Case Report
Case Report
Spinal Cord Tethering: A Case Report of Anesthetic
Considerations and Management for A Patient with
Costello Syndrome
John Sciarra* and Graham Borysko
Department of Anesthesia, University of California, USA
John Sciarra* and Graham Borysko
Department of Anesthesia, University of California, USA
John Sciarra, Department of Anesthesia, University of California, California, USA.
Received Date: October 08, 2019; Published Date: November 27, 2019
Abstract
Costello syndrome (CS), aka faciocutaneoskeletal or FCS syndrome, is a rare genetic disorder that affects very few individuals worldwide, with reported estimates from as few as one in 300,000 to one in 1.25 million people. As it affects so few, there are not many studies and research available on this condition; it can present with any number of complications that can complicate the safe administration of anesthesia. These complications run the gamut from difficult airways to cardiac, neurological, gastrointestinal (GI), and many other issues as well. In this report, we discuss the administration of general anesthesia to a patient with CS who presented for surgery to correct spinal cord tethering. Hopefully, this case summary will lead to a greater understanding of the genetic mutation and will help anesthesia practitioners identify patients with CS.
Introduction
Costello syndrome (CS), aka faciocutaneoskeletal, commonly called FCS syndrome, is a rare genetic disorder that affects many parts of the body with reported estimates ranging from as few as one in 300,000 to one in 1.25 million people [1]. Because it affects so few, there is a paucity of studies and research available on this condition. CS can present with any number of complications that can complicate the safe administration of anesthesia; many of them are thought to result from cell overgrowth and abnormal cell division [1], and run the gamut from difficult airways to cardiac, neurological, GI, and many others issues as well. In this case report, we discuss the administration of general anesthesia to a patient with CS who presented for a laminectomy to correct spinal cord tethering.
Case Presentation
The patient was a 13-year-old female (25.95 kg), presenting for lumbar laminectomy for untethering of the spinal cord. Past medical history is significant for CS, with presumably associated developmental delay, and cardiomyopathy. The preoperative exam showed a well-nourished female with facial features characteristic for CS. She also demonstrated bilateral ulnar deviation of the wrists and fingers. Past surgical history includes Nissen fundoplication, gastrostomy tube placement and removal, port placement and removal, pelvic sarcoma removal, and bilateral femoral osteotomy.
The patient’s vital signs on presentation in the preop holding area, were heart rate 88 bpm, blood pressure 94/55 mm Hg, peripheral capillary oxygen saturation (SpO2) 100% breathing room air, and temperature of 33.2 degree centigrade. She was noticeably anxious and so her father was to be present for induction. Prior to being taken to the operating room, the patient was pre-medicated with 20mg oral midazolam. Upon arrival in the operating room, she was transferred to the operating table and pre-oxygenated while the American Society of Anesthesiologists (ASA) standard monitors were applied. The induction of anesthesia was initiated by the inhalation of 8% sevoflurane and oxygen set at an FiO2 of 1.0. Subsequent mask ventilation was not difficult, so vascular access was obtained by placing a 22g intravenous catheter in the right hand. Neuromuscular blockade was then obtained by giving 3mg of vecuronium prior to attempting endotracheal intubation. Direct laryngoscopy was performed using a Mac 3 laryngoscope, and a Cormack-Lehane grade I view was obtained. A 5.5 mm endotracheal tube was then gently inserted between the vocal cords, and the cuff was inflated with air until an air seal was achieved.
The tube was then secured with tape at 18cm at the teeth. A second 22g intravenous catheter was then placed in the left hand. Anesthesia was maintained with 0.8% sevoflurane in a mixture of oxygen and nitrous oxide to a fraction of inspired oxygen (FIO2) of 0.4, combined with propofol (25 mcg/kg/min) and dexmedetomidine (bolus of 1 mcg/kg over 10 min, then 1 mcg/ kg/hr). A total of 50 mcg fentanyl was administered intravenously. No problems were encountered during the procedure, which was finished in just under 120 minutes. Prior to extubation, decurarization was achieved with 0.8mg glycopyrrolate and 4mg neostigmine. After reestablishing spontaneous ventilation, the patient was extubated without difficulty. She was then taken to the post-anesthesia care unit (PACU) and subsequently discharged to the floor without any issues.
Discussion
CS, aka faciocutaneoskeletal or FCS syndrome, is a rare genetic disorder that can affect multiple body systems. It is caused by mutations in the HRAS gene (a proto-oncogene), located on chromosome 11 (11p15.5). HRAS makes the H-Ras protein, which is a GTP-ase that is part of a pathway that helps control cell growth and division [1]. The mutations lead to an abnormally turned on (active) protein, and many of the signs and symptoms of CS are thought to result from cell overgrowth and abnormal cell division [1]. However, although it is inherited in an autosomal dominant manner, almost all reported cases have resulted from novel mutations, and have occurred in people with no familial history of the disorder. Signs and symptoms of CS overlap considerably with cardiofaciocutaneous syndrome (CFC) and Noonan syndrome, similar disorders with altered gene products that are part of the same signaling pathway as the H-Ras protein [2].
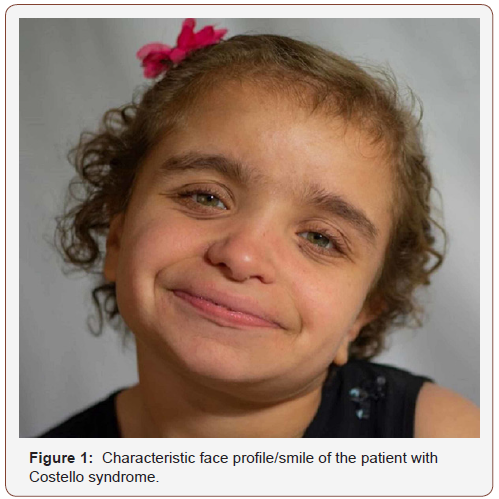
General characteristics of CS include delayed development, intellectual disability, and short stature. Distinctive facial features including a large mouth (Figure 1). Regarding the extremities CS children have tight Achilles tendons, ulnar deviation of wrists/ fingers, loose folds of skin (particularly on the hands and feet), and unusually flexible joints (Figure 2). There is commonly weak muscle tone (hypotonia), and dental and vision problems. As infants, these patients tend to have feeding difficulties. As a result, many CS patients may have a surgical history for correction of pyloric stenosis, or Nissen fundoplication [3]. A thorough review of their surgical history should be obtained from any available records and/or the patient’s guardian if possible. The characteristic smile and hands are considered consistent, and classic traits of the syndrome (Figures 1,2).

There are a number of other concerns across multiple other body systems of which the anesthesiologist should be aware. First, the patients with CS can prove to have a difficult airway due to a constellation of characteristics including macrocephaly, short neck, macroglossia, or the presence of oral or laryngeal papilloma’s. Also, CS patients are prone to sleep apnea due to a high prevalence of obstructive sleep-related respiratory disorders. Cardiac problems are common in patients with CS. Arrhythmias and structural defects are both commonly found in this population. These patients are often followed by a cardiologist from a very early age, and every effort should be made to review any available records prior to any surgical procedure. The most common arrhythmias seen are SVTs and atrial fibrillation, and the most common structural abnormalities encountered include pulmonary valve stenosis, and hypertrophic cardiomyopathy. Management of each of these conditions is per normal standard of care. Individuals with CS may require relatively high doses of medication for sedation/induction. Keep this in mind when choosing which medications to use and, if possible, do a review of available medical records documenting previously given dosages for guidance [4,5].
Patients with CS can have a number of neurological complications, as well. Prominent among them is the presence of a Chiari I malformation, along with any associated abnormalities, including hydrocephalus, syringomyelia, seizures, and a tethered cord. A review of the associated literature demonstrates that general anesthesia may be safely administered in patients with these complications, so long as care is used with the method of intubation and positioning to account for possible associated cervical instability or Craniocervical abnormalities [6]. Also, spinal anesthesia may best be avoided in the presence of co-existing Chiari malformations, as there are case reports which described the onset of signs and symptoms up to two weeks after Dural puncture. As with the previously mentioned cardiac issues, these neurological complications are known in this population. As such, they have likely been followed by a neurologist/neurosurgeon from an early age [7-9].
Conclusion
Patients with CS may present with a wide range of potential complications that an anesthesia provider should be aware of prior to the start of any surgical procedure. Most CS families, by the time the diagnosis is made, have seen multiple specialists, and it is important to leverage this information, and encourage the family to bring in all consults and past admission records. This patient had already had a G-tube placement, Nissen procedure, and abdominal surgery for sarcoma, followed by chemotherapy. They family had much information, and the printed documents, such as a recent cardiac echo, was essential to evaluate for cardiac arrhythmias, and structural abnormalities (i.e. Pulmonic stenosis, hypertrophic cardiomyopathy). With regards to a difficult airway, having a prior anesthesia record (as we did) was invaluable to ensure there are no surprises waiting after induction. Due to the level of hypotonia, direct laryngoscopy should not be difficult. Except for the dental issues, we did not find any literature to suggest difficult mask ventilations or tracheal intubations in CS patients. The tongue is sometimes larger than normal, so this may affect the intubation method as seen fit by the anesthesia provider.
In the operative phase, CS children are often hypermetabolic, and can metabolize drugs faster than normal. Post-operatively, due to the developmental delay, these children may be hard to console, and we found it helpful to involve the family as soon as possible in the recovery room. This is a rare syndrome, and an anesthesia provider may only see one in his or her career. If the diagnosis has already been made, as later in life for these children, then the task is much easier. If the diagnosis has not been made, as is common in infancy, then recognizing physical signs as described above, my clue in the provider, to suspect the syndrome. As is often the case, the well-read and experienced peri-operative physician makes the diagnosis first.
Acknowledgement
None.
Conflict of Interest
No conflict of interest.
References
- Aoki Y, Niihori T, Kawame H, Kurosawa K, Ohashi H, et al. (2005) Germline mutations in HRAS proto-oncogene cause Costello syndrome. Nat Genet 37: 1038-1040.
- Della Marca G, Vasta I, Scarano E, Rigante M, De Feo E, et al. (2006) Obstructive sleep apnea in Costello syndrome. Am J Med Genet A 140: 257-262.
- Gripp KW, Lin AE (2019) Costello Syndrome. In: Adam MP, Ardinger HH, Pagon RA (Eds.), University of Washington, USA.
- Hullander RM, Bogard TD, Leivers D, Moran D, Dewan DM (1992) Chiari I malformation presenting as recurrent spinal headache. Anesth and Analg 75: 1025-1026.
- Katcher K, Bothwell M, Tobias JD (2003) Anesthetic implications of Costello syndrome. Pediatr Anesth 13: 257-262.
- Kim TY, Lee C, Kim JN (2012) Anesthetic management of a patient with Arnold-Chiari malformation type I with associated syringomyelia: A case report. Anesth Pain Med 7: 1-166.
- White SM, Graham JM, Kerr B, Gripp K, Weksberg R, et al. (2005) The adult phenotype in Costello syndrome. Am J Med Genet A 136: 128-135.
- (2019) Costellokids, USA.
- (2019) Costello syndrome, USA.
-
John Sciarra, Graham Borysko. Spinal Cord Tethering: A Case Report of Anesthetic Considerations and Management for A Patient with Costello Syndrome. Anaest & Sur Open Access J. 1(3): 2019. ASOAJ.MS.ID.000512.
-
Costello syndrome, FCS syndrome, Genetic disorder, Spinal cord, Cardiomyopathy, Laryngoscopy, Pyloric stenosis, Macroglossia, Spinal anesthesia, Hypotonia.
-

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.