Research Article
Research Article
Tezepelumab in Mexican Patients with Severe Asthma TH2 predominant: A series of cases
Herrera García JC*
Puebla Clinical Research Unit and Pulmonary Function Unit, Hospital Ángeles Puebla. México.
Puebla Clinical Research Unit and Pulmonary Function Unit, Hospital Ángeles Puebla. México.
Received Date: September 17, 2025; Published Date:October 17, 2025
Abstract
Background: Tezepelumab, a monoclonal antibody targeting thymic stromal lymphopoietin (TSLP) has demonstrated efficacy for severe
asthma patients in clinical trials and real-world evidence in limited data. We describe the characteristics of five patients in a pulmonary clinic in
Puebla, Mexico initiating Tezepelumab in a real-world setting.
Methods: We presented 5 cases from a series of cases in a center of pulmonary unit in Puebla City, Mexico to evaluate the real-world effectiveness
of Tezepelumab in patients with severe asthma. Eligible patients were adults with a confirmed diagnosis of severe asthma treated with Tezepelumab.
Data with symptoms, pulmonary function and corticosteroid use were collected and analyzed at baseline, six months and 1 year follow up.
Results: A total of 5 patients were included. The average age was 52 years. 100% of the patients had more than 2 exacerbations previous
year. 40% had prior biologic therapy and 60% had eosinophil count <300 cells/μL. At follow up, 100% reduction in annual exacerbations. There
were significant improvements in forced expiratory volume in one second (FEV1) and Fraction Exhaled of nitric oxide (FENO). All the patients
discontinued oral corticosteroids and 80% continued with triple therapy.
Conclusion: A series of cases presented with Tezepelumab from 6 months to 1 year follow up were improved asthma control, reductions
in exacerbations, discontinued corticosteroid use and very low symptoms. Tezepelumab demonstrated that is a safety biologic treatment and
improvement symptoms.
Keywords:Severe asthma; Biologic therapy; Tezepelumab; Cases
Introduction
Tezepelumab is a human monoclonal antibody that targets and blocks thymic stromal lymphopoietin (TSLP) — an epithelial cytokine that plays a key role in initiating and maintaining airway inflammation in asthma. Now is a novel biologic therapy (IgG2λ monoclonal antibody) with target in TSLP receptor (not downstream cytokines like IL-4, IL-5, or IL-13), the form of application is subcutaneous injection (administered once every 4 weeks) and was FDA-approved in December 2021 for severe asthma in adults and adolescents (≥12 years). TSLP is released by airway epithelial cells in response to triggers like allergens, viruses, and pollutants it means entire pulmonary epithelium by the route and activates multiple downstream inflammatory pathways involved in asthma — both Type 2 (T2) and non-T2, provocated this actions: reducing eosinophilic inflammation, diminishing airway hyperresponsiveness, decreasing mucus production and suppressing both innate and adaptive immune responses. This effective mechanism gives tezepelumab a wider range of action compared to biologics that only target IL-5, IL-4/13, or Ige and can improve all phenotypes. Tezepelumab nowadays is a novel treatment that have clinical benefits based in Trials and real world use by beginning trial NAVIGATOR in which demonstrated decrease exacerbations more than 56% reduction overall (70% T2 high), improve lung function and asthma control and quality of life, decrease biomarkers like eosinophils, FENO and IgE.
Methods
We presented a series of cases from a pulmonary clinic in Puebla City, Mexico. Eligible patients to the study were: adults >18 years, diagnosed with severe asthma according to European Respiratory Society/American Thoracic Society/Latinoamerican thoracic Society (ERS/ATS/ALAT) definition, that are receiving Tezepelumab per routine clinical practice 210 mg every 4 weeks for severe asthma. All the patients were treated with triple therapy (Dual bronchodilators and steroid)
Patients data and data analysis
Patients data were collected from electronic files (medicalfile. io), during routine clinic visits and follow up between May 2024 to May 2025. We focused on 6 months and 1 year follow up, using the most adjacent follow up data for each patient. Patients with at least 6 months follow up were included in our analysis. Data were collected into a standard medical file in our center, baseline data refers to data prior to treatment initiation and included patient characteristics, exacerbation history and maintenance treatment, blood test and pulmonary function tests. Exacerbation and maintenance oral corticosteroids were averaged from a similar time duration. In all patients use triple therapy for control and completed definition of severe asthma. Asthma control test (ACT) results were used from patients and collected in files notes before treatment and after treatment with a scale of symptoms.
The cohort included 5 patients from pulmonology unit in Puebla city, Mexico, in private clinic where patients went to continue follow for severer asthma treatment and follow up. Two patients of the cohort received a prior biologic during pre-treatment period; the rest were included with naïve biologic treatment. 3 patients with peripheral eosinophils <150 and 2 patients with >300 cells. Pulmonary functions included forced expiratory volume in 1 second (FEV1) and fraction exhaled of nitric oxide (FENO) as part of the routine follow up in the center with the patients with biologics, spirometry is being performed every visit, Asthma exacerbations were defined from increase of symptoms with triple therapy, oral corticosteroids use, emergency department visit of hospitalization and rescue medication fail. The study outcomes include symptoms scale (CAT, mMRC, BORG) before and after treatment. We included patients diagnosed with severe asthma having completed at least 6 months of tezepelumab treatment. Patients treated with treated with Tezepelumab for less than 6 months or having other severe respiratory illness and not accepting participation in the study were excluded from the work.
Analysis
We analyzed clinical characteristics, drug tolerance, and effectiveness. A descriptive analysis variable was performed. Absolute frequencies and percentage values were used for qualitative variables, and mean, standard deviation, median, interquartile range and maximum and minimum were used for quantitative variables and the student T Test for paired samples were used to compare baseline data and data at 6 and 12 months and statistical significant with p<0.05. All patients signed informed consent to report series of cases and was approved by the local ethics committee of our private hospital.
Discussion
Results
We presented a series of five patients diagnosed with severe asthma and treated with tezepelumab. All the patients finished the study, but 50% of them had light adverse effects like: numb hands and hip pain. The baseline patient characteristics are shown in table 1. The mean age was 52 years. 60% were women and 40% men. 4 patients were allergic (80%), 2 patients with < 150 blood eosinophils/μL, 4 patients with FENO > 25 ppb and 1 patient had T2 low biomarker (eosinophils < 150 blood eosinophils/ μL and FENO < 25 ppb) 2 patients were treated previously with other biologic drug (omalizumab/Benralizumab). Table 2 shows the results with tezepelumab treatment. All of the patients had at least 2 exacerbations previous year. Three patients took oral corticosteroids daily (60%), 100 % of the patients took and continue with triple therapy treatment. 100% of the patients suspended oral corticosteroids therapy at the end of the study (Table 1 &2).
Table 1: Demographics of patients hospitalised for first episode of wheeze pre-COVID and during COVID
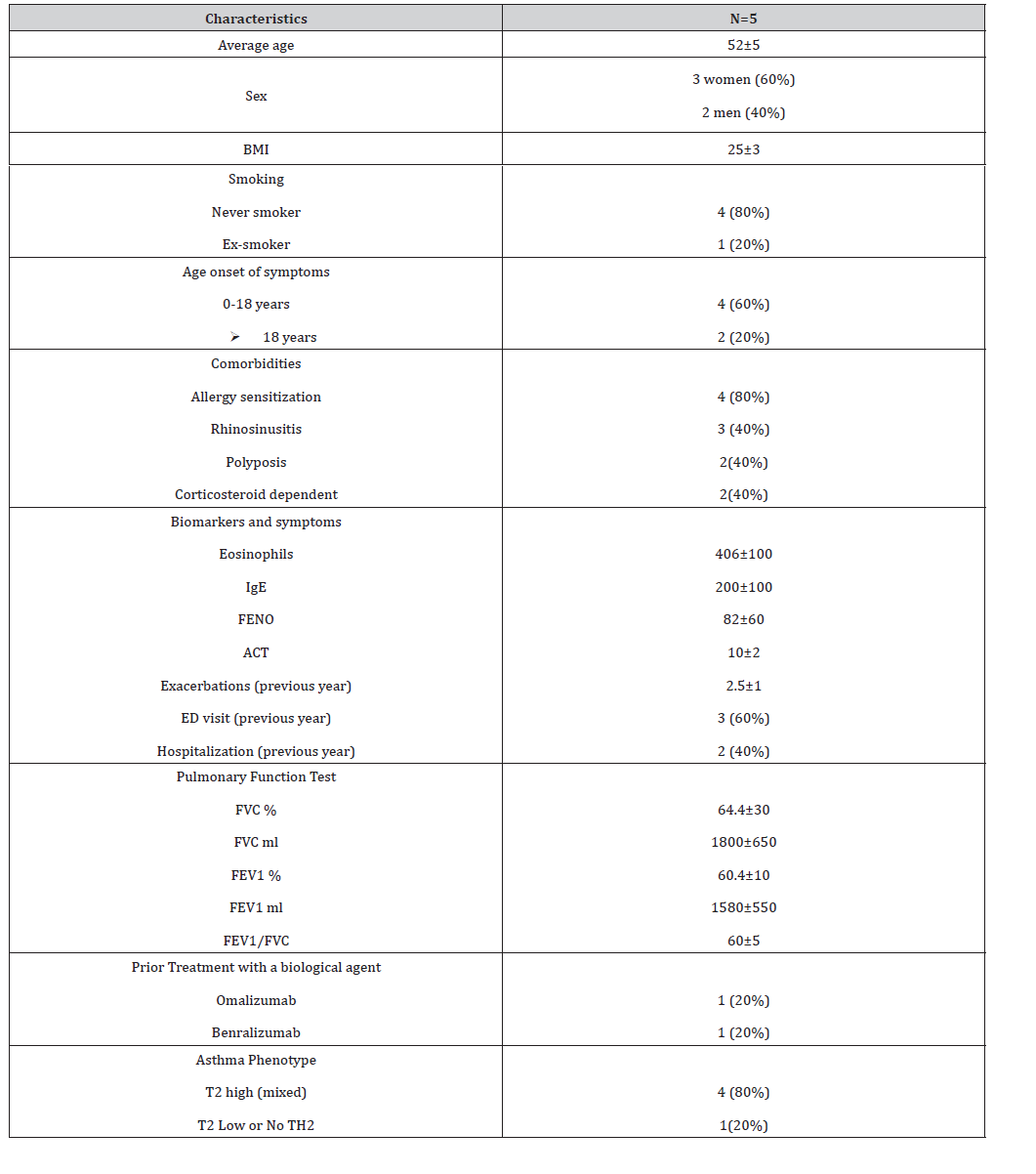
IgE: Immunoglobulin E
FVC: Forced vital capacity
FEV1: Forced expiratory volume in 1 second
ACT: Asthma control test
FENO: Fraction exhaled nitric oxide
BMI: Body mass index
Table 2: Demographics of patients hospitalised for first episode of wheeze pre-COVID and during COVID
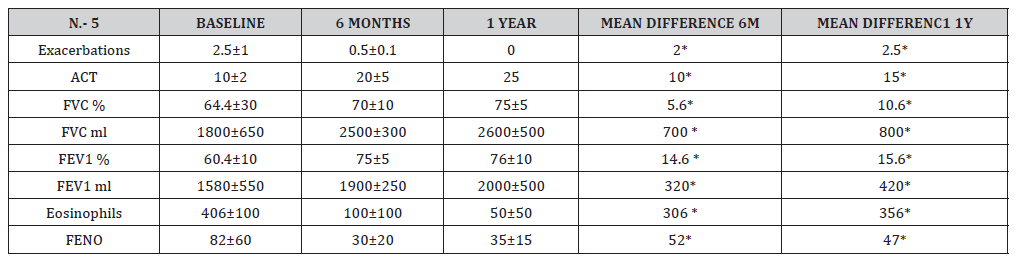
* p<0.05
Discussion
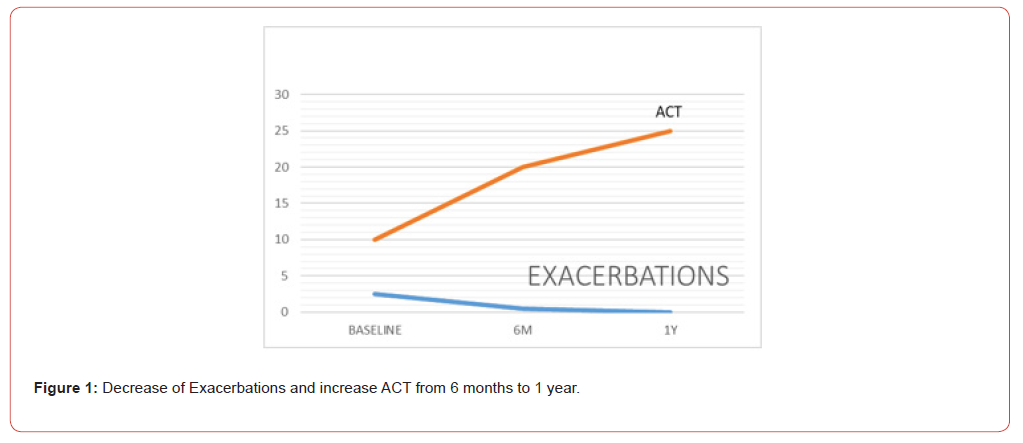
All the patients in the study improvement symptoms, exacerbations decreased from baseline to zero (from 2.5 to 0.5 in 6 months and 0 at 1 year) this kind of improvement is incredible, they have not gone to emergency visits and they continued with better quality of life. ACT improvement and decreased majority of their symptoms. 80% of the patients were never smokers, 60% began their disease before 18 years, 80% were allergic, 2 patients were with polyposis and had 2 polypectomies before biologic treatment. Eighty percent of the patients had mixed phenotypes, and one had T2 low. In 6 months were descriptive than patient’s improvement symptoms, decreased eosinophils and increase ACT, but we don’t have decrease significantly in FENO, but all the patient’s improvement in pulmonary function tests, I think is one of the best news to use this drugs in this biologic era, 1 patient were in Omalizumab for 5 years and increase this symptoms and exacerbations and changed to Tezepelumab and improvement her quality of life. The other patient was in benralizumab for 2 years and increase exacerbations and changed to Tezepelumab and improvement symptoms too. Now we know that there are a lot of different patients in real world, different ages, comorbidities, and responses to this disease. This real-world study shows that all the patients with severe asthma are different and need to prove and all of them respond differently (Figure 1-3).
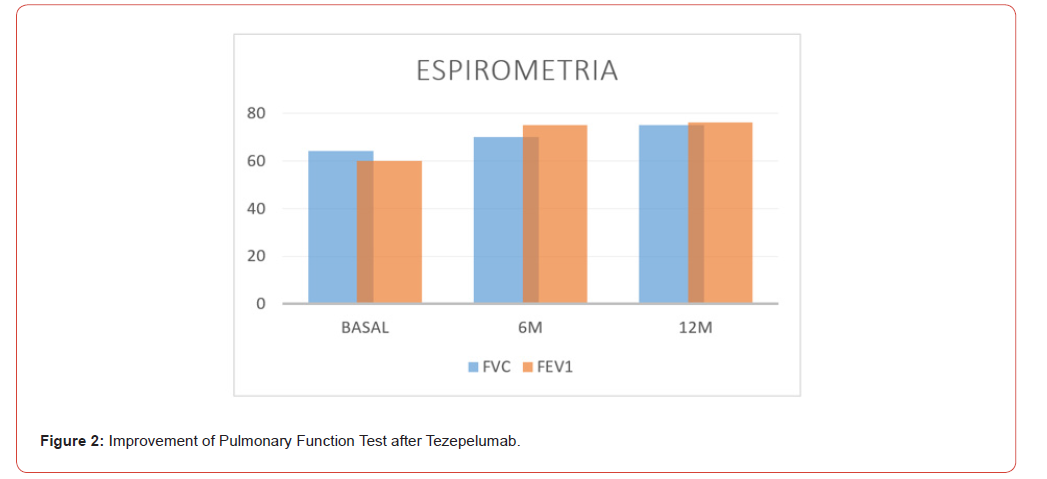
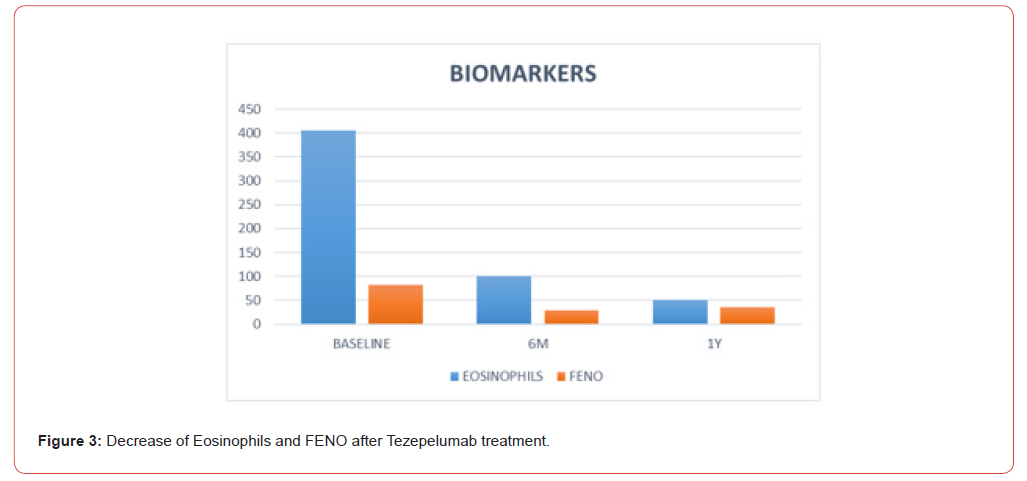
In this series of cases, we found better benefits of the treatment like significant reduction in annual exacerbations, improved ACT scores and lung function, corticosteroid decrease and discontinued and effective in both biologic nave and prior biologic experienced patients. Is important to mentioned that in all of kind of biologics as a treatment since omalizumab the clinicals trials and real world data demonstrated in many results that biologic treatment safe and don’t have severe or serious side effects and the studies mentioned and demonstrated in conclusion they are very safe, but we need to mentioned some symptoms that patients mentioned like: Sore throat, join/back pain and injection site reactions.
Conclusion
We presented a series of cases with severe asthma and descriptive response to tezepelumab in real world, we observed significant lessening of exacerbations and improved asthma control, quality of life in symptoms and lung function tests after 6 months to a year with Tezepelumab therapy in patients with failure to other biological drugs. In our study. Tezepelumab was effective in patients with or without allergies as well in mixed phenotypes as with or without high T2, T2 low or No TH2.
Acknowledgement
None.
Conflict of Interest
No Conflict of interest.
References
- Khateed Jasmin (2025) Real- world evidence of Tezepelumab for severe asthma - a retrospective multicenter cohort. ERJ Open Research 11(5): 00314-2025.
- Biener L (2024) Real-World Data on Tezepelumab in Patients with Severe Asthma in Germany. Journal of Allergy and Clinical Immunology: In Practice.
- Chung KF (2014) International ERS/ATS guidelines on definition, evaluation, and treatment of severe asthma. European Respiratory Journal 43(2): 343–73.
- Corren J (2017) Tezepelumab in Adults with Uncontrolled Asthma. New England Journal of Medicine 377(10): 936–46.
- Lipworth BJ (2025) Tezepelumab in Adults with Severe Chronic Rhinosinusitis with Nasal Polyps. New England Journal of Medicine.
- Wechsler ME (2024) Clinical response and on-treatment clinical remission with tezepelumab in a broad population of patients. The European respiratory journal 26: 64(6).
- Nopsopon T (2023) Comparative efficacy of tezepelumab to mepolizumab, benralizumab, and dupilumab in eosinophilic asthma: A Bayesian network meta-analysis. The Journal of Allergy and clinical immunology.
- Betancor D (2024) Four-month real-life response to Tezepelumab in patients with multi-failure to other biologics. Allergologia et immunopathologia.
- Plaza V (2023) Efficacy and Potential Positioning of Tezepelumab in the Treatment of Severe Asthma. Open respiratory archives.
- Canonica GW (2023) Severe Asthma Network Italy Definition of Clinical Remission in Severe Asthma: A Delphi Consensus. The journal of allergy and clinical immunology in practice.
- Matucci A (2023) Severe asthma and biologics: Managing complex patients. J Investig Allergol Clin Immunol.
- Miralles-López J (2024) PositioninG of tezepelumab in severe asthma. J Investig Allergol Clin Immunol.
-
Herrera García JC*. Tezepelumab in Mexican Patients with Severe Asthma TH2 predominant: A series of cases. Archives in Respiratory & Pulmonary Medicine. 1(4): 2025. ARPM.MS.ID.000518.
-
Severe asthma; Biologic therapy; Tezepelumab; Cases
-

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.