 Research Article
Research Article
Efficiency of Different Foaming Agents for Enhanced Oil Recovery: Bulk and Dynamic Flooding Screening
El Mahdy Osama1*, Al Quraishi Abdulrahman A2 and Belhaij Azmi M1
1Petroleum and Natural Gas Engineering Department, College of Engineering, King Saud University, Saudi Arabia
2National Center for Oil & gas Technology, King Abdulaziz City for Science and Technology, Saudi Arabia
El Mahdy Osama, Petroleum and Natural Gas Engineering Department, College of Engineering, King Saud University, P.O. Box 800, Riyadh 11421, Saudi Arabia.
Received Date: November 28, 2019; Published Date: December 04, 2019
Abstract
Foamability and foam stability are of main concerns in foam displacement for enhanced oil recovery. This work presents the output of systematic bulk and Dynamic screening of several foaming agents (surfactants). Solution salinity and oil presence effects were explored in static bulk tests. The most effective foaming agents were then investigated further through dynamic flooding in porous medium. The results indicate the foamability of all surfactants except for Triton X-405. Zonyl FSO and Hitenol H-10 were superior in term of foam stability with more stability as surfactants concentration increases. Equivalent optimum foam volumes were obtained for both surfactants but at higher concentrations of Hitenol H-10. Increasing solution salinity affected Zonyl FSO foam stability negatively at low concentration. Observation indicates that oil stayed in the lamella’s skeleton and plateau boarders with no drain out when Zonyl FSO solution was used. To the contrary, Hitenol H-10 was able to lift good portion of oil column but oil was drained out of the foam structure within a short period of time. Dynamic tests in porous medium indicate relatively better performance for Hitenol H-10 over Zonyl FSO as indicated by the increase in pressure drop and delayed gas breakthrough time.
Keywords: Foam; Agent; Oil recovery; Foam stability; Dynamic flooding
Introduction
Gas flooding is a common mean of improving oil recovery. Gases have high mobility due to their low viscosity and density compared to reservoir fluids causing gas to bypass and override the resident oil leading to inefficient sweep. Foam injection was first introduced by Bond and Holbrook [1] to overcome the high mobility associated with gas drive. Later, it was proved that foam can act as a gasblocking agent [2].
Foams are usually formed in systematic hexagonal texture due gas dispersion through a continuous surfactant solution [3]. Foams are generally described in terms of their foamability and foam stability describing the variations of foam height or volume with time, immediately after foam generation [4]. The two terms are interrelated and the more stable the foam films, the greater is the solution’s foamability.
Foam stability is dependent on electric double-layer repulsion [5,3], gravity drainage, capillary suction, surface elasticity, dispersion force attraction, steric repulsion and surface and bulk viscosity [3]. One of the main parameters to determine the success of foam flooding is the selection and concentration of surfactant used as foaming agents. Accordingly, the careful investigation of surfactant type and concentration is necessary for strong bulk foam generation. Foam generation generally increases with increasing surfactant concentration up to the critical micelle concentration (CMC) above which surfactant concentration has little effect [6]. Marsden and Khan [7] indicated that apparent viscosity increases with the increase of surfactant concentration.
Foam stability in presence of oil in bulk and flooding through porous medium scales is related to foam-oil interaction. Literatures indicated the role of oil presence on foam stability [3,8-11] and foams in presence of oil behave distinctly and the choice of surfactant is important for the success of the process [12-14]. Three major mechanisms have been considered for the antifoaming property of the dispersed oils. These are aqueous film thinning rate during oil entry, oil spreading on the water surface, and thin water film bridging [15,16]. Simjoo et al. [17] suggested that foam decay in presence of oil starts as small and rapid decay dominated by gravity drainage, followed by a stabilized foam volume, and then a second continuous decay due to bubble coalescence over a relatively long time. The oil foam destabilization is more pronounced for oils with a smaller molecular weight.
Salinity has been proved to be one of the main factors affecting foam stability. Prior investigations indicate that more stable foam can be generated with low salinity solutions and the effect of salinity is more pronounced in presence of oil [18]. Stability ensured by a disjoining pressure at the lamella between two foam bubbles is directly dependent on the concentration of surfactants and salt that are used in foam flooding in porous media [19].
Bulk foam test is an easy and quick mean to determine the formability and foam stability as opposed to time consuming dynamic foam flooding. Multiple investigations found no correlation between the results of the two methods in presence of oil [14,20], however, a correlation was observed in absence of oil [14]. In this work we investigated the bulk foamability and foam stability of several surfactants at different surfactant concentrations and different solution salinities in presence and absence of crude oil. The bulk tests were based on foam column height. Flooding tests were further conducted to correlate foam performance in porous medium with that obtained using the static tests. The flooding was performed in absence and presence of crude oil but only those in absence of oil will be presented in this article.
Experimental Procedure
The application of the t1/2 parameter for metastable foams comparison can be time consuming. Lunkenheimera and Malysa [21] proposed a parameter called R5 to examine the foam stability of the surfactant solutions. Accordingly, this parameter was used to screen the foaming agents in bulk tests. R5 parameter is determined as the ratio of the foam height at five minutes after foaming (h5) to the initial formed foam height (h0). Accordingly, foams with R5 of 50% are considered as metastable, whereas lower values indicate foams of low stability.
The setup used for bulk tests consists of a glass column 100cm long and 1.6cm inner diameter equipped with glass frit placed at the base of the column. Foam was generated by injecting gas into the solution using a syringe pump for gas supply. The experiments were conducted by slowly pouring surfactant solution (12.5 mL) at two different salinity levels into the glass column. Carbon Dioxide gas was introduced at predetermined rate into solution and when gas injection ceases, the stopcock connecting the column with the syringe pump is immediately closed, and the initial foam and solution heights are measured. Foam height and solution level are then measured in five minutes time in order to determine the R5 parameter.
The foam stability was investigated in presence of oil by adding oil to 4% salinity surfactants solutions before gas injection. The amount of oil was calculated as a volume fraction of the surfactant solution and 5.0 % by volume is poured into the column. All bulk measurements were repeated 2 to 3 times for every solution concentration at ambient conditions of pressure and temperature.

Based on the bulk screening, the best performance surfactants were tested in porous medium saturated with brine of 4 % salinity. Baseline CO2 gas injection and CO2 foam flooding were conducted in fresh core sample for each run. A schematic of the flooding unit is shown in Figure 1. Immiscible CO2 was injected into the core at constant flow rate of 0.6 to 0.7cc/min (Same flow rate was used for all experiments) until brine production ceases. At the end, the core was re-saturated with several pore volumes of brine from both ends to displace CO2 and restore the core to its original state where complete saturation was checked with permeability measurements. Surfactants were pre-injected at either 0.5 or 1.0 pore volumes slug sizes. The process was started by injecting a predetermined slug of chosen surfactant solution at flow rate of 1.0 cc/min (Same flow rate was used for all experiments) followed by continuous injection of CO2 gas until brine production ceases. The produced fluid was collected in digital mass balance and mass of recovered brine, injection rate of both gas and surfactant solution and overall pressure drop across the core sample were monitored via data acquisition system connected to personal computer. Recovery and pressure drop were used to test the in-situ generated foam efficiency in controlling gas mobility and improving displacement efficiency as evaluated by the apparent foam viscosity and mobility reduction factor (MRF) defined as the pressure drop encountered during foam flooding to that encountered during gas flooding.
Materials
Several surfactants were investigated. These surfactants are classified as nonionic (Triton X-100, Triton X-405, Zonyl FSO, Noigen N-10 and Noigen N-20) and anionic Hitenol H-10. Surfactant solutions of different concentrations were prepared by diluting them in brines of 4% and 10% salinities (Table 1).
Table 1: Composition of 4.19 % brine.
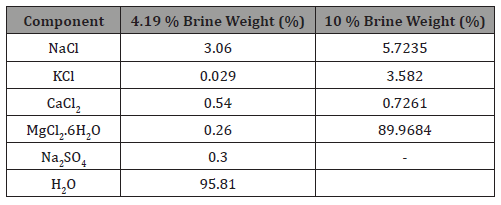
Table 1 lists the compositions of brine solutions. Arab crude oil (23 API, 90 cp) was used and high purity Carbon Dioxide as oleic and gas phases. Dynamic foam flooding was conducted in Berea core samples 10.1 cm long and 3.8 cm diameter with properties listed in Table 2.
Table 2: Petrophysical properties and summary of obtained results.

Results and Discussion
Bulk foam tests
To investigate the foam stability during the foam formation, each surfactant concentration was introduced to gas at average volumetric rates of 4.5 and 9 L/hr. Figure 2 is an example plot of initial foam height of Hitenol H-10 surfactant solutions as a function of surfactant concentration. The figure shows a two times greater foam height when gas injection rate was doubled indicating an initial foam volume equivalent to the dispersed gas and solution volumes lifted by the foam generated. Hence, a stable foam structure was formed with no significant rupture. Figure 3 summarizes the results for all surfactants solution investigated. All can be characterized with good foamability during foam column formation except for Triton X-405 where foam height was increased by only 50% when gas injection rate was doubled.
Figure 4 plots the R5 values as a function of surfactants concentration for all surfactant’s solutions. Except for the Zonyl FSO and Hitenol H-10, all tested surfactants solutions are characterized as poor stability foamers with full foam rupture at very short time frame of less than 5 minutes. Zonyl FSO provided a constant R5 value of 65% at 200 ppm surfactant concentrations and above. Hitenol H-10 surfactant solutions indicate a low R5 values below 50 ppm concentration with a sharp increase in R5 between 50 ppm and 150 ppm and a constant value of 60% above 150 ppm. Accordingly, both surfactants are considered metastable as indicated by R5 values above 50% with transition from low stability to high stability within different ranges of surfactant concentrations. Zonyl FSO required a wider range of concentrations to reach stability compared to Hitenol H-10 and that can be related to the different surface activities of the two surfactants.



The effect of solution salinity on foam stability was tested by comparing the stability of Zonyl FSO and Hitenol H-10 foams at two different salinities of 4% and 10% (Figure 5). The figure shows less foam stability at low concentrations of high salinity Zonyl FSO surfactant solution as indicated by the sharp increase of R5 values within a narrow range of concentrations (about 20 ppm) compared to a much wider range for low salinity solution. No effect was noticed at higher concentrations. On the other hand, matching trend of R5 values was seen at low and medium Hitenol H-10 concentrations with slight decrease of R5 values at higher concentrations, higher salinity solutions.
Figure 6 shows snapshots of the foam columns generated by low and high concentrations of Zonyl FSO and Hitenol H-10 both at 10% salinity. High concentration solutions were characterized by fine and fairly uniform distributed bubble size whereas lower concentrations clearly reveal a different foam texture with larger bubble sizes and broader distribution. These snapshots correlate well with the R5 results presented in Figure 5.
A major concern when applying foam for mobility control is its stability in presence of oil. Foam stability of Zonyl FSO and Hitenol H-10 solutions at 4% salinity were tested in presence of crude oil and results obtained were compared with those obtained previously in absence of oil (Figure 6). Results reveal that Zonyl FSO solutions indicate lower foam height at lower concentrations. No effect was seen at concentrations above 100 ppm. Observation indicate a good foam height but with some gap in the lower portion of the foam column and low oil lift at concentrations on the range 100 to 300 ppm. Above 600 ppm a good foam structure was obtained with good stability and good lifting capacity of oil at the lamella’s skeleton and plateau boarders with no drainage as shown in the snap shoots presented in Figure 7. Hitenol H-10 showed drastic lower foam height indicating very week stability in which foam vanishes as gas injection ceases at concentrations around 100 ppm. As concentration increases within the range of 150 to 300 ppm, good foam height was observed but with week structure in which foam vanishes completely within the first minute. As concentration increases above 1000 ppm, we were able to obtain a good foam height with reasonable stability and oil lifting efficiency. Figure 8 present a snapshot of Hitenol H-10 foam column showing oil phase distribution inside the foam structure accumulating at the plateau borders indicating the strength of Hitenol H-10 foam films. However, lifted oil drained out of the foam structure within a short time frame. In summary, presence of oil, at the volume fraction implemented, affects the stability of the foam columns of both surfactants. The effect depends on the surfactant type and surfactant concentration where stability decreases at low concentrations of Zonyl FSO and at all concentrations range tested of Hitenol H-10.




Dynamic foam flooding in porous medium
Foam efficiency in in porous mediums were performed in absence of oil to investigate the foam behavior of Zonyl FSO and Hitenol H-10 surfactants in porous medium. Surfactants solutions (4% solutions salinity) at concentrations of 600 ppm and 1000 ppm were utilized for Hitenol H-10 and Zonyl FSO respectively. Table 2 summarizes the runs conducted and the main results obtained.

To investigate the effect of surfactant slug size on mobility, two core samples were subjected to base line gas flood and Hitenol H-10 foam flood each. The first core was pre-injected at 0.5 pore volume and the other was pre-flooded with 1.0 pore volume slug size. Pressure drop encountered during gas flood and foam flood were compared (Figure 9). The figure indicates a peak pressure for both gas and foam flood at 1.0 pore volume slug size corresponding to gas breakthrough. Higher pressure drops and delayed gas breakthrough by 0.2 to 0.3 pore volumes of gas injection confirm the foam formation upon contact between CO2 and pre-injected slug of surfactants solutions. Consequently, foam flooding provided higher water recovery factor of 45.17% compared to 18.32% obtained by CO2 gas flooding. Foam flooding conducted with smaller 0.5 pore volume slug was compared to base line gas flooding and results indicates higher pressure drop and delayed gas breakthrough during foam flooding. Gas flooding provided water cumulative recovery factor of 17.80% compared to 39.2% when foam flooding was implemented. Foam efficiency is determined by the mobility reduction factor (MRF) defined as the ratio of pressure drop encountered during foam flood to that recorded during the gas flooding. Figure 10 compares the MRF during foam injection with 0.5 and 1.0 pore volume slug sizes of Hitenol H-10 surfactant. The figure indicates similar breakthrough time but with higher MRF for the larger slug experiment indicating formation of relatively better foam quality explaining the higher water recovery factor for the run conducted with higher surfactant slug size. The difference dissipates as gas breakthrough with curves overlapping each other and providing effective steady state MRF of 1.45 for the two slug sizes. Apparent viscosity generally used to determine the foam strength could be inferred from Darcy law using steady state pressure drop. Foam apparent viscosity of 4.1 cp and 3.98 cp were calculated for the runs conducted with 1.0 and 0.5 pore volume slug sizes respectively, indicating close foam apparent viscosities of Hitenol H-10 at the two slug sizes tested.


As we did with Hitenol H-10, two more runs in fresh cores were conducted with Zonyl FSO surfactant at 0.5 and 1.0 pore volume slug sizes. Figure 11 plot the pressure drops measured during gas and foam flooding at both slug sizes. The figure shows higher pressure drop and substantial delayed breakthrough time during foam injection with 1.0 pore volume slug size as compared to CO2 gas injection. Cumulative water recovery factor obtained during foam flooding was 39.2 compared to 21.69 for gas injection with the increase observed at late stage of injection. Similarly, injection of Zonyl FSO at slug size of 0.5 pore volume was compared to that encountered during gas flooding. Small delay was noticed in break through time with insignificant change in pressure drop at breakthrough and after. These observations indicate the formation of lower quality foam with such small slug size. Water recovery factors during the gas and foam flooding were 19.11 and 35.7 respectively. The two Zonyl FSO foam flooding runs at both slug sizes are compared in Figure 12 and clearly, larger slug size provided relatively more efficient foam at the inlet section as indicated by the higher MRF and delayed breakthrough time. However, steady state MRF was equivalent at a value of 1.1 for both slug sizes. Foam apparent viscosity of 3.17 cp and 2.9 cp were calculated for runs conducted with 1.0 and 0.5 pore volume slug sizes respectively, indicating close foam apparent viscosities.

The performance of the both surfactant solutions were compared in Figure 13 for the runs conducted at 0.5 and 1.0 pore volume slug sizes. Results obtained indicate the positive effect of increasing slug size on MRF for the two surfactants with relatively better performance for Hitenol H-10 surfactant especially at smaller slug size. Dilution of surfactant solutions with resident brine is believed to be the reason for the difference in generated foam performance where larger slug size of surfactant solution will tend to be exposed to less dilution and loose less of its efficiency.

The presented dynamic foam flooding results concentrates on the foam strength and performance in absence of oil. More runs were conducted in presence of oil to study the effectiveness of these foamers to enhance oil recovery. The outcomes of these experiments are going to be presented in a different article.
Conclusion
Foamability and foam stability of several anionic and nonionic surfactants were investigated, and foam characteristics were studied at different solutions salinities in absence and presence of crude oil. The best performed surfactants were further tested in porous medium and the following are the main outcomes of the work:
• All surfactants except Triton X-405 showed good formability but Zonyl FSO and Hitenol H-10 surfactants provided the highest foam longevity in the absence of oil. Foamability and foam stability enhances as surfactants concentrations increases.
• High concentration solutions were characterized by fine and fairly uniform distributed bubble size whereas lower concentrations clearly reveal different foam texture with larger bubble sizes and broad distribution.
• Higher salinity reduces the foam stability at low concentrations of Zonyl FSO surfactant. No effect was seen at higher concentrations. On the other hand, no effect was observed on foam stability of Hitenol H-10 solutions at different concentrations except for small drop on foam longevity at higher concentrations.
• In presence of oil, lower concentration of Zonyl FSO solutions provided lower foam stability; however, foam stability was not affected at concentrations above 100 ppm. On the other hand, lower stability was noticed at all concentration range tested of Hitenol H-10.
• Hitenol H-10 surfactant was able to lift good portion of the oil upward but that oil drained out of the foam structure. To the contrary, Oil phase was distributed inside the foam structure of Zonyl FSO accumulating at the plateau borders indicating the strength of foam films.
• The performance of the two surfactants in porous medium were compared and results indicate positive effect of increasing slug size on MRF for both surfactants with relatively better performance for Hitenol H-10 surfactant.
Acknowledgement
This project was funded by the National Plan for science, Technology and Innovation (MAARIFAH), King Abdulaziz City for Science and Technology, the Kingdom of Saudi Arabia (08-ENE402- 2).
Conflict of Interest
None.
References
- Bond DC, Holbrook OC (1958) U.S. Patent No. 2,866,507, 30th December 1958.
- Fried AN (1961) The foam drive processes for increasing the recovery of oil, Report of Investigations 5866, USBM.
- Schramm LL, Wassmuth F (1994) Foams: Basic Principles, In: Schramm LL (ed), Foams: Fundamentals & Applications in the Petroleum Industry, American Chemical Society, Washington, DC, pp. 1-43.
- Malysa K, Lunkenheimer K (2008) Foams under dynamic conditions, Current Research Article in Colloid & Interface Science 13: 150-162.
- Israelachvili JN (1991) Intermolecular & Surface Forces, 2ed, Academic Press, San Diego.
- Chiang JC, Sanyal SK, Castanier LM, Brigham WE, Sufi A (1980) Foam as a Mobility Control Agent in steam Injection Processes, paper SPE 8912 presented at the 50th Annual California Regional Meeting, Los Angeles, pp. 9-11.
- Marsden SS, Khan SA (1996) The Flow of Foam through Porous Media and Apparent Viscosity measurements. Soc Pet Eng J 6: 17-25.
- Aveyard R, Binks BP, Fletcher PDI, Peck TG, Garrett PR (1993) Entry and spreading of alkane drops at the air-surfactant solution interface in relation to foam and soap film stability, J Chem Soc Faraday Trans 89: 4313-4321.
- Denkov ND (2004) Mechanisms of foam destruction by oil-based antifoams, Langmuir 20(22): 9463-9505.
- Bergeron V, Fagan ME, Radke CJ (1993) Generalized entering coefficients - A criterion for foam stability against oil in porous media, Langmuir 9(7): 1704-1713.
- Garrett PR (1993) The mode of action of antifoams, In Defoaming: Theory and industrial applications, Edited by Garrett PR and Marcel Dekker, New York.
- Maire J, Coyer A, Fatin-Rouge N (2015) Surfactant foam technology for in situ removal of heavy chlorinated compounds-DNAPLS, Journal of Hazard Mater 299: 630-638.
- Osei-Bonsu K, Shokri N, Garassia P (2015) Foam Stability in the presence and absence of hydrocarbons: From bubble to bulk Scale, Colloids Surf A 481: 514-526.
- Jones SA, Van der Bent V, Farajzadeh R, Rossen WR, Vincent-Bonnieu (2016) Surfactant screening for foam EOR: correlation between bulk and coreflood experiments, Colloids Surf A 500: 166-176.
- Nikolov AD, Wasan DT, Huang DW, Edwards DA (1986) The effect of oil on foam stability: mechanisms and implications for oil displacement by foam in porous media. Paper SPE15443-MS presented at The Annual Technical Conference and Exhibition, 5-8 October 1986, New Orleans, Louisiana.
- Lau HC, O Brien SM “Effects of spreading and non-spreading oils on foam propagation through porus media”. SPE Res Eng 3(3): 893-896.
- Simjoo M, Rezaei T, Andrianov A, Zitha PLJ (2013) Foam stability in the presence of oil: effect of surfactant concentration and oil type, Colloids and Surfaces A 438: 148-158.
- Vikingstad Anne Kari, Skauge A, Høiland H, Aarra M (2005) Foam-oil interactions analyzed by static foam tests, Colloids and Surfaces A 260: 189-198.
- Aronson AS, Bergeron V, Fagan ME, Radke CJ (1994) The influence of disjoining pressure on foam stability and flow in porous media, Colloids and Surfaces A 83: 109-120.
- Dalland M, Hanssen JE, Kristiansen TS (1994) Oil interaction with foams under static and flowing conditions in porous media, Colloids Surf A 82(2): 129-140.
- Lunkenheimera K, Malysa K (2003) Simple and generally applicable method of determination and evaluation of foam properties, Journal of Surfactants and Detergents 6(1): 69-74.
-
El Mahdy Osama, Al Quraishi Abdulrahman A, Belhaij Azmi M. Efficiency of Different Foaming Agents for Enhanced Oil Recovery: Bulk and Dynamic Flooding Screening. Glob J Eng Sci. 4(2): 2019. GJES.MS.ID.000583.
-
Electric, Prabe, Peanuts
-

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.






