 Research Article
Research Article
Effects of Processing on the Antioxidant Potential of Brown and Gold Flaxseed Extracts
Rajwinder Kaur1, Shakaree Hale1, Harpreet Singh1, Shantrell Willis1, Martha Verghese1*
1Department of Food and Animal Sciences, Alabama Agricultural and Mechanical University, 4900 Meridian St. N, Normal, AL 35762
Martha Verghes, Department of Food and Animal Sciences, Alabama Agricultural and Mechanical University, 4900 Meridian St. N, Normal, AL 35762
Received Date: June 18, 2024; Published Date:June 18, 2024
Abstract
Flaxseed (Linum usitatissimum) is rich in bioactive compounds, which may play a role in disease prevention. Flaxseed has emerged as a functional food ingredient because of its effect on human health, especially its chemo-preventative potential. This study aimed to determine the impact of processing on brown and gold flaxseeds’ phytochemical content and antioxidant capacity. Flaxseeds were extracted with deionized water (AQ) and 80% methanol (ME). Total phenolic content (TPC), total flavonoid content (TFC), free radical scavenging ability using 2,2-diphenyl-1-picrylhydrazyl radical (DPPH), ferric reducing antioxidant power (FRAP), oxygen radical absorbance capacity (ORAC), Trolox equivalent antioxidant capacity (TEAC), and nitric oxide radical scavenging (NORS) were evaluated in flax seed extracts. Flaxseed extracts used for this study were golden raw/ whole (GRW), golden raw/ground (GRG), golden roasted/whole (GROW), golden roasted/ground (GROG), golden roasted/defatted/whole (GRDW), golden roasted/defatted/ground (GRDG), golden defatted/whole (GDW), golden defatted/ground (GDG), brown raw/whole (BRW), brown raw/ ground (BRG), brown roasted/whole (BROW), brown roasted/ground (BROG), brown roasted/defatted/whole (BRDW), brown roasted/defatted/ ground (BRDG), brown defatted/whole (BDW), brown defatted/ground (BDG). TPC (mg GAE/100g) was highest in GDG extracts (53.13) and lowest in BRW (2.03). TFC (mg CE/100 g) was highest in GDG (901.11) and lowest in GRW (27.55). DPPH scavenging ability was highest in GRDG (98.56) and lowest in GRW (33.07). FRAP (mMol Fe+2/g) ability was highest in GRDG (10.16) and lowest in GRW (0.37). TEAC (μg T.E./g) was highest in GDG (579.17) and lowest in BRW (7.48). NORS (μg A.A./100g) was highest in BDG (290.27) and lowest in BRW (1.13). ORAC (μg T.E./g) was highest in GRDG extracts (32,473.95) and lowest in GRW (19,079.02). Results showed significant differences in various extracts of flax after processing. This study suggests that processing may improve the health benefits of flaxseed with implications for functional food product development. Results showed significant differences in various extracts of flax after processing.
Abbreviations: Aqueous- AQ; Methanol- ME; Total phenolic content- TPC; Total flavonoid content- TFC; 2,2-diphenyl-1-picrylhydrazyl radical- DPPH; ferric reducing antioxidant potential- FRAP; oxygen radical absorbance capacity- ORAC; Trolox equivalent antioxidant capacity- TEAC; nitric oxide radical scavenging- NORS; golden raw/whole- GRW; golden raw/ground- GRG; golden roasted/whole- GROW; golden roasted/ground- GROG; golden roasted/defatted/whole- GRDW; golden roasted/defatted/ground- GRDG; golden defatted/whole- GDW; golden defatted/ground- GDG; brown raw/whole- BRW; brown raw/ground- BRG; brown roasted/whole- BROW; brown roasted/ground- BROG; brown roasted/defatted/whole- BRDW; brown roasted/defatted/ground- BRDG; brown defatted/whole- BDW; brown defatted/ground- BDG; Defatted- DF; Roasted & Defatted- R&DF.
Introduction
Phytochemicals, from the Greek word “phyto meaning,” “plant,” are chemical compounds naturally found in plants, herbs, spices, fruits, vegetables, seeds, and nuts. Plants naturally produce these compounds; in some cases, secondary metabolites are produced in response to stress or attack, making phytochemicals an essential part of plant defense mechanisms [1]. In some plants, various phytochemicals are responsible for plant characteristics such as color or flavor. Phytochemicals are classified into compounds that include flavonoids, isoflavonoids, and antioxidants [2]. Humans are constantly exposed to exogenous factors such as environmental toxins and ultraviolet rays, leading to oxidative stress. Normal cellular metabolism produces reactive oxygen species (ROS) and free radicals essential in cell signaling pathways [3]. However, an imbalance can damage important biomolecules and cells and the development of cancer [4]. Flax is one of the oldest crops cultivated in the United States, originating in ancient Egypt [5]. A member of the Linaceae family, flax is scientifically known as Linum Usitatissimmum, meaning “most useful” (Amin & Thakur, 2014). The seeds obtained from the flax plant are known as flaxseeds or linseed. Flaxseeds are flat oval-shaped and have two primary varieties: brown and gold. Flaxseed is grown worldwide as an oil or fiber crop, with Canada being the largest producer of flaxseed [7]. Flax is rich in fats (omega-3 fatty acids), lignans, dietary fiber, and antioxidants [8]. Flaxseed continues to increase in interest due to its anticarcinogenic properties [10]. Flaxseed can be incorporated into several foods and served as functional ingredients in baked goods. α- Linolenic acid (ALA) and lignans present in flaxseed could potentially contribute to the chemo-preventive and anti-inflammatory function associated with the consumption of flaxseed (Marghescu, 2012). Commercial flaxseed processing includes seed cleaning, flaking, cooking, defatting, solvent extraction, and removal. The amount of heat used in the processing of flaxseeds can cause physiochemical changes and influence the nutritional quality of the final product. Roasting has been shown to increase the porosity of flaxseeds, allowing for better extractability of antioxidants (Waszkowiak et al., 2015), and defatting increases protein and carbohydrate content (Oomah & Giuseppe, 1998). However, in terms of stability, it is essential to determine if it is feasible to incorporate flaxseed into baked food products.
Therefore, the objective of the present study was to determine the effects of processing on phytochemical content and antioxidant potential of brown and gold flaxseeds. Total phenolic content, total flavonoid content, free radical reducing ability (1,1- Diphenyl – 2- picrylhydrazyl), ferric reducing antioxidant power (FRAP), trolox equivalent antioxidant capacity (TEAC), nitric oxide radical assay, 2,2’ – azinobis (3-ethylbenzothiazoline-6-sulfonate) (NORS), and oxygen radical absorbance capacity (ORAC) was determined of methanol and hot water extractions of raw, roasted and defatted brown and gold flaxseed.
Materials and Methods
Chemicals and reagents
Eighty percent methanol (Fisher Scientific), hexane (Fisher Scientific), Folin-Ciocateu reagent (MP Biomedicals), 7% sodium carbonate (Fisher Scientific), ferulic acid (Fisher Scientific), 5% sodium nitrite (Fisher Scientific), 10% aluminum chloride (Fisher Chemical), 1M sodium hydroxide (Fisher Scientific), catechin (Fisher Scientific), acetate buffer (sodium acetate, acetic acid glacial), 1,1-diphenyl-2-picrylhydrazyl (DPPH) radical (Fisher Scientific), 10 mM 2,4,6-tri(2-pyridyl)-s-triazine (TPTZ) (Fisher Scientific), 40 mM hydrochloric acid (Fisher Chemical), 20 mM ferric chloride (Fe- Cl3.6H2O), ferrous sulfate (Fisher Scientific), 20mM fluorescin, 2,2 azobis 2-amidinopropane dihydrochloride (AAPH), 7 mM 2,2’- azinobis (3- ethylbenzothiazoline 6- sulfonate) radical cation, trolox (Fisher Scientific), potassium persulfate (Fisher Scientific), 10mM sodium nitroprusside (Fisher Scientific), Griess reagent (Fisher Scientific).
Instrumentation
All experiments were carried out using a conventional oven (Isotemp Oven, Fisher Marietta, OH, model 6925), a Black and Decker coffee grinder, Thermo Scientific Orbital shaker, Branson Sonicator, Buchi Vacuum Controller Rotavapor, Thermo Scientific Legend XTR Centrifuge, and Biotek Synergy HT Microplate reader (Highland Park Winooski, VT).
Flaxseed material
Brown and gold flaxseeds were purchased from a local health food store in Huntsville, Alabama, United States. Flaxseeds were stored at 2°C until further use.
Flaxseed roasting
Brown and gold flaxseeds were roasted in a conventional oven (Isotemp Oven, Fisher Marietta, OH, model 6925) at 482°F (250°C) for three minutes. Flaxseeds were allowed to reach room temperature before grinding. Whole seeds were ground using a coffee grinder (Black and Decker).
Defatting
Defatting was performed using hexane. Five grams of flaxseeds were soaked in hexane with continuous shaking for four hours, followed by sonication (Branson 5800, Branson Ultrasonics Corporation, Danbury, CT 06810, USA). Defatted seeds were centrifuged at 10,000 rpm for 10 minutes. The supernatant was filtered using Whatman filter paper No.1 and stored at -80°C until further use. The defatted flaxseed residue was stored at -80°C until used for methanol and aqueous extractions.
Methanol and aqueous extractions
Methanol (ME) and aqueous extracts were performed using 80% methanol and deionized water heated to 60°C. Five grams of flaxseed were soaked in 80% methanol and hot water (60°C) with continuous shaking for two hours, followed by sonication. Extracts were centrifuged at 10,000 rpm for 10 minutes. The supernatant was filtered using Whatman filter paper No. 1 and evaporated to dryness using a rotary evaporator (Buchi Rotavapor R-215, US). The concentrate was reconstituted and stored at -80°C until further use.
Determination of Total Phenolic Content
The total phenolic content of brown and gold flaxseeds was determined by the Folin-Ciocalteu colorimetric method [11]. The absorbance was measured against a blank (distilled water) at 760 nm and compared to a ferulic acid standard curve. Ferulic acid was used instead of gallic acid because it is more predominant in flaxseeds. The results are expressed as mean (mg ferulic acid equivalents/ 100 ml) ± SD for three replications.
Determination of Total Flavonoid Content
The aluminum colorimetric method determined the total flavonoid content of brown and gold flaxseed (Sun & Lui, 2006) [12]. The absorbance was measured against a blank (distilled water) at 510 nm and compared to a flavone-C standard curve. Flavone-C was used instead of catechin because it is more predominant in flaxseeds. The absorbance was measured at 510 nm. Results are expressed as mg FCE/100 ml.
1,1-diphenyl-2-picrylhydrazyl (DPPH) radical scavenging assay
The antioxidant activity of brown and gold flaxseed was evaluated by 1-diphenyl-2-picrylhydrazyl (DPPH) radical. The absorbance was read at 517 nm. Samples were expressed in terms of IC50 (concentration required for a 50% decrease in absorbance of DPPH, radical) relative to the control (100%).
Ferric Reducing Antioxidant Power (FRAP)
The ability of brown and gold flaxseed to reduce ferric iron to ferrous iron was evaluated by the ferric reducing antioxidant power (FRAP) assay as described by Benzie and Strain (1996). The absorbance was read at 590 nm. The absorbance is compared to the ferrous sulfate standard (0.1mM1 – 0mM).
Oxygen Radical Absorbance Capacity (ORAC)
ORAC of brown and gold flaxseed was determined according to the protocol suggested by Huang et al., 2002. The fluorescence decay rate was examined using a microplate reader, and calculations were made using the area under the fluorescent decay curve and a Trolox standard curve.
Trolox Equivalent Antioxidant Capacity (TEAC) Determination
The Trolox equivalent antioxidant capacity of brown and gold flaxseed was determined according to the protocol suggested by Rice-Evans Miller, 1994. Absorbance was read at 734 nm.
Nitric Oxide Radical Scavenging Activity (NORS)
The nitric oxide radical scavenging activity (NORS) of brown and gold flaxseed was determined (Ebrahimzadeh, Nabavi, Bahramian, & Bekhradnia, 2010). The absorbance was read at 546 nm.
Results and Discussion
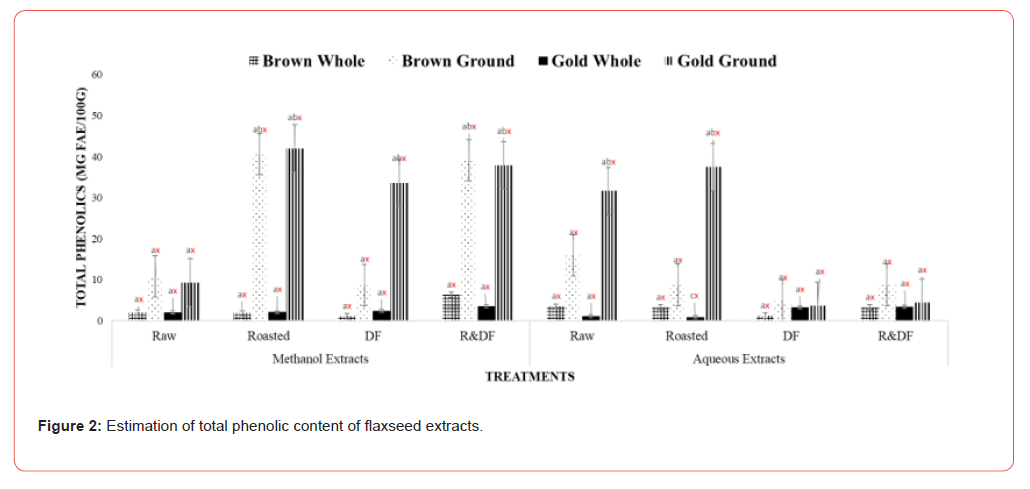
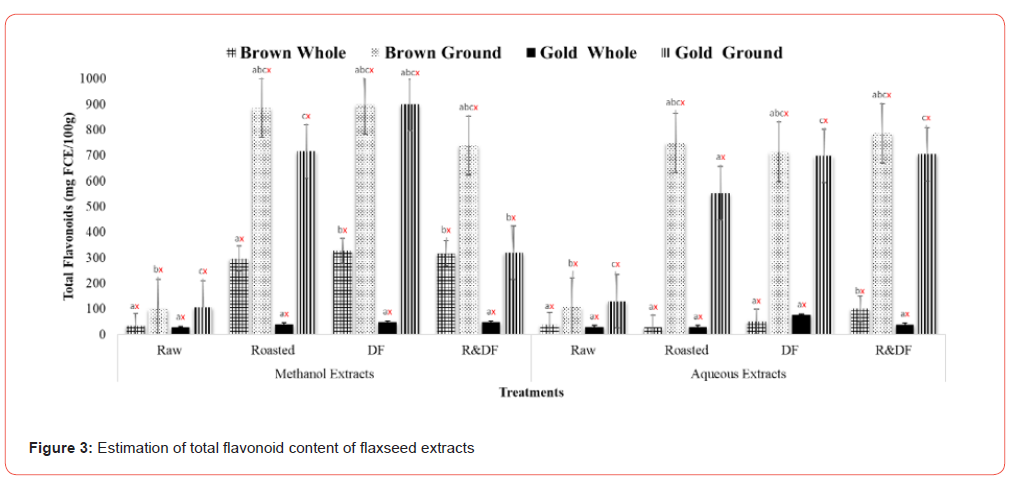
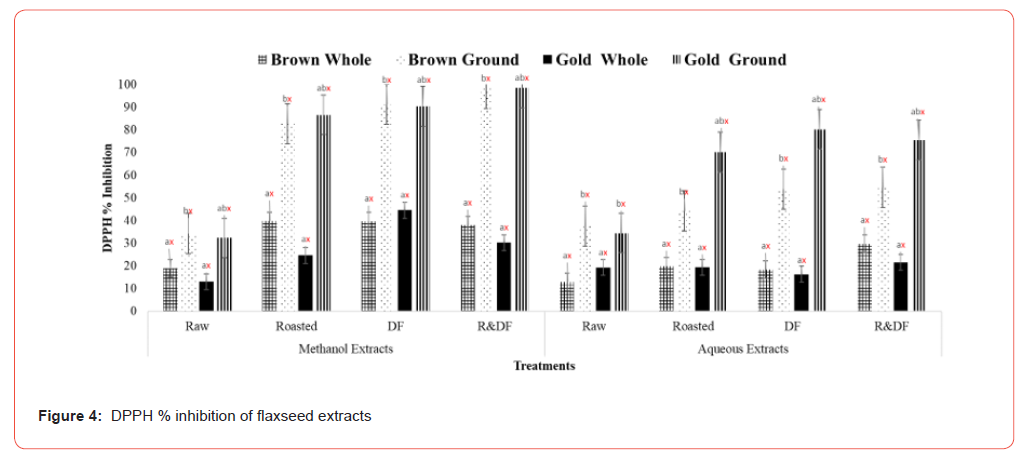
The effects of processing on the phytochemical and antioxidative activities of brown and gold flaxseed extracts were determined using in vitro methods. Results of total phenolic content (Figure 1) show significant differences between ground, roasted, and defatted flaxseed extracts when compared to unroasted and whole-fat flaxseed extracts. There are no significant differences between varieties. This indicates that processing methods such as roasting can potentially improve the extractability of polyphenolic compounds by increasing the porosity of the flaxseed [12]. Flaxseeds comprise a class of phenolic compounds known as lignans, which can act as antioxidants and phytoestrogens. These lignans have weak estrogenic effects, which are beneficial in reducing the growth of cancerous tumors, especially hormone-sensitive ones such as breast and prostate [14-16]. Flaxseed lignan secoisolariciresinol diglucoside (SDG) has also reduced blood vessel cell formation in female rats [17]. One study showed that when type 2 diabetes patients were fed defatted flaxseeds for two months, patients showed a significant reduction of plasma glucose and lipid peroxidation [18]. Total flavonoid content results (Figure 2) showed significant differences between ground, roasted, and defatted flaxseed extracts compared to whole, unprocessed extracts. There were no significant differences between brown and gold varieties. Results of DPPH % inhibition (Figure 3) shows significant differences between ground roasted and defatted flaxseed extracts when compared to raw, whole-fat flaxseed extracts. These results coincide with Waszkowiak et al., 2015 suggesting that roasted flaxseeds show higher antioxidant potential compared to raw.
The antioxidant activity of flaxseed has been shown to reduce total cholesterol, triglyceride levels, and platelet aggregation, which can have implications in patients with a higher risk of cardiovascular disease [19-21] investigated the anti-atherogenic capacity of flaxseed in an animal model. Results from this study indicated that supplementation of a cholesterol-enriched diet with ground flaxseed lowered plasma cholesterol and saturated fatty acids, increased plasma content of α-linoleic acid, and inhibited plaque formation in the aorta and aortic sinus compared with mice fed a diet supplemented with only dietary cholesterol [22]. This study suggests dietary flaxseed can inhibit atherosclerosis by reducing circulating cholesterol levels [23,24]. Results show a more significant ferric-reducing antioxidant potential (Table 1) of roasted and defatted flaxseed extracts compared to unprocessed extracts. Ground flaxseed results in greater antioxidant activity, which can benefit patients with type 2 diabetes and hypertension. Caligiuri et al., 2014 examined the effects of flaxseed consumption on altering plasma oxylipins in a manner that influenced blood pressure in patients with hypertension. This study suggests that α- linolenic acid in flaxseed may have inhibited soluble epoxide hydrolase, which altered oxylipin concentrations that contributed to the antihypertensive effects in patients with peripheral arterial disease [25].
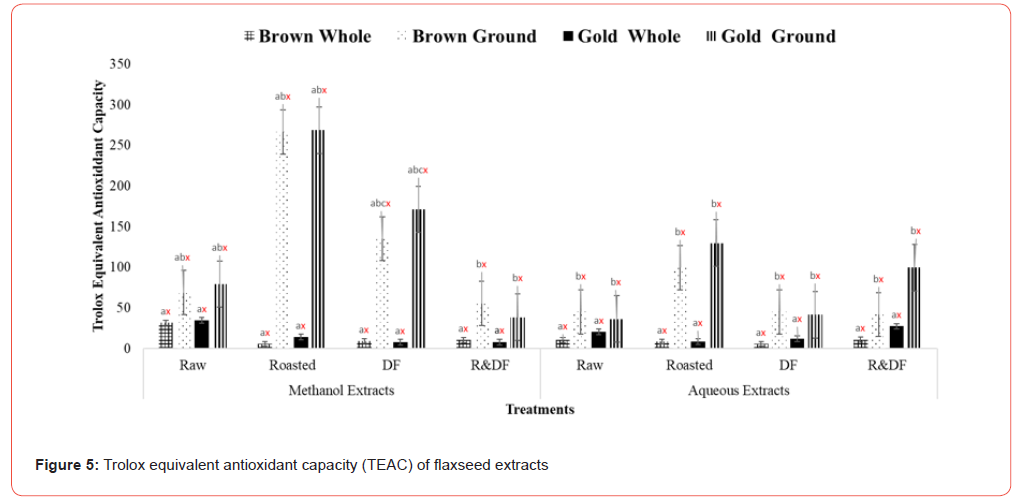
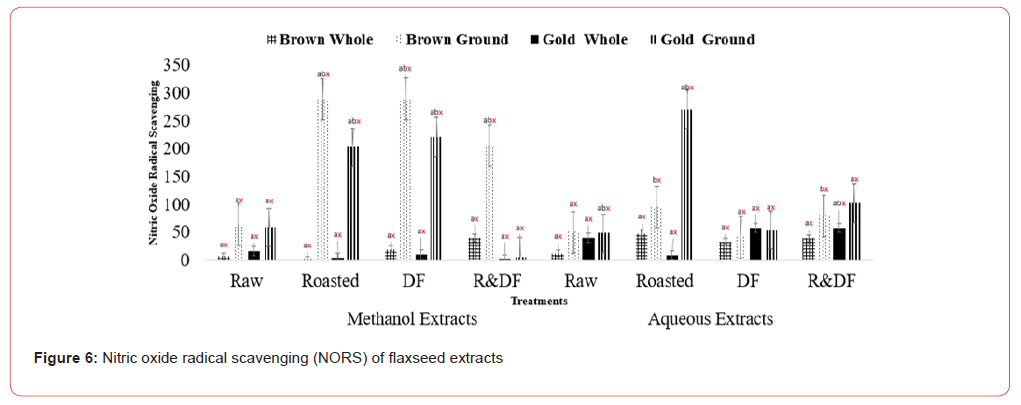
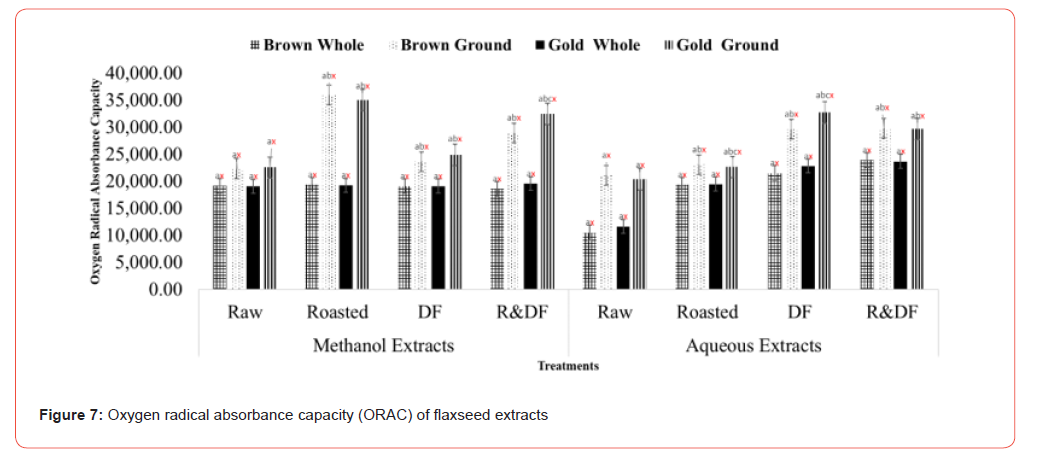
Results of Trolox equivalent antioxidant capacity (Figure 4), nitric oxide radical scavenging (Figure 5), and oxygen radical absorbance capacity (Figure 6) show significant differences between ground roasted and defatted flaxseed extracts when compared to raw, whole-fat flaxseed extracts. There were no significant differences between brown and gold varieties of flaxseed (Figure 7).
This study revealed that ground, roasted, and/or defatted flaxseed extracts yielded a significantly higher antioxidant potential when compared to whole, unprocessed flaxseed extracts. Results showed no significant differences in TF and TP contents and antioxidant potential of brown and gold flaxseed extracts. However, significant differences between whole and ground and whole and roasted flaxseeds. Results indicate that flaxseed constituents are stable during processing. This study suggests that processing may impact the health effects of flaxseed with industry applications and implications in functional food product development.
Values (n=3) are means ± SEM. Bars with subscripts (abc) represent differences amongst treatments. Bars with subscripts (xyz) represent differences between varities. Bars with superscripts (abc/xyz) without a common letter differ significantly p ≤ 0.05. Abbreviations: FAE – Ferulic acid equivalents, DF- defatted, R&DFroasted/ defatted.
Values (n=3) are means ± SEM. Bars with subscripts (abc) represent differences amongst treatments. Bars with subscripts (xyz) represent differences between varities. Bars with superscripts (abc/xyz) without a common letter differ significantly p ≤ 0.05. Abbreviations: FA – Flavone-C equivalents, DF- defatted, R&DF- roasted/ defatted.
Values (n=3) are means ± SEM. Bars with subscripts (abc) represent differences amongst treatments. Bars with subscripts (xyz) represent differences between varities. Bars with superscripts (abc/xyz) without a common letter differ significantly p ≤ 0.05. Abbreviations: DPPH - 1,1-diphenyl, 2-picrahydrazyl, DF- defatted, R&DF- roasted/defatted.
Table 1:Ferric reducing antioxidant potential (FRAP) of flaxseed extracts
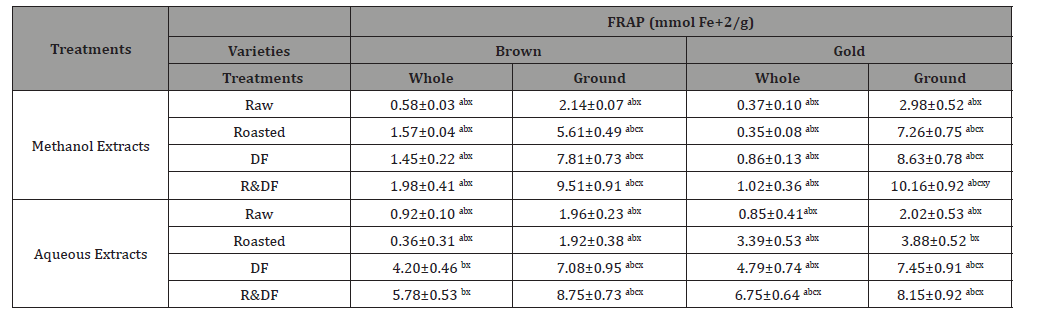
Values (n=3) means ± SEM. Superscripts (abc) represent differences amongst treatments which are shown in rows. Bars with superscripts (xyz) represent differences between varities. Bars with superscripts (abc/xyz) without a common letter differ significantly p ≤ 0.05. Abbreviations: FRAP, ferric reducing antioxidant potential, DF- defatted, R&DF- roasted/defatted.
Values (n=3) are means ± SEM. Bars with subscripts (abc) represent differences amongst treatments. Bars with subscripts (xyz) represent differences between varities. Bars with superscripts (abc/xyz) without a common letter differ significantly p ≤ 0.05. Abbreviations: TEAC - Trolox equivalent antioxidant capacity, DF- defatted, R&DF- roasted/defatted.
Values (n=3) are means ± SEM. Bars with subscripts (abc) represent differences amongst treatments. Bars with subscripts (xyz) represent differences between varities. Bars with superscripts (abc/xyz) without a common letter differ significantly p ≤ 0.05. Abbreviations: A.A. – Ascorbic acid, DF- defatted, R&DF- roasted/ defatted.
Values (n=3) are means ± SEM. Bars with subscripts (abc) represent differences amongst treatments. Bars with subscripts (xyz) represent differences between varities. Bars with superscripts (abc/xyz) without a common letter differ significantly p ≤ 0.05. Abbreviations: DF- defatted, R&DF- roasted/defatted.
Application Of Real Samples And Conclusions
In recent years, consumers have become increasingly aware of food ingredients and the implications between diet and health [26]. The rise in obesity and chronic diseases has become a national public health concern and scientists are now turning to food and food constituets to help combat these concerns. Since flaxseeds are rich in lignans and antioxidants, the seeds may have anti-cargenogenic activity and can be used to develop funtional food products [27]. This study was adapted to determine the amount of phenolic compounds present; which may be responsible for the chemo-preventive effects of flaxseed and also to investigate the effects of processing on phytochemicals. Since flaxseeds are often subjected to heat processing and grinding when incoporated into foods, it is essential to understand the effects on the stability of these active compounds. This study showed promising results, as we continue to investigate the functinality of flaxseeds in foods.
Acknowledgement
None
Conflicts of Interests
No conflicts of interest.
References
- Almario RU, Karakas, SE (2013) Lignan content of the flaxseed influences its biological effects in healthy men and women. Journal of the American College of Nutrition32(3): 194-199.
- Amin T, Thakur M (2014) Linum usitatissimum L. (Flaxseed) A Multifarious Functional Food. Online Int. Interdisciplinary Res. IV (I): 220-238.
- Azrad M, Vollmer RT, Madden J, Dewhirst M, Polascik TJ, et al. (2013) Flaxseed-derived enterolactone is inversely associated with tumor cell proliferation in men with localized prostate cancer. Journal of Medicinal Food, 16(4): 357-360.
- Azrad M, Turgeon CE, Demark-Wahnefried W (2013) Current evidence linking polyunsaturated fatty acids with cancer risk and progression. Frontiers in Oncology3:
- Bernacchia R, Preti R, Vinci G (2014) Chemical composition and health benefits of flaxseed. Austin J Nutri Food Sci 2(8): 1045.
- Benzie IF, Strain JJ (1996) The ferric reducing ability of plasma (FRAP) as a measure of “antioxidant power”: the FRAP assay. Analytical Biochemistry239(1): 70-76.
- Bhaskaran S, Hardley F (2002) Buyer beliefs, attitudes and behaviour: foods with therapeutic claims. Journal of Consumer Marketing19(7): 591-606.
- Blomhoff R, Blomhoff HK (2006) Overview of retinoid metabolism and function. Developmental Neurobiology, 66(7): 606-630.
- Brand-Williams W, Cuvelier ME, Berset CLW T (1995) Use of a free radical method to evaluate antioxidant activity. LWT-Food science and Technology28(1): 25-30.
- Caligiuri SP, Aukema HM, Ravandi A, Guzman R, Dibrov E, et al. (2014) Flaxseed consumption reduces blood pressure in patients with hypertension by altering circulating oxylipins via an α-linolenic acid-induced inhibition of soluble epoxide hydrolase. Hypertension 64: 53-59.
- Dupasquier CM, Dibrov E, Kneesh AL, Cheung PK, Lee KG, Alexander HK, et al. (2007) Dietary flaxseed inhibits atherosclerosis in the LDL receptor-deficient mouse in part through antiproliferative and anti-inflammatory actions. See comment in PubMed Commons below Am J Physiol Heart Circ Physiol. 293: H2394-2402
- Ebrahimzadeh MA, Nabavi SM, Nabavi SF, Bahramian F, Bekhradnia AR (2010) Antioxidant and free radical scavenging activity of H. officinalis L. var. angustifolius, V. odorata, B. hyrcana and C. speciosum. Pak J Pharm Sci23(1): 29-34.
- Ganorkar PM, Jain RK (2013) Flaxseed--a nutritional punch. International Food Research Journal, 20(2).
- Goyal A, Sharma V, Upadhyay N, Gill S, Sihag M (2014) Flax and flaxseed oil: an ancient medicine & modern functional food. Journal of Food Science and Technology51(9): 1633-1653.
- Kanter M (1998) Free radicals, exercise and antioxidant supplementation. Proceedings of the Nutrition Society57(1): 9-13.
- Leyva DR, Zahradka P, Ramjiawan B, Guzman R, Aliani M, et al. (2011) The effect of dietary flaxseed on improving symptoms of cardiovascular disease in patients with peripheral artery disease: rationale and design of the FLAX-PAD randomized controlled trial. Contemporary clinical trials, 32(5): 724-730.
- Lindahl T, Baghaei F, Fagerber Blixter I, Gustafsson K, Stigendal L, et al. (2011) Effects of the oral, direct thrombin inhibitor dabigatran on five common coagulation assays. Thrombosis and Haemostasis, 105(2): 371-378.
- Ou B, Huang D, Hampsch-Woodill M, Flanagan JA, Deemer EK (2002) Analysis of antioxidant activities of common vegetables employing oxygen radical absorbance capacity (ORAC) and ferric reducing antioxidant power (FRAP) assays: a comparative study. Journal of Agricultural and Food Chemistry50(11): 3122-3128.
- Rajesha J, Murthy KNC, Kumar MK, Madhusudhan B, Ravishankar GA (2006) Antioxidant potentials of flaxseed by in vivo model. Journal of Agricultural and Food Chemistry54(11): 3794-3799.
- Rhee Y, Brunt A (2011) Flaxseed supplementation improved insulin resistance in obese glucose intolerant people: a randomized crossover design. Nutrition journal10(1): 44.
- Rice-Evans C, Miller NJ (1994) [241 Total antioxidant status in plasma and body fluids. In Methods in Enzymology 234: 279-293. Academic Press.
- Robert I, Lin S (1994) Phytochemicals and antioxidants. In Functional FoodsSpringer, Boston, MA pp. 393-449.
- Saggar JK, Chen J, Corey P, Thompson LU (2010) The effect of secoisolariciresinol Di glucoside and flaxseed oil, alone and in combination, on MCF-7 tumor growth and signaling pathways. Nutrition and Cancer62(4): 533-542.
- Shabbira MA, Khana MR, Shehzada A, Dinb A, Khana MI, et al. (2014) Flaxseed segregations as an imperative tool for its neutraceutical implication. Pakistan Journal of Food Sciences, 24(2): 64-74.
- Singleton VL, Orthofer R, Lamuela-Raventós RM (1999) [14] Analysis of total phenols and other oxidation substrates and antioxidants by means of folin-ciocalteu reagent. In Methods in Enzymology299: 152-178. Academic press.
- Sun J, Liu RH (2006) Cranberry phytochemical extracts induce cell cycle arrest and apoptosis in human MCF-7 breast cancer cells. Cancer Letters241(1): 124-134.
- Thompson LU, Chen JM, Li T, Strasser-Weippl K, Goss PE (2005) Dietary flaxseed alters tumor biological markers in postmenopausal breast cancer. Clinical Cancer Research, 11(10): 3828-3835.
- ISSN: 2644-2981 4.05.000604
- Volume 5 - Issue 3, 2025
- Open Access
-
Rajwinder Kaur, Shakaree Hale, Harpreet Singh, Shantrell Willis, Martha Verghese*. Effects of Processing on the Antioxidant Potential of Brown and Gold Flaxseed Extracts. 5(1): 2024. GJNFS.MS.ID.000604
-
Flaxseed, Gold Flaxseed Extracts, phyto meaning, plants, bioactive compounds
-

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.






