Research Article
Research Article
Post-Acute Care for Chronically Critically Ill
Kathleen J Haley1*, Jacinta Lomba1, Robert N Nace2, Anthony F Massaro1 and Karl Laskowski1
1 Brigham and Women’s Hospital, Boston, MA
2 Spaulding Hospital Cambridge
K Haley, Brigham and Women’s Hospital, Boston, MA, USA.
Received Date: October 17, 2025; Published Date:October 31, 2025
Abstract
Patients dependent on advanced technology for survival are referred to as “chronically critically ill.” Their care often lacks continuity due to transfers between various services within and between hospitals. Disrupted care transitions may result in less effective management. A new care model was developed to improve continuity of care between the acute care hospital and the long-term acute care hospital [LTACH] by utilizing a multidisciplinary team that operates in both settings. The model has been well-received by patients, families, and staff at both institutions. Patients managed under this model have experienced reduced thirty-day readmission rates at the acute hospital despite stable Elixhauser scores. These findings suggest that an approach emphasizing continuity of care between institutions is an effective strategy for managing chronically critically ill patients.
Keywords:Chronic critical illness; Continuity of care; Long-term acute care
Introduction
Over the past two decades, the management of critically ill patients has advanced significantly, leading to reduced mortality in this patient population. These improvements have resulted in the emergence of a new patient population: survivors of acute crises who continue to experience severe organ dysfunction. These individuals remain dependent on advanced medical technology such as mechanical ventilators or hemodialysis and have been termed “chronically critically ill” [1]. This population is highly vulnerable, with one-year mortality approaching 50% [2-4], and only one in ten reaching functional independence one year after their initial illness [5-7]. Caring for these patients demands substantial resources: although fewer than 10% of critically ill patients develop chronic critical illness, they account for 30% of total intensive care unit occupancy, and annual hospital costs exceed 20 billion dollars [8].
It is widely recognized that current acute critical illness management approaches have many systemic limitations that negatively impact the care of chronically critically patients [2,9,10]. One of the most important limitations is suboptimal continuity of care. Constant team rotations, frequent intra- and inter-facility transfers, and interruptions in long-term provider involvement make it challenging to maintain consistent patient care and effective communication with families. Extended hospital stays frequently result in primary care physicians losing contact with their patients, who then interact with multiple care teams across different facilities. These ongoing transitions strain therapeutic relationships and complicate discussions about prognosis. In addition, despite efforts to maintain consistent and accurate data transmission for these patients, the multiple clinical hand-offs often result in loss of critical patient information [11-13].
Prior research aimed at improving communication and continuity of care between acute hospitals and long-term acute care hospitals [LTACHs] has produced mixed results regarding patient outcomes and hospital utilization rates [14-17]. These interventions typically focused on increasing the amount and consistency of information shared with patients, families, and providers. Although the studied interventions often achieved the goal of enhanced communication [15-19], the impact on patient outcomes and therapeutic goals varied [17-21]. Notably, interventions that facilitated the bedside team’s ability to establish a therapeutic alliance with patients and families were associated with greater changes in patient outcomes and decision-making.
In 2013, the Brigham and Women’s Physician Organization (BWPO) allocated seed funds for care redesign projects. Our team received funds through this program to develop a new care delivery model to address the management challenges for chronically critically patients, particularly the challenge of maintaining continuity of care. This model involved forming an integrated, multidisciplinary team to connect the acute hospital with the LTACH. The initiative centered around providing ongoing support to clinicians at both sites and establishing long-term therapeutic relationships with patients and their families. We hypothesized that this approach would improve outcomes for these chronically critically ill patients. Here, we describe our approach to the longitudinal management of patients with chronic critical illness and the results of the program.
Methods/Approach
The pilot project was designed as a Quality Improvement (QI) initiative at the Brigham and Women’s hospital, and as such the initial clinical work was not formally supervised by the Institutional Review Board (RB) per their policies. The subsequent protocol for the research analyses was approved in advance by the IRB. The analysis of the results was performed after the completion of the initial Quality Improvement pilot program. The Brigham and Women’s Hospital (BWH) is a 793-bed tertiary care hospital located in Boston, MA. Our care redesign focused on patients initially treated in the 20-bed BWH medical intensive care unit (MICU) and then transferred to an LTACH. A review of the BWH MICU patients previously discharged to the LTACH level of care determined that Spaulding Hospital Cambridge (SHC) received the largest percentage of MICU patients for post-ICU care. Based on these findings, the intervention focused on patients transferred to SHC after a MICU hospitalization of at least one overnight stay. Our care redesign project was named The Integrated, Patient-Centered Care in Chronic Critical Illness (IP4CI).
Patient Population
The IP4CI program included all patients who received care in the BWH MICU for at least one overnight and then were transferred to SHC for rehabilitation, except for the lung transplant patients. These patients were excluded because these patients had a preexisting robust program for post-acute hospital management. The control group consisted of all BWH MICU patients with at least an overnight stay in the two years before the start of the IP4CI program. The acuity of the patients in both groups was assessed by comparing the Elixhauser comorbidity index scores and by comparing the use of mechanical ventilation and renal replacement therapy in both groups of patients. The Elixhauser comorbidity index is an established assessment of patient comorbidities and has been shown to be predictive of patient prognosis in multiple settings [22-26].
Functional Assessment
We used the Activity Measure Post Acute Care basic mobility form (AMPAC) to evaluate the functional status of our patients. The short form AMPAC is a well-validated tool [27,28] that uses six common activities of daily living to determine functional status: turning in bed, moving from the supine to a seated position in the bed, moving from bed to chair, sitting in a chair, walking in the room, and climbing stairs. Some of the IP4CI patients were so debilitated that they could not attempt even the most basic movement scored by the AMPAC. We therefore modified the scale to include “0” for assessing these extremely weak patients. Our modified AMPAC scores ranged from 0 – 24.
Statistical Analyses
Differences in categorical variables were compared by Fisher exact tests. Continuous variables were compared using Student’s t-tests. Nonparametric data were compared using the Mann- Whitney test. Multivariate logistic regression was used to identify associations among variables. Differences were accepted as significant when P < 0.05. The logistic regression model “goodness of fit” was assessed by the Hosmer-Lemeshow test.
IP4CI Program - Development
Key stakeholders were engaged at both institutions for collaborative planning and to align incentives. The primary goals were to 1) improve the continuity of care for chronically critically ill patients, 2) improve communication between the acute hospital and the LTACH, and 3) align the care plan with achievable functional and medical goals. During the planning for this project, we focused on three areas: needs assessment, procedure development, and identification of objective outcomes.
A needs assessment was conducted using surveys of clinicians at both the acute hospital and the LTACH. The results indicated that most clinicians at both institutions considered clear communication between the acute hospital and LTACH to be an important factor in providing care for chronically critically ill patients, and the assessment identified notable communication gaps between the two facilities.
For the development of this procedure, a new two-person team was proposed to maintain communication between the MICU and LTACH. This “continuity team” met with clinicians, patients, and families prior to transfer from the acute hospital to LTACH in order to establish therapeutic goals aligned with the patient’s values. After transfer, the IP4CI team promoted continuity by meeting with LTACH hospitalists weekly to assess each patient’s progress toward these goals and remained in contact with patients and their families. Weekly team meetings were held to develop, review, and refine processes using feedback from patients and clinicians.
The outcome measures were developed by the stakeholder group in conjunction with the BWPO and by identifying consistent themes in the discussion with patients. Two outcome targets were identified. The first was a 25% decrease in the early readmission rate from LTACH to an acute hospital, defined as an admission lasting at least 24 hours that occurred within thirty days of discharge from the acute care facility. The second outcome was discharge to home from the LTACH, which was identified by most of the patients as their most important goal.
IP4CI Approach - Implementation
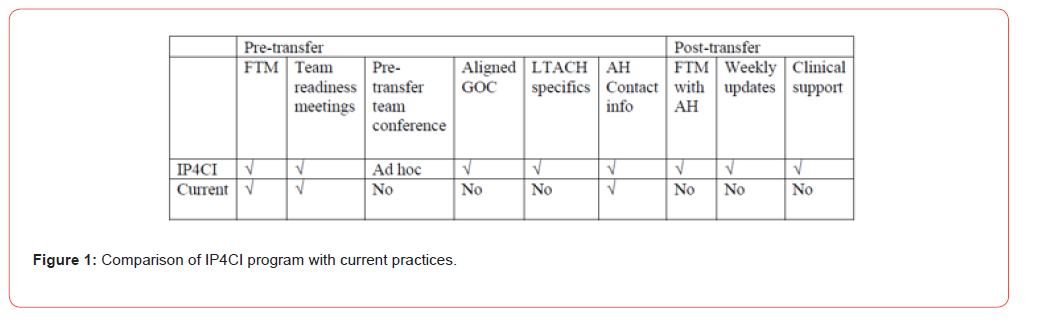
Figure 1 provides a summary of the IP4CI approach alongside a comparison to usual care. To facilitate effective communication between the acute facility and the LTACH, several strategies were implemented. First, the “continuity team” was charged with facilitating communication between the acute facility and the LTACH. Second, we initiated weekly discussions between acute care MICU and LTACH clinicians to provide a forum for ongoing communication of specific patient information and responses to therapy. One of the acute care MICU clinicians was available to LTACH providers via pager 24/7 to address questions arising at the LTACH and to help with any significant new issues. This same physician was also available to the BWH Emergency Department to address questions regarding program patients having evaluations there and to provide ED physicians with information about the care available at the LTACH. Third, following the implementation of the IP4CI program, the stakeholders continued to meet weekly to review implementation issues and to discuss any areas of concern.
Prior to the acute hospital to LTACH transfer, the “continuity team” met with the clinical team at the acute hospital and also provided the patients and families individualized counseling regarding the environment and care they would receive while at the LTACH. These meetings identified achievable therapeutic goals and identified any issues that might hinder a successful LTACH transfer. To provide a consistent structure, rigor, and transparency to these conversations, the continuity team used the “Serious Illness Conversation Guide”, which was developed by the Palliative Care Consultation service at the Dana Farber Cancer Institute as a guide for physicians discussing life-threatening illness with their patients [29]. While initially focused on guiding conversations with patients who have a cancer diagnosis, the “Serious Illness Conversation Guide” has been successfully adapted for use in other life-threatening illnesses [30,31]. These meetings were designed to provide transition coaching in preparation for the transfer to LTACH, so that the patients and their families were prepared for the differences they would experience following transfer from the acute hospital to LTACH.
The continuity team remained in contact with the patients and their families after LTACH transfer. Additionally, the continuity team maintained regular communication with the LTACH clinicians via weekly patient progression reviews and were available via email and pager for questions that arose outside of the regular conferences. Thus, the continuity team provided the LTACH clinicians, the patients, and their families a longitudinal perspective on their illness and convalescence.
Weekly stakeholder operations reviews have proven to be an important component of the program. Some issues that impacted the effectiveness of the program only became apparent after initial implementation. For example, these meetings identified that real-time, effective communication with the BWH Emergency Department required the development of a specific novel mechanism to facilitate these conversations. The regular operations meetings also allowed the program to use real-time feedback to “fine tune” the operational procedures. Finally, the operations meetings also enabled the teams to quickly adjust to hospital organizational changes, and to incorporate new team members efficiently.
Results
Patient Characteristics
Patient characteristics before and during the initiation of the IP4CI program are provided in Table 1. Compared to the two years prior to the start of the IP4CI program, the distribution of patients’ age, gender and ethnicity was not statistically different after the initiation of the IP4CI program (Table 1). The BWH is a tertiary care facility, and this is reflected in the high Elixhauser comorbidity index scores in our patients, which were available for 110 control patients and the first 250 IP4CI patients. The average Elixhauser comorbidity index scores in the control patients (10.25 ± 6.5) and after the IP4CI program (10 ± 7.6) were not significantly different. This indicates that patient acuity remained stable before and after the IP4CI program was started. There were more patients transferred to LTACH on mechanical ventilation following the start of the IP4CI program compared to the control period, but this did not reach statistical significance. The number of patients transferred to LTACH on renal replacement therapy was significantly greater after the start of the IP4CI program [P < 0.05] (Table 1).
Program Outcomes
The early (<30 days after acute hospital transfer to LTACH) readmission rate back to the BWH MICU in fiscal year 2012, prior to the IP4CI program, was 40%. For the first 241 patients followed by the IP4CI program, the early readmission rate was 28%. Of note, the decreased early readmission rate has been sustained over the subsequent years of the program. Together with the stable Elixhauser score, as shown above, this indicates that the decreased readmission rate achieved by the IP4CI program was not due to any decrease in the acuity of the patients sent to LTACH.
The characteristics of the IP4CI patients are outlined in Table 2. 58 of the 241 patients expired during their LTACH admission. Compared to patients who survived their LTACH admission, the patients who did not survive had significantly greater rates of mechanical ventilation at the time of transfer, more frequent early readmission to the acute hospital, more transfers between the LTACH and the acute hospital, and a longer length of stay at the acute hospital (Table 2). Logistic regression analysis identified significant associations between death during LTACH admission and the following variables: requirement for mechanical ventilation at the time of transfer to LTACH (P <0.05), readmission to the acute hospital less than 30 days following transfer to LTACH (P < 0.01), and multiple readmissions to the acute hospital from LTACH (P < 0.01) (Table 2).
Table 1: Demographics of patients hospitalised for first episode of wheeze pre-COVID and during COVID
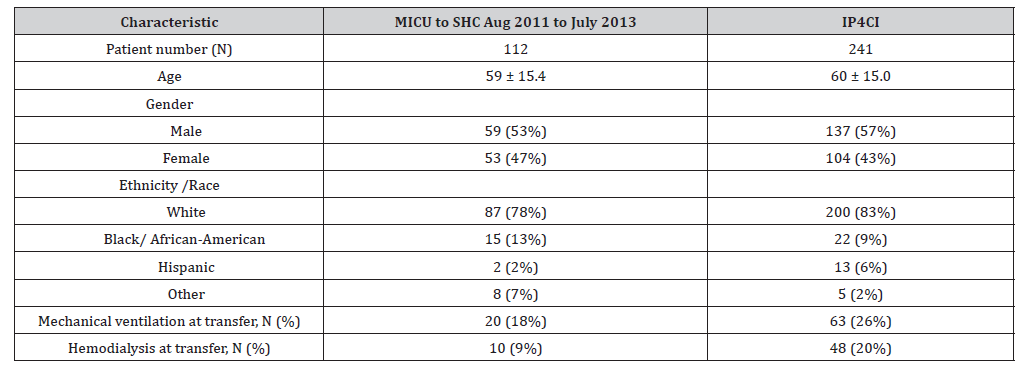
Table 2: Demographics of patients hospitalised for first episode of wheeze pre-COVID and during COVID
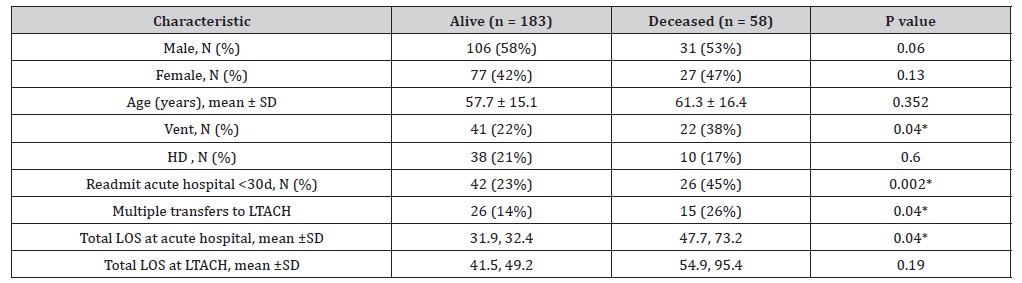
Abbreviations: N = number of patients, SD = standard deviation, Vent = mechanical ventilator support at time of transfer, HD = hemodialysis at time of transfer, d = days, LTACH = long-term acute care hospital, LOS = length of stay, * designates statistical significance with P < 0.05.
30% of the IP4CI patients were discharged directly to home from the LTACH. A LTACH admission of at least 30 consecutive days was significantly associated with discharge to home (P < 0.001, Table 3). Additionally, the patients who did not have a home discharge had statistically longer lengths of stay at the acute hospital (P < 0.05, Table 3). Logistic regression analysis showed a significant association (P = 0.004) between a LTACH length of stay of at least 30 days and discharge to home. Of note, multiple transfers from the acute hospital to LTACH were not correlated with discharge to home. 8 of the 73 patients with a home discharge had multiple LTACH transfers compared to 33 of 168 patients who were not discharged to home. Negative predictors of a home discharge included a requirement for mechanical ventilation at the time of transfer from the acute hospital (Table 3).
Table 3: Demographics of patients hospitalised for first episode of wheeze pre-COVID and during COVID
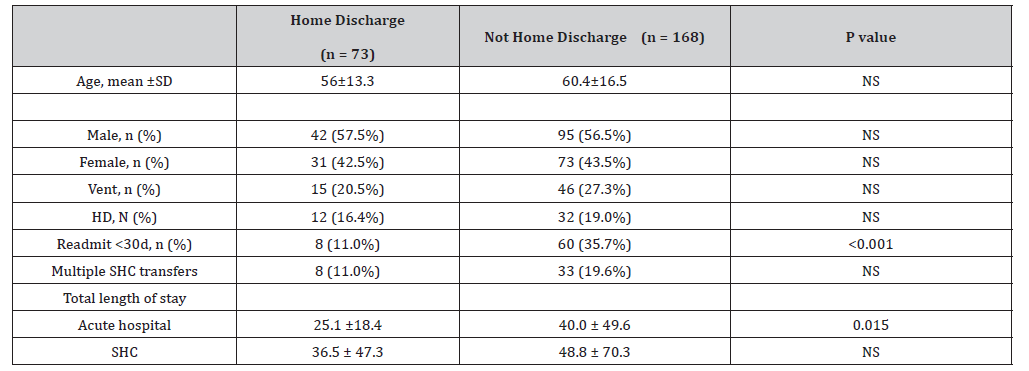
Abbreviations: N = number, SD = standard deviation, NS = not statistically significant, SHC = Spaulding Hospital Cambridge, Vent = mechanical ventilation support at time of transfer, HD = hemodialysis at time of transfer, * designates statistically significant with P < 0.05.
The Importance of Functional Outcomes
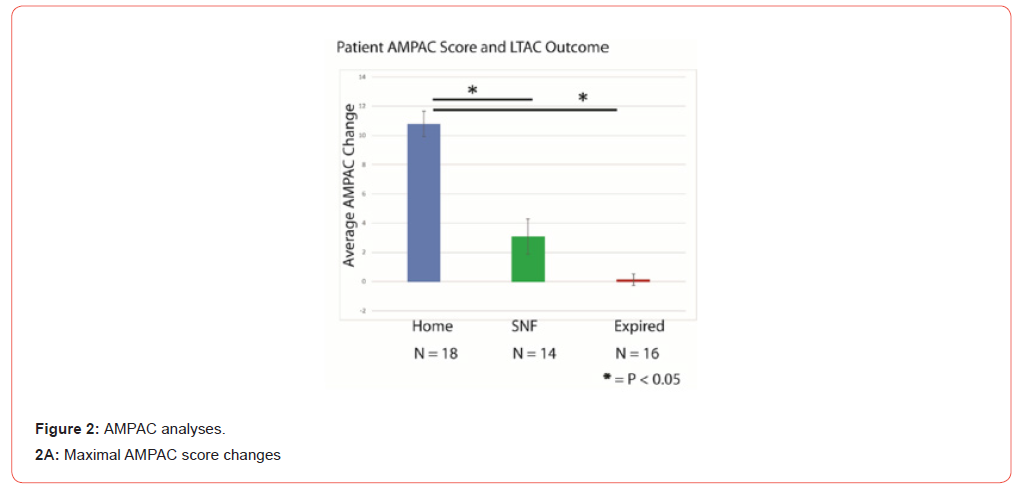
We tested multiple potential clinical factors to determine possible associations with a home discharge. Unexpectedly, factors that have been shown to be predictive of outcome in acutely critically ill patients, such as serum albumin [32,33] and Provent Scores [34], were not predictive of this outcome in our population [data not shown]. We next evaluated functional status, which has been shown to be a key predictor of clinical outcomes in acutely critically ill patients, including stroke survivors and patients requiring long-term mechanical ventilation [35-37]. We used the AMPAC basic score to compare the functional status of a subgroup of 48 IP4CI patients to determine if the functional status at LTACH transfer or changes in functional status while at LTACH predicted clinical outcome in our chronically critically ill patients. The 48 patients in this subgroup included 18 patients discharged to home, 14 patients discharged to a skilled nursing facility [SNF] and 16 patients who expired during their LTACH admission.
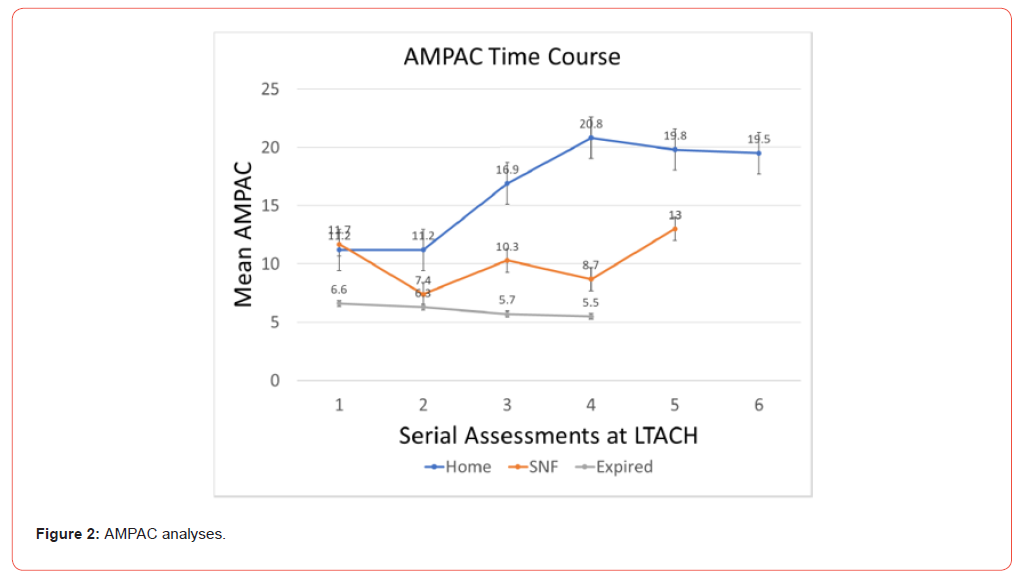
The initial AMPAC score for all three groups was very low: 11.2 ± 3.7 [average and standard deviation] for patients discharging to home, 11.7 ± 3.9 for patients discharging to SNF and even lower for patients who expired during their LTACH admission: 6.3 ± 5.6. The change in the AMPAC score during the LTACH admission was calculated from the Physical and Occupational Therapists’ interval assessments, which occurred approximately every two weeks. The patients discharged to home had a significantly greater increase in their AMPAC score while at LTACH compared to patients who either discharged to SNF or who expired during their LTACH admission (Figure 2A). As shown in Figure 2B, the AMPAC scores of the three groups diverged by the second LTACH assessment, with the patients who discharged to home increasing to an average AMPAC 16.9 ±4.6, the patients discharging to SNF with a significantly smaller increase to 10.3 ±4.4 and the patients who expired during their LTACH admission with little change from their admission AMPAC: 5.6 ±2.3 (Figure 2A & 2B).
Discussion
This report introduces an innovative care model for chronically critically ill patients: the IP4CI program. This approach utilises a dedicated team that coordinates care between clinical staff at the acute facility and LTACH to provide a comprehensive, longitudinal view of patient progress. Through sustained engagement with LTACH clinicians and patients, the IP4CI program improves continuity of care for individuals who often experience gaps in connection with their primary providers. This high continuity, longitudinal approach enhances the development of therapeutic relationships with patients and facilitates periodic reassessments of the therapeutic plan to maintain alignment between the treatment plan and the patient’s goals. By providing ongoing clinical support to the LTACH providers, our approach also strengthens the collaboration between the acute hospital and the LTACH.
This approach has been well-received by patients, their families, and clinicians at both the acute hospital and the LTACH. One of the SHC hospitalists told our clinician that with the IP4CI program, “you know that you’re never alone”. Additionally, this approach has been associated with a sustained decrease in the early readmission rate from the LTACH to the acute hospital. Our high continuity approach builds on the work of earlier investigators who identified the important role of palliative care consultation and family communication for critically ill patients [20, 38-47]. While several different models are used for such consultation [41], the most effective approaches typically involve repeated interactions between the consultant and the treatment team and/or family over time. In contrast, models that are not characterized by repeated interactions have been less effective [17, 19]. We speculate that the longitudinal relationship developed between our continuity team and the program patients supported effective therapeutic alliances and thereby enhanced the care for these chronically critically ill patients.
An unexpected finding was the absence of an association between variables previously shown to correlate with outcomes in acutely critically patients and clinical outcomes in our cohort. It is possible that the differences in our cohort from prior investigations were caused by the established chronic critical illness in our patients. Thus, it is possible that in such a debilitated population, the discriminatory power of the assessments that were predictive in acutely critically ill patients was lost.
Another unexpected finding was the association between a LTACH admission of at least thirty days and the eventual discharge to home. While this might be caused by healthier patients being able to remain at LTACH at least thirty days, we speculate that additional factors modulated patient outcomes in our cohort. Decreasing the early (<30 day) readmission rate was a primary outcome metric of the IP4CI program. Indeed, the clinical support provided to the LTACH assisted the LTACH hospitalists in keeping patients who otherwise would have been readmitted to the acute hospital. We speculate that the association between a LTACH stay of at least 30 days and discharge to home could reflect the effects of uninterrupted intensive physical and occupational therapy provided to the patients at LTACH. The patients at SHC receive physical rehabilitation at least five days a week. This sustained focus on regaining strength and endurance potentially allowed patients to regain functional capacity and to resume enough activities of daily living as to make a discharge to home feasible. Our functional data support this contention. All the IP4CI patients arrived at LTACH with low AMPAC scores. The patients who were discharged to home showed significant and sustained improvement, but this improvement became apparent only after approximately 4 weeks at the LTACH. Previous investigators have also identified the functional status as a crucial determinant of patient outcome [37,48,49]. Our work builds on their findings by showing that sustained improvement in functional status takes several weeks to become apparent. Our data also show that both the total amount of improvement and the pattern of functional recovery is predictive of patient outcome. Based on the results from our patients, we propose that having regular physical and occupational therapy for at least 30 days contributed substantially to these patient’s eventual home discharge.
There are multiple limitations to our approach, primarily related to the question of generalizability. This is the experience of a single program. Also, the initial patient population in our IP4CI program included only MICU patients, and so this approach may not generalize to other chronically critically ill patients.
Our program also possesses multiple strengths. Ironically, one of the strengths of our approach is also a limitation. The longitudinal approach we implemented depends heavily on the relationships between the IP4CI team, the patient, the patient’s family, the MICU clinicians, and the LTACH clinicians. This approach can therefore be rendered ineffective by the disruption of these relationships. We have addressed this limitation in two ways. First, the IP4CI team meets weekly to discuss programmatic issues, and to facilitate the onboarding of new team members. These meetings maintain team cohesion and facilitate the adjustment of processes that are not functioning optimally. Second, video conferences are used to support IP4CI team participation in family conferences at the LTACH. These face-to-face interactions have served as a means to maintain direct communication, which is considered important for this program. These two approaches have allowed the IP4CI team to maintain productive and therapeutic relationships with our patients and the LTACH clinicians over their prolonged LTACH admissions. Our strong team approach has proven durable and has allowed us to sustain this program despite multiple personnel and funding changes.
Conclusion
In summary, patients with chronic critical illness are a highly vulnerable population with multiple comorbid processes. Their care is characterized by frequent changes in healthcare providers. The poor continuity of care exacerbates the management difficulties for these patients. We present a new care paradigm to improve the continuity of care for these patients. Our innovative approach is associated with sustained decreases in the early readmission rate for LTACH patients without significant changes in mortality rates. We propose that an approach that maintains high continuity of care for chronically critically patients is feasible and may improve outcomes for these patients.
Funding
This work was supported by the Brigham Care Redesign Incubator and Start-Up Program [BCRISP]. This funding source was not involved in the development, writing, or decision to submit this work for publication.
Acknowledgments
The authors thank Mr. Gabriel Santos, Ms. Lantharra Langlois and Ms Christina Exilhomme for their assistance in data collection and Drs. Hillary Goldberg and Rachel Putman for their thoughtful comments during the manuscript preparation. The authors also thank the members of the IP4CI team, including Jill Pedrick, Andre Remillard, Caitlyn Ryan, and Julie Zuis.
References
- K Girard, T A Raffin (1985) The chronically critically ill: to save or let die? Respir Care 30: 339–347.
- J E Nelson, C E Cox, AA Hope, SS Carson (2010) Chronic critical illness. Am J Respir Crit Care Med 182: 446–454.
- D J Scheinhorn, DC Chao, MS Hassenpflug, DR Gracey (2001) Post-ICU weaning from mechanical ventilation: the role of long-term facilities. Chest 120: 482S–484S.
- DJ Scheinhorn (1999) Outcomes after acute care: analysis of 133 mechanically ventilated patients. Am J Respir Crit Care Med 160: 1788–1789.
- MS Herridge, Catherine M Tansey, Andrea Matté, George Tomlinson, Natalia Diaz-Granados, et al. (2011) Functional disability 5 years after acute respiratory distress syndrome. The New England journal of medicine 364: 1293–1304.
- MS Herridge, Angela M Cheung, Catherine M Tansey, Andrea Matte-Martyn, Natalia Diaz-Granados, et al. (2003) One-year outcomes in survivors of the acute respiratory distress syndrome. The New England journal of medicine 348: 683–693.
- MS Herridge (2011) Recovery and long-term outcome in acute respiratory distress syndrome. Crit Care Clin 27: 685–704.
- JM Kahn, Tri Le, Derek C Angus, Christopher E Cox, Catherine L Hough, Douglas B White, et al. (2015) The epidemiology of chronic critical illness in the United States. Crit Care Med 43: 282–287.
- JM Kahn, SS Carson (2013) Generating evidence on best practice in long-term acute care hospitals. JAMA 309: 719–720.
- DJ Scheinhorn (2000) Outcomes of post-intensive care unit care: once more, the half-full/empty glass. Crit Care Med 28: 257–258.
- S Studeny, Lauren Burley, Kelsey Cowen, Melanie Akers, Kelly O'Neill, et al. (2017) Quality improvement regarding handoff. SAGE Open Med 5: 2050312117729098.
- C Fryman, C Hamo, S Raghavan, N Goolsarran (2017) A Quality Improvement Approach to Standardization and Sustainability of the Hand-off Process. BMJ Qual Improv Rep 6.
- NA Rattray, Patricia Ebright, Mindy E Flanagan, Laura G Militello, Paul Barach, et al. (2018) Content counts, but context makes the difference in developing expertise: a qualitative study of how residents learn end of shift handoffs. BMC Med Educ 18: 249.
- EB Rudy, B J Daly, S Douglas, H D Montenegro, R Song et al. (1995) Patient outcomes for the chronically critically ill: special care unit versus intensive care unit. Nurs Res 44: 324–331.
- CE Cox, Carmen L Lewis, Laura C Hanson, Catherine L Hough, Jeremy M Kahn, et al. (2012) Development and pilot testing of a decision aid for surrogates of patients with prolonged mechanical ventilation. Crit Care Med 40: 2327–2334.
- SS Carson, Maihan Vu, Marion Danis, Sharon L Camhi, Leslie P Scheunemann, et al. (2012) Development and validation of a printed information brochure for families of chronically critically ill patients. Crit Care Med 40: 73–78.
- SS Carson, Christopher E Cox, Sylvan Wallenstein, Laura C Hanson, Marion Danis, et al. (2016) Effect of Palliative Care-Led Meetings for Families of Patients With Chronic Critical Illness: A Randomized Clinical Trial. JAMA 316: 51–62.
- N Braus, Toby C Campbell, Kristine L Kwekkeboom, Susan Ferguson, Carrie Harvey, et al. (2016) Prospective study of a proactive palliative care rounding intervention in a medical ICU. Intensive Care Med 42: 54–62.
- (1995) A controlled trial to improve care for seriously ill hospitalized patients. The study to understand prognoses and preferences for outcomes and risks of treatments (SUPPORT). The SUPPORT Principal Investigators. JAMA 274: 1591–1598.
- A Lautrette, Michael Darmon, Bruno Megarbane, Luc Marie Joly, Sylvie Chevret, et al. (2007) A communication strategy and brochure for relatives of patients dying in the ICU. The New England journal of medicine 356: 469–478.
- A Lautrette, M Ciroldi, H Ksibi, Elie Azoulay (2006) Azoulay, End-of-life family conferences: rooted in the evidence. Crit Care Med 34: S364–372.
- A Elixhauser, C Steiner, DR Harris, RM Coffey (1998) Comorbidity measures for use with administrative data. Med Care 36: 8–27.
- ME Menendez, V Neuhaus, CN van Dijk, D Ring (2014) The Elixhauser comorbidity method outperforms the Charlson index in predicting inpatient death after orthopaedic surgery. Clin Orthop Relat Res 472: 2878–2886.
- NR Thompson, Youran Fan, Jarrod E Dalton, Lara Jehi, Benjamin P Rosenbaum, et al. (2015) A new Elixhauser-based comorbidity summary measure to predict in-hospital mortality. Med Care 53: 374–379.
- C van Walraven, PC Austin, A Jennings, H Quan, AJ Forster (2009) A modification of the Elixhauser comorbidity measures into a point system for hospital death using administrative data. Med Care 47: 626–633.
- F Zhang, Yida Chiu, Joie Ensor, Mohamed O Mohamed, George Peat, et al. (2022) Elixhauser outperformed Charlson comorbidity index in prognostic value after ACS: insights from a national registry. J Clin Epidemiol 141: 26–35.
- S M Haley, Wendy J Coster, Patricia L Andres, Larry H Ludlow, Pengsheg Ni, et al. (2004) Activity outcome measurement for postacute care. Med Care 42: I49–61.
- DU Jette, Mary Stilphen, Vinoth K Ranganathan, Sandra D Passek, Frederick S Frost, et al. (2014) Validity of the AM-PAC "6-Clicks" inpatient daily activity and basic mobility short forms. Phys Ther 94: 379–391.
- R Bernacki, Mathilde Hutchings, Judith Vick, Grant Smith, Joanna Paladino, et al. (2015) Development of the Serious Illness Care Program: a randomised controlled trial of a palliative care communication intervention. BMJ Open 5: e009032.
- Z Cooper, , Luca A Koritsanszky, Christy E Cauley, Julia L Frydman, Rachelle E Bernacki, et al. (2016) Recommendations for Best Communication Practices to Facilitate Goal-concordant Care for Seriously Ill Older Patients with Emergency Surgical Conditions. Ann Surg 263: 1–6.
- Z Cooper, Katherine Corso, Rachelle Bernacki, Angela Bader, Atul Gawande, et al. (2014) Conversations about treatment preferences before high-risk surgery: a pilot study in the preoperative testing center. J Palliat Med 17: 701–707.
- B Can, Ozgur Kara, Muhammet Cemal Kizilarslanoglu, Gunes Arik, Gozde Sengul Aycicek, et al. (2017) Serum markers of inflammation and oxidative stress in sarcopenia. Aging Clin Exp Res 29: 745–752.
- JM Clochesy, BJ Daly, HD Montenegro (1995) Montenegro, Weaning chronically critically ill adults from mechanical ventilatory support: a descriptive study. Am J Crit Care 4: 93–99.
- SS Carson, Jeremy M Kahn, Catherine L Hough, Eric J Seeley, Douglas B White, et al. (2012) A multicenter mortality prediction model for patients receiving prolonged mechanical ventilation. Crit Care Med 40: 1171–1176.
- FH Chang, P Ni, AM Jette (2014) Does activity limitation predict discharge destination for postacute care patients? Am J Phys Med Rehabil 93: 782–790.
- SM Haley, Pengsheng Ni, Jin-Shei Lai, Feng Tian, Wendy J Coster, et al. (2011) Linking the activity measure for post acute care and the quality of life outcomes in neurological disorders. Arch Phys Med Rehabil 92: S37–43.
- A Jubran, Brydon J B Grant, Lisa A Duffner, Eileen G Collins, Dorothy M Lanuza, et al. (2019) Long-Term Outcome After Prolonged Mechanical Ventilation: A Long-Term Acute-Care Hospital Study. Am J Respir Crit Care Med 199(12):1508-1516.
- PD Treece, Ruth A Engelberg, Sarah E Shannon, Elizabeth L Nielsen, Theresa Braungardt, et al. (2006) Integrating palliative and critical care: description of an intervention. Crit Care Med 34: S380–387.
- JE Nelson, Kusum S Mathews, David E Weissman, Karen J Brasel, Margaret Campbell et al. (2015) Integration of palliative care in the context of rapid response: a report from the Improving Palliative Care in the ICU advisory board. Chest 147: 560–569.
- JE Nelson, J Randall Curtis, Colleen Mulkerin, Margaret Campbell, Dana R Lustbader, et al. (2013) Choosing and using screening criteria for palliative care consultation in the ICU: a report from the Improving Palliative Care in the ICU (IPAL-ICU) Advisory Board. Crit Care Med 41: 2318–2327.
- JE Nelson et al. (2010) Models for structuring a clinical initiative to enhance palliative care in the intensive care unit: a report from the IPAL-ICU Project (Improving Palliative Care in the ICU). Crit Care Med 38: 1765–1772.
- R. Curtis et al. (2016) Randomized Trial of Communication Facilitators to Reduce Family Distress and Intensity of End-of-Life Care. Am J Respir Crit Care Med 193: 154–162.
- JR Curtis, Patsy D Treece, Elizabeth L Nielsen, Lois Downey, Sarah E Shannon, et al. (2008) Integrating palliative and critical care: evaluation of a quality-improvement intervention. Am J Respir Crit Care Med 178: 269–275.
- JR Curtis et al. (2011) Effect of a quality-improvement intervention on end-of-life care in the intensive care unit: a randomized trial. Am J Respir Crit Care Med 183: 348–355.
- JR Curtis et al. (2012) Development and evaluation of an interprofessional communication intervention to improve family outcomes in the ICU. Contemp Clin Trials 33: 1245–1254.
- CM Lilly, LA Sonna, KJ Haley, AF Massaro (2003) Intensive communication: four-year follow-up from a clinical practice study. Crit Care Med 31: S394–399.
- CM Lilly, D L De Meo, L A Sonna, K J Haley, A F Massaro, et al. (2000) An intensive communication intervention for the critically ill. Am J Med 109: 469–475.
- DU Jette, Mary Stilphen 2, Vinoth K Ranganathan 3, Sandra D Passek 4, Frederick S Frost, et al. (2014) AM-PAC "6-Clicks" functional assessment scores predict acute care hospital discharge destination. Phys Ther 94: 1252–1261.
- B Joseph, FS Jehan (2017) The Mobility and Impact of Frailty in the Intensive Care Unit. Surg Clin North Am 97: 1199–1213.
-
Kathleen J Haley*, Jacinta Lomba1, Robert N Nace, Anthony F Massaro and Karl Laskowski. Post-Acute Care for Chronically Critically Ill Patients – Building a Bridge. Archives in Respiratory & Pulmonary Medicine. 1(4): 2025. ARPM.MS.ID.000519.
-
Chronic critical illness; Continuity of care; Long-term acute care
-

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.