the intraoperative dose of local steroids can only be administered once, whereas multiple doses of systemic steroids were administered up to 48 hours postoperatively [24]. Studies using local steroids consistently reported statistically significant differences in the incidence and severity of dysphagia through 3 months postoperatively, however, results were more inconsistent in systemic steroid studies [24]. Taken together, these results suggest that local steroids are more cost-effective and possibly more effective in preventing dysphagia/odynophagia [24].
Low Back Pain
Low back pain is a common symptom in people of all ages and socioeconomic status. The global point prevalence of low back pain (acute, subacute, and chronic) was 7.33% in 2017, with 577 million people affected at one time [32]. People quit their jobs more often because of low back pain than diabetes, hypertension, neoplasia, asthma, heart and respiratory diseases combined [33]. We found limited evidence of steroids compared to NSAIDs (one study) for reducing disability [34]. Acute low back pain is defined as: 4 weeks of symptoms or less, with or without radiculopathy. It is often caused by herniated disc or stenosis of the canal or foramen, but it can also have a nonspecific presentation and imaging. Radicular pain includes radiating pain or paresthesia involving one or both lower extremities, due to compression of one or more nerve roots [20].
Gastaldi et al, [35] in a randomized, multicenter, doubleblind trial in patients with acute lumbar radicular pain due to intervertebral disc herniation, where they assessed the short-term efficiency and tolerance of ketoprofen and methylprednisolone, showed that methylprednisolone (60 mg/day) or ketoprofen (200 mg/day) did not show a significant difference between the groups compared to placebo [35]. Another double-blind randomized controlled trial by Friedman et al, [36] evaluated the effects of a single dose of intravenous steroid on non-radicular back pain with acute symptoms; there were no statistically or clinically significant differences in the numerical pain rating scale at one month of follow-up compared to placebo [36].
Finally, Chou et al, [37] in a systematic review of thirteen trials (1047 participants) conclude that the effects of systemic corticosteroids in people with non-radicular low back pain are unclear and systemic corticosteroids are probably ineffective for spinal stenosis [37]. A single dose or a short course of systemic corticosteroids for low back pain does not appear to be associated with an increased risk of any serious adverse events [37]. Systemic corticosteroids appear to be slightly effective in improving pain and function in the short term in people with low back radicular pain not due to spinal stenosis, and may slightly improve long-term function [37]. For low back pain due to spinal stenosis, limited evidence suggested that systemic corticosteroids are probably no more effective than placebo for short-term pain or function [35].
Spinal Infiltration of Steroids
Depending on the pathology, spinal infiltrations include interlaminar epidural (IEI), transforaminal (TF), sacroiliac, and facet infiltrations. Common pathologies include herniated disc, spinal stenosis, facet or sacroiliac joint pain [20]. According to NASS (North American Spine Society) guidelines, [38] epidural spinal infiltrations are indicated when two of the following four criteria are met:[38]
• Severe pain that causes functional disability
• four weeks of pain or unresponsiveness to 4 weeks of
non-invasive care.
• Presence of radiculopathy or referred dermatome pain
• Correlative nerve involvement on CT or MRI.
IEIs should be performed under image guidance with twodimensional imaging (AP and lateral) with the use of contrast (if you are not allergic) [20]. Historically, these infiltrations have been performed in a series of three, which is no longer recommended. Repeat injections are based on the previous response, and if no relief was obtained with the first IEI, the patient should be reevaluated before being automatically enrolled for the second injection. A maximum of four IEIs can be performed in 6 months and six IEIs in 12 months [20]. Up to two TF infiltrations are permitted, and IEIs or flow rates should not be performed at the same time with a TF. Contraindications to injections include allergy to intravenous contrast, local or systemic infection, uncontrolled congestive heart failure, uncontrolled diabetes, pregnancy, and inability to obtain percutaneous access [20].
Lumbar Interlaminar Epidural Infiltration
The clinical effectiveness and usefulness of epidural infiltrations have been studied extensively, but differences in study designs, methodology, type of procedures, medications used, and diagnostic criteria make comparisons very difficult [39]. Some initial studies and reviews of non-fluoroscopy-guided infiltrations showed very little success and no major benefit when comparing epidural infiltrations of steroids with intramuscular injections or saline injections [39]. A recent systematic review, followed by a version published in Spine, concluded that a review of 20 placebocontrolled trials provided moderate-quality evidence that epidural corticosteroid injections are effective, although the effects are small and short-term [40]. The defect of this conclusion is that local anaesthetics are not harmless. The extensive anti-inflammatory effects, long-lasting effects beyond the pharmacological profiles of steroids, and the non-significant difference between steroids and local anaesthetics and the abundance of experimental and clinical evidence leads, in our opinion, to an inaccurate synthesis of evidence [40].
Manchikanti et al,[41] in their meta-analysis published in 2021 in which 21 clinical trials were evaluated, with at least 6 months of follow-up and carried out under fluoroscopic guidance, with evaluation of methodological quality, synthesis of qualitative and quantitative evidence, showed level I or strong evidence for the effectiveness of lumbar epidural infiltrations with local anesthetic and steroids and level II to I or moderate to strong evidence of local anesthetic only in the management of lumbar radiculopathy or sciatica secondary to lumbar disc herniation[41].
Cervical Epidural Infiltration
Epidural infiltration of steroids (IEE) and local anesthetics (LA) achieves a high concentration of the treatment agent within the epidural space to inhibit inflammation and reduce nociceptive afferent signalling [42]. The indications for cervical epidural steroid infiltration are: cervical radicular pain, radiculitis, and cervical radiculopathy [42]. The techniques available to access the cervical epidural space include the interlaminar (IL) and transforaminal (TF) approaches [43]. The TF route has become more common because it can place the medication more directly at the site of the proposed cause of pain [43]. The varied techniques of IEI and the diversity of radicular pain etiologies make it difficult to compare studies, and it is likely that all types of IEIs are not equally effective for all causes of radiculopathy [43]. The systematic review conducted by Manchikanti et al. [44] shows the synthesis of the best qualitative evidence with level II evidence of the efficacy of cervical interlaminar epidural infiltrations with local anesthesia with or without steroids [44]. Diwan et al,[45] al report that the evidence is good for radiculitis secondary to herniated disc with local anesthetics and steroids, limited only with local anesthetics, while it is limited for local anesthetics with or without steroids, for axial or discogenic pain, spinal stenosis pain, and post-surgical pain syndrome [45].
Transforaminal Lumbar Infiltration
Transforaminal (or periradicular infiltration) of corticosteroids under fluoroscopic guidance delivers high concentrations of therapeutic agents to the site of pathology [39]. However, the evidence for transforaminal steroid injection is more controversial, and there are only a few randomized controlled trials examining the efficacy of this treatment in relieving radicular pain and disability [39]. In a meta-analysis conducted by Quraish et al, [46] they reported that transforaminal infiltrations show a significant reduction in pain scores in patients with lumbar radiculopathy compared to doing nothing (conservative treatment without infiltration therapy and probably lumbar interlaminar epidural infiltrations), in this meta-analysis, no additional benefit was found from the addition of steroids to the local anesthetic and this effect was maintained at 12 months [46]. On the other hand, MacVicar et al, [47] conclude that a substantial proportion of patients with lumbar radicular pain caused by contained disc herniations, lumbar transforaminal infiltration of corticosteroids is effective in reducing pain, restoring function, reducing the need for further medical care, and avoiding surgery [47]. The evidence supporting this conclusion was revealed by a thorough review of all published data and was found to be much more convincing than it would have been if the literature review had been of the limited scope of a traditional “systematic review” of randomised and controlled trials only [47].
Conclusions
Corticosteroids have a powerful anti-inflammatory effect and are useful in different conditions from the surgical point of view that affect the spine, however, the use of these drugs is associated with side effects that affect different organs and systems, so they must be administered with caution and strategically and above all based on the evidence of their real benefit on patients.
• There is no consistent evidence to support the use of
steroids in acute spinal cord injury, on the contrary, it increases
the risk of adverse effects that increase morbidity and mortality
in patients with this type of disease.
• Regarding metastatic compression of the spinal cord and
as in acute spinal cord injury there are no clear guidelines on
how corticosteroids should be administered for patients with
metastatic compression of the spinal cord, however, a decrease
in vasogenic edema is observed, but the use of steroids does not
improve patient survival.
• The usefulness of steroids in the management of
postoperative dysphagia in anterior cervical discectomy and
fusion is well documented, as is the role of topical steroids and
their potential use in this type of surgery at the cervical level.
• The literature does not support the use of corticosteroids
in the setting of acute low back pain, the effects of systemic
corticosteroids in people with non-radicular low back pain are
unclear, and systemic corticosteroids are probably ineffective
for spinal stenosis.
• Corticosteroids can also be administered by interlaminar,
transforaminal, sacroiliac and facet epidural infiltrations,
thereby achieving a high concentration of the treatment agent
within the epidural space to inhibit inflammation and reduce
nociceptive afferent signaling and thus contribute to the
treatment of pain.
• Current practice cites the optimal balance between the
efficacy of steroid treatment and the control of side effects,
so studies on the dosage and duration, as well as the type of
corticosteroid to be administered, are needed in order to
achieve this balance between efficacy and control of side effects.
Acknowledgements
None
Conflict of Interest
No Conflict of Interest.
References
- Ericson-Neilsen W, Kaye A (2014) Steroids: pharmacology, complications, and practice delivery issues. Ochsner J. Summer 14(2): 203-207.
- Williams DM (2018) Clinical Pharmacology of Corticosteroids. Respir Care 63(6): 655-670.
- Stewart PM, Krone NP (2011) The adrenal cortex. In: Melmed S, Polonsky K, Larsen PR, Kronenberg H (eds.) Williams Textbook of Endocrinology. (12thedn). Philadelphia, PA: Saunders. In. eds.
- Tantisira KG, Damask A, Szefler SJ, Schuemann B, Markezich A, et al. (2012) Investigators. Genome-wide association identifies the T gene as a novel asthma pharmacogenetic locus. Am J Respir Crit Care Med 185(12): 1286-1291.
- Samuel, Sophie Nguyen, Thuy Choi H (2017) Pharmacologic Characteristics of Corticosteroids. Journal of Neurocritical Care.
- Ahuja CS, Wilson JR, Nori S, Kotter MRN, Druschel C, et al. (2017) Traumatic spinal cord injury. Nat Rev Dis Primers 27(3): 17018.
- Canseco JA, Karamian BA, Bowles DR, Markowitz MP, DiMaria SL, et al. (2021) Updated Review: The Steroid Controversy for Management of Spinal Cord Injury. World Neurosurg 1-8.
- Bracken MB, Shepard MJ, Collins WF, Holford TR, Young W, et al. (1990) A randomized, controlled trial of methylprednisolone or naloxone in the treatment of acute spinal-cord injury. Results of the Second National Acute Spinal Cord Injury Study. N Engl J Med 322(20): 1405-1411.
- Bracken MB, Shepard MJ, Holford TR, Leo-Summers L, Aldrich EF, et al. (1997) Administration of methylprednisolone for 24 or 48 hours or tirilazad mesylate for 48 hours in the treatment of acute spinal cord injury. Results of the Third National Acute Spinal Cord Injury Randomized Controlled Trial. National Acute Spinal Cord Injury Study. JAMA 277(20): 1597-1604.
- Sunshine JE, Dagal A, Burns SP, Bransford RJ, Zhang F, (2017) Methylprednisolone Therapy in Acute Traumatic Spinal Cord Injury: Analysis of a Regional Spinal Cord Model Systems Database. Anesth Analg 124(4): 1200-1205.
- Liu Z, Yang Y, He L, Pang M, Luo C, et al. (2019) High-dose methylprednisolone for acute traumatic spinal cord injury: A meta-analysis. Neurology 93(9): e841-e850.
- Sultan I, Lamba N, Liew A, Doung P, Tewarie I, et al. (2020) The safety and efficacy of steroid treatment for acute spinal cord injury: A Systematic Review and meta-analysis. Heliyon 6(2): e03414.
- Penas-Prado M, Loghin ME (2008) Spinal cord compression in cancer patients: review of diagnosis and treatment. Curr Oncol Rep 10(1): 78-85.
- Kumar A, Weber MH, Gokaslan Z, Wolinsky JP, Schmidt M, et al. (2017) Metastatic Spinal Cord Compression and Steroid Treatment: A Systematic Review. Clin Spine Surg 30(4): 156-163.
- Sørensen S, Helweg-Larsen S, Mouridsen H, Hansen HH (1994) Effect of high-dose dexamethasone in carcinomatous metastatic spinal cord compression treated with radiotherapy: a randomised trial. Eur J Cancer 30A (1): 22-27.
- Skeoch GD, Tobin MK, Khan S, Linninger AA, Mehta AI (2017) Corticosteroid Treatment for Metastatic Spinal Cord Compression: A Review. Global Spine J 7(3): 272-279.
- Maranzano E, Latini P, Beneventi S, Perruci E, Panizza BM, at al. (1996) Radiotherapy without steroids in selected metastatic spinal cord compression patients. A phase II trial. Am J Clin Oncol 19(2): 179-183.
- Daniels AH, Riew KD, Yoo JU, Ching A, Birchard KR, at al. (2008) Adverse events associated with anterior cervical spine surgery. J Am Acad Orthop Surg 16(12): 729-738.
- Riley LH, Vaccaro AR, Dettori JR, Hashimoto R (2010) Postoperative dysphagia in anterior cervical spine surgery. Spine (Phila Pa 1976) 35(9 Suppl): S76-85.
- Nourbakhsh A, Harrison K (2023) Use of Steroids in Spine Surgery. J Am Acad Orthop Surg 31(13): 651-659.
- Papavero L, Heese O, Klotz-Regener V, Buchalla R, et al. (2007) The impact of esophagus retraction on early dysphagia after anterior cervical surgery: does a correlation exist? Spine (Phila Pa 1976) 32(10): 1089-1093.
- Lee SH, Kim KT, Suk KS, Park KJ, Oh KI, et al. (1976) Effect of retropharyngeal steroid on prevertebral soft tissue swelling following anterior cervical discectomy and fusion: a prospective, randomized study. Spine (Phila Pa 1976) 36(26): 2286-2292.
- Song KJ, Lee SK, Ko JH, Yoo MJ, Kim DY, at al. (2014) The clinical efficacy of short-term steroid treatment in multilevel anterior cervical arthrodesis. Spine J 14(12): 2954-2958.
- Adenikinju AS, Halani SH, Rindler RS, Gary MF, Michael KW, et al. (2017) Effect of perioperative steroids on dysphagia after anterior cervical spine surgery: A systematic review. Int J Spine Surg 11(2): 9.
- Cui S, Daffner SD, France JC, Emery SE (2019) The Effects of Perioperative Corticosteroids on Dysphagia Following Surgical Procedures Involving the Anterior Cervical Spine: A Prospective, Randomized, Controlled, Double-Blinded Clinical Trial. J Bone Joint Surg Am 101(22): 2007-2014.
- Jeyamohan SB, Kenning TJ, Petronis KA, Feustel PJ, Drazin D, et al. (2015) Effect of steroid use in anterior cervical discectomy and fusion: a randomized controlled trial. J Neurosurg Spine 23(2):137-143.
- Nam TW, Lee DH, Shin JK, Goh TS, Lee JS (2013) Effect of intravenous dexamethasone on prevertebral soft tissue swelling after anterior cervical discectomy and fusion. Acta Orthop Belg 79(2): 211-215.
- Koreckij TD, Davidson AA, Baker KC, Park DK (2016) Retropharyngeal Steroids and Dysphagia Following Multilevel Anterior Cervical Surgery. Spine (Phila Pa 1976). 41(9): E530-534.
- Kim HJ, Alluri R, Stein D, Lebl D, Huang R, et al. (2021) Effect of Topical Steroid on Swallowing Following ACDF: Results of a Prospective Double-Blind Randomized Control Trial. Spine (Phila Pa 1976) 46(7): 413-420.
- Jenkins TJ, Nair R, Bhatt S, Rosenthal BD, Savage JW, et al. (2018) The Effect of Local Versus Intravenous Corticosteroids on the Likelihood of Dysphagia and Dysphonia Following Anterior Cervical Discectomy and Fusion: A Single-Blinded, Prospective, Randomized Controlled Trial. J Bone Joint Surg Am 100(17): 1461-1472.
- Yu H, Dong H, Ruan B, Xu X, Wang Y (2021) Intraoperative Use of Topical Retropharyngeal Steroids for Dysphagia after Anterior Cervical Fusion: A Systematic Review and Meta-Analysis. Dis Markers.
- (2018) Institute for health metrics and evaluation, Available: http://www.healthdata.org/data-visualization/gbd-compare
- Schofield P (2008) Assessment and management of pain in older adults with dementia: a review of current practice and future directions. Curr Opin Support Palliat Care 2: 128-132.
- Gianola S, Bargeri S, Del Castillo G, Corbetta D, Turolla A, et al. (2022) Effectiveness of treatments for acute and subacute mechanical non-specific low back pain: a systematic review with network meta-analysis. Br J Sports Med 56(1): 41-50.
- Gastaldi R, Durand M, Roustit M, Zulian M, Monteiro I, et al. (2019) Short-term Efficiency and Tolerance of Ketoprofen and Methylprednisolone in Acute Sciatica: A Randomized Trial. Pain Med. 20(7):1294-1299.
- Friedman BW, Holden L, Esses D, Bijur PE, Choi HK, et al. (2006) Parenteral corticosteroids for Emergency Department patients with non-radicular low back pain. J Emerg Med. 31(4): 365-370.
- Chou R, Pinto RZ, Fu R, Lowe RA, Henschke N, et al. (2022) Systemic corticosteroids for radicular and non-radicular low back pain. Cochrane Database Syst Rev 10(10): CD012450.
- (2020) North American Spine Society: Epidural Steroid Injection and Selective Spinal Nerve Blocks. North American Spine Society (NASS).
- Rivera CE (2018) Lumbar Epidural Steroid Injections. Phys Med Rehabil Clin N Am 29(1):73-92.
- Oliveira CB, Maher CG, Ferreira ML, Hancock MJ, Oliveira VC, et al. (2020) Epidural Corticosteroid Injections for Sciatica: An Abridged Cochrane Systematic Review and Meta-Analysis. Spine (Phila Pa 1976) 45(21): E1405-E1415.
- Manchikanti L, Knezevic E, Knezevic NN, Sanapati MR, Thota S, et al. (2021) Epidural Injections for Lumbar Radiculopathy or Sciatica: A Comparative Systematic Review and Meta-Analysis of Cochrane Review. Pain Physician 24(5): E539-E554.
- House LM, Barrette K, Mattie R, McCormick ZL (2018) Cervical Epidural Steroid Injection: Techniques and Evidence. Phys Med Rehabil Clin N Am 29(1): 1-17.
- Stout A (2011) Epidural steroid injections for cervical radiculopathy. Phys Med Rehabil Clin N Am 22(1): 149-59.
- Manchikanti L, Nampiaparampil DE, Candido KD, Bakshi S, Grider JS, et al. (2015) Do cervical epidural injections provide long-term relief in neck and upper extremity pain? A systematic review. Pain Physician 18(1): 39-60.
- Diwan S, Manchikanti L, Benyamin RM, Bryce DA, Geffert S, et al. (2012) Efficacy of cervical epidural injections in the management of chronic neck and upper extremity pain. Pain doctor 15(4): E405-434.
- Quraishi NA (2012) Transforaminal injection of corticosteroids for lumbar radiculopathy: systematic review and meta-analysis. Eur Spine J 21(2): 214-219.
- MacVicar J, King W, Landers MH, Bogduk N (2013) The effectiveness of lumbar transforaminal injection of steroids: a comprehensive review with systematic analysis of the published data. Pain Med 14(1): 14-28.
-
Juan Ulises Villanueva Morales1*, Ricardo Erick Robles Ortíz et al. Corticosteroids in Spine Surgery. Glob J Ortho Res. 5(3): 2026. GJOR.MS.ID.000614..
-
Corticosteroids, Spinal cord injury, Spinal cord compression, Low back pain, Cervical surgery, Spinal infiltration, Spinal tumors, Dexamethasone treatment
-

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.
- Abstract
- Introduction
- Corticosteroid physiology: hypothalamic-pituitaryadrenal axis
- Pharmacology of Steroids
- Mechanism Of Action
- Pharmacokinetics and Pharmacodynamics
- Acute Spinal Cord Injury
- Adverse Reactions
- Spinal Tumors
- Adverse Reactions
- Spinal Cord Compression Without Neurological Involvement
- Cervical Surgery: Discectomy and Anterior Cervical Fusion
- Topical Steroid Use
- Low Back Pain
- Spinal Infiltration of Steroids
- Lumbar Interlaminar Epidural Infiltration
- Cervical Epidural Infiltration
- Transforaminal Lumbar Infiltration
- Conclusion
- Acknowledgements
- Conflict of Interest
- References






