 Research article
Research article
Reduction of Obesity can be Predicted by Early Weight Loss under Testosterone Therapy (TTh)
Raidh Talib Alzubaidi1,2, Aksam Yassin1,2,3*, Anas Albudairat1, Mohamed Hatem1, Hasan Abdallah1,Abdelaziz Saleh1, Hatem Kamkoum1, Abdulla Al-Ansari1,2
1Hamad Medical Corporation, Aisha Al Attiyya Hospital, Andrology & Men’s Health Unit, Qatar
2Weill Cornell Medical School, NY, USA
3Dresden International University, Preventive Medicine Program, Dresden Germany
Aksam Yassin, MD, PhD, EdD, Head of Men’s Health Unit, Hamad Medical Corporation, Doha, Qatar
Received Date: June 16, 2025; Published Date:June 24, 2025
Abstract
We and others have previously shown that testosterone replacement therapy (TRT) results in sustained weight loss in the majority of middleaged hypogonadal men. Previously, however, a small proportion failed to lose at least 5% of their baseline weight. The reason for this is not yet understood. In the present study, we sought to identify early indicators that may predict successful long-term weight loss, defined as a reduction of at least 5% of total body weight relative to baseline weight (T0), in men with hypogonadism undergoing TRT. Eight parameters measured were assessed as potential predictors of sustained weight loss: loss of 3% or more of baseline weight after 1 year of TU treatment, severe hypogonadism, BMI, waist circumference, International Prostate Symptom Score (IPSS), glycated hemoglobin (HbA1C), age and use of vardenafil. Among the eight measured parameters, three factors were significantly associated with sustained weight loss over the entire period of TU treatment: (1) a loss of 3% of the baseline body weight after 1 year of TRT; (2) baseline BMI over 30; and (3) a waist circumference >102 cm. Age was not a predictor of weight loss.
Keywords: Testosterone; obesity; hypogonadism; BMI; waist circumference; predictors; weight loss
Introduction
Male hypogonadism is defined as a clinical syndrome resulting from the failure of the testes to produce physiological levels of testosterone. This leads to a number of symptoms including erectile dysfunction, loss of libido, impaired cognitive function, depression, fatigue, and lack of physical strength [1-4]. Recent guidelines by the International Society for Andrology (ISA) and International Society for the Study of the Aging Male (ISSAM) [5] state that testosterone therapy is required when total testosterone concentrations fall below 12 nmol/L. It is thought that up to 40% of men over the age of 45 may be hypogonadal to some degree [6]. It is well established that low testosterone levels are strongly associated with increases in abdominal obesity, body mass index (BMI; kg/m2) and waist circumference, and high circulating low-density lipoprotein (LDL) and triglyceride concentrations [7-10]. The hypogonadal-obesity cycle suggests that in the hypogonadal state, there is an increase in abdominal adipose tissue which further reduces testosterone, resulting in progressively more severe hypogonadism [11,12].
Weight loss in obese men has been shown to produce a significant rise in both total and free testosterone concentrations in proportion to the degree of weight loss [13]. Moreover, restoring testosterone levels in obese hypogonadal men produces a significant and sustained weight loss, accompanied by reductions in BMI, waist circumference and abdominal obesity, improvements in psychological and physical health-quality indicators, and cardiovascular risk factors [14-22]. Patients undergoing testosterone undecanoate (TU) treatment also show an improvement in body composition which comprises an increase in lean muscle mass and a decrease in fat tissue mass [23]. Such improvements are seen to deteriorate when TU is withdrawn for 1–2 years, suggesting that these men may in fact require lifelong TU treatment [24,25]. Conversely, prostate cancer patients receiving gonadotropin-releasing hormone agonist (GnRH) therapy, which induces a state of hypogonadism, display an increase in fat tissue mass and a reduction in lean muscle mass [26].
Abdominal obesity and low testosterone concentrations are also associated with increased risk of metabolic syndrome, type 2 diabetes (T2D), and cardiovascular disease (CVD), and the weight loss observed with testosterone replacement therapy (TRT) reduces the risk of these diseases [15,16]. We and others have previously reported that TRT results in sustained weight loss in the majority of middle-aged hypogonadal men [27]. However, in our cohort of 261 men with hypogonadism, a small proportion failed to lose at least 5% of their baseline weight [27]. The reason for this is not understood. One previous study however has observed that certain characteristics in men, such as extreme obesity and increased waist circumference, may predict the success of testosterone therapy to reduce body weight [28]. In the present study, we sought to identify further early indicators that may predict successful longterm weight loss, defined as a reduction of at least 5% of total body weight relative to baseline weight (T0), in men with hypogonadism undergoing TRT.
Methods
Patients and Study Design
From November 2004, 261 patients (mean age 58 years) diagnosed with hypogonadism were treated with long-acting Testosterone Undecanoate (TU; Nebido®, Bayer Pharma, Berlin, Germany) in a prospective observational and longitudinal registry study. All individuals included in the study gave informed written consent. The study was conducted according to the ethical guidelines formulated for observational studies by the German Ärztekammer (German Medical Association) and followed the principles outlined in the Helsinki Declaration of 1975, as revised in 1983. Men with a total serum testosterone concentration of ≤3.5 ng/mL (12 nmol/L) and documented symptoms of erectile dysfunction (ED) (Sexual Health Inventory for Men (SHIM) score ≤21) met the inclusion criteria. Men received intramuscular injections of 1000 mg TU at day 1 approximately 3 weeks after diagnosis, again at week 6, and then once every 3 months thereafter. Patients were entered into a cumulative registry database once they had received treatment for at least 1 year and followed for a maximum of 5 years.
Assessment of Outcome
Eight parameters measured in men with hypogonadism were assessed as potential predictors of sustained weight loss: loss of 3% or more of the baseline weight after 1 year of TU treatment; severe hypogonadism at T0 (TT < 7.8 nmol/L; 225 ng/dL); BMI at T0 > 30 kg/m2; waist circumference at T0 > 102 cm; IPSS at T0 < 1; glycated hemoglobin (HbA1C; 4.8–5% or 22.4–39.9 mmol) at T0 > 7%; age at T0 > 70 years, ≤1 use of the phosphodiesterase 5 inhibitor vardenafil (Levitra®, Bayer). These independent variables were coded in binary fashion, where the presence of the variable in a patient = 1 and its absence = 0. Binary logistic regression was performed using the eight binary-coded independent variables, with weight loss of 5% or more as the dependent variable (5% or more weight loss over the course of TU treatment = 1; <5% weight loss = 0; median period over which body weight was measured was 4.25 years). Statistical analysis was performed using SPSS Statistics software (IBM, Armonk, NY). Parameters whose regression coefficient (B) were significantly different from zero (p < 0.05; Wald test) were significantly associated with 5% weight loss over the course of TU treatment.
Results
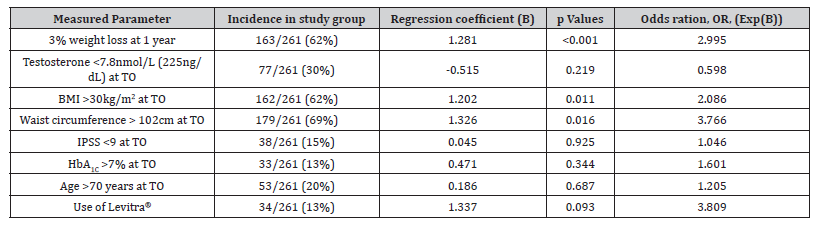
Amongst the eight parameters measured in men with hypogonadism, either at the onset of TU treatment or at the end of the first year of treatment, three of these factors were found to be significantly associated with sustained weight loss over the entire period of TU treatment (Table 1). The first significant association was a loss of 3% of the baseline body weight after 1 year of TRT (p < 0.001, Wald test; odds ratio, OR = 2.995; 95% confidence interval). The second was a BMI over 30 (kg/m2) at the onset of treatment (p = 0.011; OR = 2.086), and the third was a waist circumference >102 cm at the onset of treatment (p = 0.016; OR = 3.766). Sustained long-term weight loss was not significantly associated with severe hypogonadism at T0 (p = 0.219), IPSS at T0 < 1 (p = 0.925), glycated hemoglobin at T0 > 7% (p = 0.344), age over 70 years at T0 (p = 0.687), or use of vardenafil (p = 0.093).
Discussion
The results from this present study demonstrate that of the parameters measured, sustained weight loss in men with hypogonadism undergoing TRT may be more likely to be achieved by men who lose 3% of baseline body weight within the first year, men with a BMI over 30 (kg/m2), or men with a waist circumference measurement greater than 102 cm at the onset of TRT treatment (T0). Obese patients (elevated BMI and waist circumference) tend to sustain a higher weight loss. TRT was the only intervention that the current cohort was exposed to. Previously, our group observed a significant reduction between baseline and the respective last visit of every patient in the following parameters: mean waist circumference, mean weight, mean percentage weight change (loss), and mean BMI. Our 261 patients lost a total of 2621 kg over the period of the study. However, in this cohort of 261 men with hypogonadism, around 15% failed to lose 5% of their pretreatment weight and the underlying cause of this is not understood.
Table 1:Binary logistic regression: predictors of sustained weight loss.
In a previous study by our group, whereby weight loss was measured in a mixed group of obese and nonobese men, the proportion of men who failed to lose 5% of their baseline body weight was 20% [22], much higher due to the fact a high baseline BMI is a predictor of weight loss, implying that nonobese men are less likely to lose a significant proportion of their body weight. Conversely, the proportion of men failing to lose 5% of their body weight was considerably less in a study analyzing only obese men [20]. Indeed, weight loss studies primarily recruit obese subjects whereby proportional reduction in mass is often increased. Our registry study has included obese, overweight, and normal weight men and demonstrates that while weight loss is greatest in the obese, an overall weight reduction was apparent. Weight loss in overweight and normal weight men may also be desirable dependent upon the body mass depot that is reduced. For example, testosterone is known to decrease fat mass while increasing lean mass to improve body composition in men [29].
We have also previously reported that waist circumference as an indicator of central fat mass is the most successful predictor of sexual health status and health-related quality of life in hypogonadal men [30]. This study aimed to identify easily assessed factors that predict the likelihood of successful long-term weight loss due to TRT alone, and therefore the same parameters may also be used to predict failure. This is crucial because men suffering from hypogonadism who do not present with the parameters predicting success, TRT alone may not be sufficient to reduce obesity. Weight loss induced by TRT leads to improvement in quality of life and benefits the patient not just regarding their weight per se but also reducing their risk of CVD, T2D, and metabolic syndrome. If such patients can be identified at the start of TRT or in the early stages of treatment, weight loss may be achieved through other means, either additional medical interventions or simply exercise and other lifestyle changes. Our findings that sustained weight loss in men undergoing TRT may be predicted by losing 3% of baseline body weight in the first year, and a baseline BMI over 30 kg/m2 and waist measurement greater than 102 cm are consistent with that of a previous study in which 110 hypogonadal men with a mean age of 59.6 received TU [28].
Saad et al. reported that higher obesity and waist circumference predicted the greatest reduction in weight, waist circumference, and BMI over the 2-year period. Contrary to our findings, Saad et al. reported that age predicted a beneficial effect of TRT on waist circumference, glucose and all lipids with age, BMI, and C-reactive protein considered independent predictors of the strongest benefit of TRT on the metabolic syndrome [28]. A meta-regression analysis by Corona et al. illustrated that following TU treatment, the changes observed in BMI were significantly lower in older patients and were increased as a function of trial duration and/or baseline BMI, i.e. maximum weight loss is obtained in younger, more obese men treated for a longer time [31]. In addition to the parameters that may predict successful weight loss in men with hypogonadism, this study importantly highlights parameters that do not significantly affect the outcome of TRT in terms of a reduction in obesity. A popular myth persists that older people with hypogonadism would not benefit from TRT, however being over 70 years of age was not significantly associated with reduced odds of successful TRT in terms of long-term weight loss and therefore testosterone administration should not be withheld from this older cohort of men.
These data are confirmed in a previous study by Saad et al. whereby men over the age of 65 with hypogonadism were found to benefit as much from TRT as younger men demonstrating age is not a contraindication to TRT in hypogonadal men [32]. Similarly, the use of the PDE5 inhibitor vardenafil did not affect the outcome of TRT, suggesting that TU and vardenafil may be prescribed together without affecting the likelihood of success of TRT. However, the actual frequency of vardenafil use throughout this trial could not be accurately assessed and may limit the interpretation of this result. Total testosterone levels, HbA1C, and IPSS score were also not found to be associated with differences in TRT-induced weight loss. Several studies have indicated that early weight loss in the first year predicts the success of further and long-term weight loss [33,34], which prognostically and clinically means that continuation of TRT will be beneficial. Furthermore, research into the ability to predict the presence and/or improvement of comorbidities in hypogonadal men undergoing TRT has identified erectile dysfunction as a strong predictor of obesity, abnormal lipid pattern, and a history of type 1 and 2 diabetes [35]. We can also recommend more readings [36-42] to enhance the knowledge on innovations and paradigm shifts in testosterone treatment.
A limitation of the present study is the nature of the registry design. This single-center, open-label study was not a randomized controlled trial (RCT) and therefore may limit the scope of interpretation of the presented findings. However, many of the benefits (and risks) of medicines fall outside the bounds of the classical RCT and as such including data in such studies, routinely collected in the usual practice of medicine, are more reflective of the heterogeneous patients seen in real-world practice settings.
Conclusion
This prospective, observational, and longitudinal analysis demonstrates that sustained weight loss in men with hypogonadism undergoing TRT may be more likely achieved in men losing 3% of baseline body weight in the first year of the trial, men with a BMI over 30, and men with a waist measurement greater than 102 cm at the onset of TRT.
Disclosure
Authorship has no conflict of interest to report
References
- Bhasin S, Cunningham GR, Hayes FJ, Matsumoto AM, Snyder PJ, et al. (2010) Testosterone therapy in men with androgen deficiency syndromes: an endocrine society clinical practice guideline. J Clin Endocrinol Metab 95(6): 2536-2559.
- Wald M, Meacham RB, Ross LS, Niederberger CS (2006) Testosterone replacement therapy for older men. J Androl 27(2): 126-132.
- Harman SM, Metter EJ, Tobin JD, Pearson J, Blackman MR (2001) Longitudinal effects of aging on serum total and free testosterone levels in healthy men. Baltimore Longitudinal Study of Aging. J Clin Endocrinol Metab 86(2): 724-731.
- Purifoy FE, Koopmans LH, Mayers DM (1981) Age differences in serum androgen levels in normal adult males. Hum Biol 53(4): 499-511.
- Lunenfeld B, Mskhalaya G, Zitzmann M, Arver S, Kalinchenko S, et al. (2015) Recommendations on the diagnosis, treatment and monitoring of hypogonadism in men. Aging Male 18(1): 5-15.
- Mulligan T, Frick MF, Zuraw QC, Stemhagen A, McWhirter C (2006) Prevalence of hypogonadism in males aged at least 45 years: the HIM study. Int J Clin Pract 60(7): 762-769.
- Derby CA, Zilber S, Brambilla D, Morales KH, McKinlay JB (2006) Body mass index, waist circumference and waist to hip ratio and change in sex steroid hormones: the Massachusetts Male Ageing Study. Clin Endocrinol 65(1): 125-31.
- Phillips GB (1993) Relationship between serum sex hormones and the glucose-insulin-lipid defect in men with obesity. Metab Clin Exp 42(1): 116-120.
- Haffner SM, Valdez RA, Stern MP, Katz MS (1993) Obesity, body fat distribution and sex hormones in men. Int J Obes Relat Metab Disord 17: 643-649.
- Seidell JC, Bjorntorp P, Sjostrom L, Kvist H, Sannerstedt R (1990) Visceral fat accumulation in men is positively associated with insulin, glucose, and C-peptide levels, but negatively with testosterone levels. Metab Clin Exp 39(9): 897-901.
- Kapoor D, Malkin CJ, Channer KS, Jones TH (2005) Androgens, insulin resistance and vascular disease in men. Clin Endocrinol 63(3): 239-250.
- Cohen PG (1999) The hypogonadal-obesity cycle: role of aromatase in modulating the testosterone-estradiol shunt – a major factor in the genesis of morbid obesity. Med Hypotheses 52(1): 49-51.
- Camacho EM, Huhtaniemi IT, O’Neill TW, Finn JD, Lee DM, et al. (2013) Age-associated changes in hypothalamic-pituitary-testicular function in middle-aged and older men are modified by weight change and lifestyle factors: longitudinal results from the European Male Aging Study. Eur J Endocrinol 168(3): 445-455.
- Almehmadi Y, Yassin AA, Nettleship JE, Saad F (2016) Testosterone replacement therapy improves the health-related quality of life of men diagnosed with late-onset hypogonadism. Arab J Urol 14(1): 31-36.
- Haider A, Yassin AA, Haider KS, Doros G, Saad F, et al. (2016) Men with testosterone deficiency and a history of cardiovascular diseases benefit from long-term testosterone therapy: observational, real-life data from a registry study. Vasc Health Risk Manage 12: 251-261.
- Yassin AA, Nettleship J, Almehmadi Y, Salman M, Saad F (2016) Effects of continuous long-term testosterone therapy (TTh) on anthropometric, endocrine and metabolic parameters for up to 10 years in 115 hypogonadal elderly men: real-life experience from an observational registry study. Andrologia 48(7): 793-799.
- Haider A, Saad F, Doros G, Gooren L (2014) Hypogonadal obese men with and without diabetes mellitus type 2 lose weight and show improvement in cardiovascular risk factors when treated with testosterone: an observational study. Obes Res Clin Pract 8(4): 339-349.
- Haider A, Yassin A, Doros G, Saad F (2014) Effects of long-term testosterone therapy on patients with “diabesity”: results of observational studies of pooled analyses in obese hypogonadal men with type 2 diabetes. Int J Endocrinol 683515.
- Francomano D, Lenzi A, Aversa A (2014) Effects of five-year treatment with testosterone undecanoate on metabolic and hormonal parameters in ageing men with metabolic syndrome. Int J Endocrinol 527470.
- Saad F, Yassin A, Doros G, Haider AH (2015) Effects of long-term treatment with testosterone on weight and waist size in 411 hypogonadal men with obesity Classes I–III: observational data from two registry studies. Int J Obes 40: 162-170.
- Saad F, Haider A, Doros G, Traish A (2013) Long-term treatment of hypogonadal men with testosterone produces substantial and sustained weight loss. Obesity 21(10): 1975-1981.
- Yassin AA, Doros G (2013) Testosterone therapy in hypogonadal men results in sustained and clinically meaningful weight loss. Clin Obes 3(3-4): 73-83.
- Aversa A, Bruzziches R, Francomano D, Spera G, Lenzi A (2010) Efficacy and safety of two different testosterone undecanoate formulations in hypogonadal men with metabolic syndrome. J Endocrinol Invest 33(11): 776-783.
- Yassin A, Nettleship JE, Talib RA, Almehmadi Y, Doros G (2016) Effects of testosterone replacement therapy withdrawal and retreatment in hypogonadal elderly men upon obesity, voiding function and prostate safety parameters. Aging Male 19(1): 64-69.
- Saad F, Yassin A, Almehmadi Y, Doros G, Gooren L (2015) Effects of long-term testosterone replacement therapy, with a temporary intermission, on glycemic control of nine hypogonadal men with type 1 diabetes mellitus – a series of case reports. Aging Male 18(3): 164-168.
- Hamilton EJ, Gianatti E, Strauss BJ, Wentworth J, Lim-Joon D, et al. (2011) Increase in visceral and subcutaneous abdominal fat in men with prostate cancer treated with androgen deprivation therapy. Clin Endocrinol 74(3): 377-383.
- Yassin DJ, Doros G, Hammerer PG, Yassin AA (2014) Long-term testosterone treatment in elderly men with hypogonadism and erectile dysfunction reduces obesity parameters and improves metabolic syndrome and health-related quality of life. J Sex Med 11(6): 1567-1576.
- Saad F, Haider A, Giltay EJ, Gooren LJG (2011) Age, obesity and inflammation at baseline predict the effects of testosterone administration on the metabolic syndrome. Horm Mol Biol Clin Investig 6(1): 193-199.
- Kelly DM, Jones TH (2015) Testosterone and obesity. Obes Rev 16(7): 581-606.
- Yassin AA, Nettleship JE, Salman M, Almehmadi Y (2016) Waist circumference is superior to weight and BMI in predicting sexual symptoms, voiding symptoms and psychosomatic symptoms in men with hypogonadism and erectile dysfunction. Andrologia 49(4).
- Corona G, Maseroli E, Maggi M (2014) Injectable testosterone undecanoate for the treatment of hypogonadism. Expert Opin Pharmacother 15(13): 1903-1926.
- Saad F, Yassin A, Haider A, Doros G, Gooren L (2015) Elderly men over 65 years of age with late-onset hypogonadism benefit as much from testosterone treatment as do younger men. Korean J Urol 56(4): 310-317.
- Smith SR, O’Neil PM, Astrup A, Finer N, Sanchez-Kam M, et al. (2014) Early weight loss while on lorcaserin, diet and exercise as a predictor of week 52 weight-loss outcomes. Obesity 22(10): 2137-2146.
- Fujioka K, O’Neil PM, Davies M, Greenway F, Lau DCW, et al. (2016) Early weight loss with liraglutide 3.0 mg predicts 1-year weight loss and is associated with improvements in clinical markers. Obesity 24(11): 2278-2288.
- Almehmadi Y, Yassin DJ, Yassin AA (2015) Erectile dysfunction is a prognostic indicator of comorbidities in men with late onset hypogonadism. Aging Male 18(3): 186-194.
- Yassin A, Abdallah H, Kamkoum H, Alzubaidi RT, Albudairat A, et al. (2025) Intramuscular Testosterone and the Gel in the Current Treatment Era. Biomed J Sci & Tech Res 61(4): 53877-53882.
- Yassin A, Kamkoum H, Alzubaidi RT (2025) Is There a Need for Testosterone Therapy in Older Men? J Advances Med Sci 2(1): 1-3.
- Yassin A, Alzubaidi RT, Kamkoum H, Alzubaidi RT, Ramadan A, et al. (2025) Recent Update on Advancements in Testosterone Therapy (TTh). J Urol Ren Dis 10: 1413.
- Yassin A, Alzubaidi RT, Kamkoum H, Akkad MSE, Mahdi, et al. (2025) Effect of Testosterone Therapy (TTh) on Liver Function and Steatosis. Gastroint Hepatol Dig Dis 8(1): 1-8.
- Yassin A, Kamkoum H, Alzubaidi RT, Ansari AA (2025) Testosterone Prevention Role in Men’s Health: Diabetes Mellitus. J Diabetes Treat 10: 10141.
- Yassin A, Albaba B, Kamkoum H, Alzubaidi RT, Al-Qudimat AR, et al. (2025) Testosterone Prevention Role in Men’s Health: Cardiovascular Dieseases. Cardiol Res Cardio vasc Med 10: 280.
- Yassin A, Kamkoum H, Alzubaidi RT, Abdallah H, Assad O, et al. (2025) How Long Should We Treat with Testosterone: Stopping Testosterone Therapy (TTh) Wat is Next? Effects of Withdrawal and resumption of TTh. Ann Rev Resear 12(4): 555843.
-
Raidh Talib Alzubaidi, Aksam Yassin*, Anas Albudairat, Mohamed Hatem, Hasan Abdallah, et.all. Reduction of Obesity can be Predicted by Early Weight Loss under Testosterone Therapy (TTh). Curr Tr Clin & Med Sci. 4(2): 2025. CTCMS.MS.ID.000584.
-
Testosterone; obesity; hypogonadism; BMI; waist circumference; predictors; weight loss; iris publishers; iris publisher’s group
-

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.






