Case Report
Case Report
Investigating Fatigue and Exercise Intolerance in a
University Immunology Clinic
Julian L Ambrus1*, Paul Isackson2, Molly Moore3, John Butsch3 and Lucia Balos4
1Departments of Medicine, SUNY at Buffalo School of Medicine, USA
2Department of Pediatrics, SUNY at Buffalo School of Medicine, USA
3Department of Surgery, SUNY at Buffalo School of Medicine, USA
4Department of Pathology, SUNY at Buffalo School of Medicine, USA
Julian L Ambrus1*, Paul Isackson2, Molly Moore3, John Butsch3 and Lucia Balos4
1Departments of Medicine, SUNY at Buffalo School of Medicine, USA
2Department of Pediatrics, SUNY at Buffalo School of Medicine, USA
3Department of Surgery, SUNY at Buffalo School of Medicine, USA
4Department of Pathology, SUNY at Buffalo School of Medicine, USA
Julian L Ambrus, Departments of Medicine, SUNY at Buffalo School of Medicine, USA.
Received Date: January 31, 2020; Published Date: February 18, 2020
Abstract
Purpose: This manuscript reviews the experience of a University Immunology clinic with the evaluation of patients with idiopathic fatigue and exercise intolerance for the presence of metabolic disorders. Laboratory, biochemical and genetic studies were utilized in the evaluation.
Recent Findings: Of the 372 patients evaluated, 95% were found to have a treatable metabolic disorder. A defect in the glycogen storage pathway was found in 78 patients. Mitochondrial disorders were found in 258 patients. Myoadenylate deaminase deficiency was found in 7 patients. Various congenital myopathies were identified in 11 patients. Inflammatory myopathies were identified in 25 patients, 6 of whom had normal muscle enzymes on the initial evaluation.
Summary: The majority of patients (95%) referred with idiopathic fatigue and exercise intolerance after extensive evaluations were found to have underlying metabolic dysfunction. Frequently associated problems included gastrointestinal dysmotility disorders, recurrent infections, Raynaud’s, migraine headaches and various autoimmune diseases. Most patients showed symptomatic improvement with treatment of their metabolic dysfunction.
Keywords: Carnitine palmitoyl transferase; Mitochondria; Glycogen Storage Disease, Myoadenylate Deaminase, Asthma; Sjogren’s syndrome; Raynaud’s; Gastrointestinal dysmotility
Introduction
The last 20 years have seen dramatic increases in medical knowledge that have changed the way that we look at various clinical disorders. One of these advances has been the appreciation that metabolism is a critical part of the functioning of every cell, tissue and organism. Metabolism is defined in the Oxford dictionary as “the chemical processes that occur within a living organism that maintain life”. Metabolism is critical for the production of adenosine triphosphate (ATP) that is necessary to drive many chemical reactions. It is involved with the production of necessary DNA, RNA and proteins. It produces various chemical messengers that are critical for communication among cells and tissues. It determines structural changes that are necessary for cell movement and function. It determines the nature of cellular behavior. Not surprisingly, metabolism is part of everything a person does and therefore alterations in metabolism have clinical implications [1]. Metabolic changes can result from inherited mutations in particular genes regulating metabolism but can also occur because of various environmental and nutritional challenges, including dietary changes, changes in temperature, changes in oxygen concentration and local or systemic inflammation that may occur because of infections or toxic environmental substances [2- 6]. Many medications influence metabolism [7-11].
The first inherited metabolic disorders were identified in young children who often had mutations in genes affecting metabolism that lead to early death [12]. More recently, it has been appreciated that primary or secondary metabolic disorders can present in adults and have a more benign prognosis [13,14]. In adults, the most common presentations for metabolic disorders are fatigue and exercise intolerance [15,16]. Other common symptoms are heat intolerance, difficulty with particular medications, such as statins, metformin and propofol, recurrent infections, bowel dysmotility disorders and accelerated osteoarthritis [17-22].
When evaluating fatigue, one has to consider sleep disorders, endocrine disorders, nutritional disorders, chronic infectious disorders, autoimmune/inflammatory disorders, cardiac disorders, respiratory disorders, psychiatric disorders, malignancies and drug reactions. Recently, adult onset metabolic disorders have been included in this differential diagnosis [23,24]. In fact, fibromyalgia and chronic fatigue syndrome have been shown to be related to metabolic disorders in particular patients [25-29].
This manuscript describes the identification of metabolic disorders in a population of 372 patients referred to a University Immunology clinic over a period of 20 years(1998-2019) by primary care doctors, rheumatologists and other medical specialists after their work up had failed to identify a cause for life altering fatigue and exercise intolerance. Many of these patients had been sent to psychiatrists with the hope that depression or another psychiatric disorder would explain their clinical findings. As we will describe below, 95% of these patients were found to have some form of treatable metabolic dysfunction. Treatment of the identified metabolic disorder lead to clinical improvement in a vast majority of the patients.
Testing
The patients described were all initially evaluated by primary care doctors, rheumatologists and other medical specialists. Profound fatigue and exercise intolerance that had led to life alterations in all patients resulted in the referral to our University clinic. Work up in all patients included complete blood counts, comprehensive metabolic profiles, urinalysis, carnitine, TSH, free T4, CPK, ammonia, lactic acid and in most cases an ischemic forearm test. An elevated lactic acid rest on more than one occasion was interpreted as indicating a reliance on glycolytic metabolism, suggestive of a mitochondrial disorder [30]. Of the patients studied, 169 (46%) had an elevated lactic acid at rest along with 167 (45%) who had an elevated CPK. Serum carnitine was low in only 17 patients (5%). Eighty patients (22%) had a history of thyroid disease, but all had normal thyroid studies at the time of their evaluation in our clinic. Thyroid hormone regulates biogenesis of mitochondria [31]. The ischemic forearm test evaluates changes in lactic acid and ammonia with exercise of the arm causing reliance on anaerobic metabolism. In a normal test, both the lactic acid and ammonia rise 3-4 times the baseline levels. In an adult, a failure of the lactic acid to rise suggests a glycogen storage disease, while a failure of the ammonia to rise suggests myoadenylate deaminase deficiency or a disorder of the urea acid cycle [32,33]. Of the 263 patients who had an ischemic forearm test, 125 were abnormal (48%). In 90 patients, findings were identified in these laboratory studies that allowed a treatment strategy to be initiated and patients did not want any further work up. Eleven patients who did not show clinical improvement with initial treatment and had genetic studies done, which included whole exome sequencing and sequencing of the mitochondrial DNA, done at GeneDx in Gaithersburg, Maryland [34,35].
The remainder of the patients had biochemical studies done from muscle biopsies. The indications for these biochemical studies were normal findings on all the initial laboratory studies, confusing findings on the initial laboratory studies, failure to respond to therapy based on the initial laboratory studies and/or curiosity of the patients to want to identify the root cause for their disorder.
Biochemical studies included the evaluation of the activity of carnitine palmitoyl transferase, myoadenylate deaminase, phosphorylase, phosphorylase b kinase, phosphofructokinase, phosphoglycerate kinase, phosphoglycerate mutase, lactate dehydrogenase, acid and neural maltase, NADH dehydrogenase, NADH cytochrome c reductase, succinate dehydrogenase, succinate cytochrome c reductase, cytochrome c oxidase and citrate synthase from freshly obtained muscle biopsies. In some patients CoQ10 levels were also assessed. These studies were performed in the Robert Guthrie Genetics Laboratory as previously described [8]. Treatment strategies were determined by the biochemical studies, when possible. Some patients with normal biochemical studies in whom no treatment strategy could be suggested went on to have genetic studies.
The histology of the muscle obtained for biochemical studies was evaluated. The vast majority of the biopsies showed only fiber atrophy (67%). Other less common findings included normal (11%), inflammatory myositis (11%), consistent with denervation (2%), Ragged red fibers or abnormal mitochondria on EM (4%), inclusion body myositis (3 patients), abnormal lipid deposition (1 patient), abnormal glycogen deposition (1 patient) and vasculitis (2 patients).
The biochemical studies revealed the following: Mitochondrial Respiratory Chain:
Complex 1 activity <70% - 71/216 (33%)
Complex 1 activity <50% - 10/216 (5%)
Complex 2 activity <70% - 15/216 (7%)
Complex 2 activity <50% - 1/216 (0.5%)
Complex 3 activity <70% - 48/216 (22%)
Complex 3 activity <50% - 13/216 (6%)
Complex 4 activity <70% - 16/216 (7%)
Complex 4 activity <50% - 5/216 (2%)
CoQ10 activity < 100% - 16/72 (22%)
In addition, carnitine palmitoyl transferase activity <65% in 70/216 patients (32%), LDH activity was <20% in 58/216 patients (27%), phosphoglycerate mutase activity was < 50% in 6/216 patients (3%) and myoadenylate deaminase activity was <10% in 7/216 patients (3%).
Ultimately 83 patients had genetic studies. The notable findings were as follows:14 patients had glycogen storage diseases (4-alpha glucosidase deficiency [Pompe disease=glycogen storage disease type II], 3-myophosphorylase deficiency [McArdle’s disease = glycogen storage disease type V], 5-phosphorylase kinase B deficiency [glycogen storage disease type IX], 1 phosphofructokinase deficiency [Tarui disease = glycogen storage disease type VII], 1-beta enolase deficiency [glycogen storage disease type XIII])
25 patients had mutations in various mitochondrial respiratory chain enzymes. 8 patients had mitochondrial depletion mutations (6-DNA polymerase gamma [POLG]; 1-mitochondrial genome maintenance exonuclease 1 [MGME 1]); 1-p53 inducible ribonucleotide reductase [RRM2B]. 12 patients had mutations in other mitochondrial genes (1-uncoupling protein 3 [UCP3], 3-optic atrophy 1 [OPA 1], 2 -very long chain acyl-CoA dehydrogenase [VLCAD], 2-mitofusin 2 [MFN 2], 4-various mitochondrial tRNA). 11 had mutations in various genes associated with congenital myopathies (1-lamin A/C [LMNA], 1- Rapsyn [RAPSN], 1-calsequestrin [CASQ1], 1-Titin [TTN], 2-Kelch repeat and BTB Domain Containing 13 [KBT BD 13; Nemaline myopathy 6], 1-Collagen type VI alpha 2 chain [COL6A2], 3-Ryanodine receptor 1 [RYR 1], 1-regulator factor X, 3 [RFX 3])
5 had mutations in myoadenylate deaminase. 8 had no relevant metabolic gene mutations identified, but 6 had autoinflammatory syndromes ( 1-Tumor necrosis factor receptor -associated periodic syndrome [TRAPS], 1-Nacht, LRR and PYD domains-containing protein 12 [NLRP12] associated cryopyrin associated syndrome, 2-Familial Mediterranean fever, 2-Melvalonate kinase deficiency).
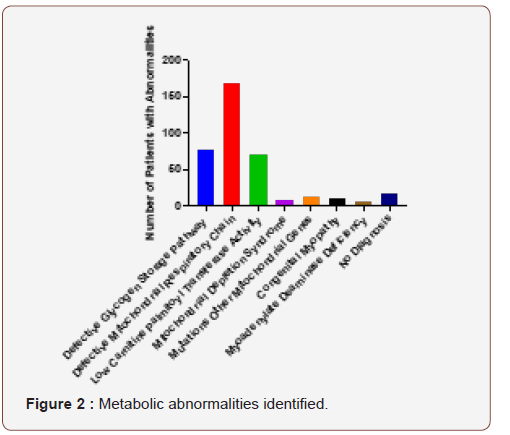
The actual work up done on each individual patient depended upon the findings, how far the patient was interested in pursuing their work up and what their insurance company was willing to pay for. Of the patients who had normal blood studies and went on to have the biochemical studies done, 36 had significant findings on the biochemical studies. There were 29 patients who had abnormal blood studies and went on to have biochemical studies that were normal. Thirteen of these patients had genetic studies that found mutations consistent with their phenotype. There were 11 patients who had normal blood studies, did not want to have a muscle biopsy and had significant findings on their genetic work up. There were 6 patients who had abnormal blood studies but got no further elucidation of their disorder from a combination of biochemical and genetic studies. Overall, most patients arrived at a treatable diagnosis with the combination of the above studies. Figure 1 demonstrates the studies that ultimately led to a treatable diagnosis in this patient population-20% from laboratory studies, 21% from genetic studies and 58% from biochemical studies. Figure 2 demonstrates the diagnoses that were reached in these patients that allowed development of a treatment strategy-78 had abnormalities of an enzyme in the glycogen storage pathway, 168 had abnormalities of a mitochondrial respiratory chain function, 8 had mitochondrial depletion syndromes, 12 had defects in other mitochondrial proteins, 11 had mutations in proteins associated with congenital myopathies, 7 had abnormal myoadenylate deaminase and 70 had low carnitine palmitoyl transferase activity. Of the 372 patients in this manuscript 16 had blood and biochemical studies that did not reveal a treatable diagnosis (and never had genetic studies done) and 2 had blood, biochemical and genetic studies that did not reveal a treatable diagnosis. Therefore only 18 of 372 patients (5%) failed to get a diagnosis by the end of the studies. The majority of these patients showed improvement with treatment.
Treatment
The treatment of mitochondrial dysfunction or mitochondrial respiratory chain disorders involved a combination of medications shown to enhance respiratory chain function and ATP production. CoQ10, which transports electrons between the complex I and complex III of the respiratory chain, folic acid, which is a cofactor for several respiratory chain enzymes, carnitine, which helps import fatty acids into the mitochondria, alpha lipoic acid, which is a strong anti-oxidant and creatine, which generates ATP through the creatine phosphate shuttle [26,36-42]. Patients with low carnitine palmitoyl transferase activity were also put on diets minimizing long chain fatty acids [43,44].
Patients with glycogen storage diseases were placed on diets restricting complex carbohydrates with larger amounts of simple sugars [45,46]. In patients with low activity of lactate dehydrogenase, patients were also given the medications to treat mitochondrial dysfunction, because of the influence of lactic acid on mitochondrial functions [47,48].
Patients with myoadenylate deaminase deficiency were treated with ribose [49]. In addition, they were given the medication for mitochondrial dysfunction because loss of ammonia could lead to depletion of fumarate which is necessary for the normal functioning of the citric acid cycle [32,50], although this affect has been questioned in one study [51].
Doses of these medications were adjusted as needed for individual patients. At the same time, it was necessary to optimize management of other clinical issues that contributed to the patients’ symptoms, such as sleep apnea, food hypersensitivities and Raynaud’s.
Additional Clinical Findings
All of the patients were referred with fatigue and exercise intolerance. Eighty-one percent of the patients carried a diagnosis of fibromyalgia given them by an Internist or Rheumatologist. Eleven percent of the patients were labeled as having difficult to treat depression. Eighteen of the patients (5%) had significant dyspnea, the cause for which could not be identified and was felt to be secondary to respiratory muscle dysfunction. Seventeen percent had been diagnosed with asthma, which is roughly double the incidence in the adult population [52]. Two patients had recurrent bouts of rhabdomyolysis. Twenty-five patients (7%) were ultimately found to have some form of inflammatory myositis. Six of these patients had normal CPK at their initial evaluation and would not have normally been evaluated for an inflammatory process. Interestingly, accelerated osteoarthritis involving the spine, hips and knees was found in 15% of the patients, presumably due to poor muscle function supporting the joints [22,53]. Congestive heart failure was present in 7 patients (2%), possibly related to the effect of a metabolic disorder on heart muscle function.
Interestingly, a majority of the patients (73%) had some form of gastric dysmotility, which included gastroesophageal reflux, gastroparesis, nausea, vomiting, constipation, diarrhea and bouts of pseudo-obstruction. Many patients (45%) had various food hypersensitivities including celiac sprue, so that elimination diets were a critical part of their management. Only 13 patients (3%) had a definitive diagnosis of inflammatory bowel disease. Another very common problem in these patients was recurrent infections (44%). Many of them had been worked up for B and T cell disorders. Eight patients (2%) had hypogammaglobulinemia, which in two cases was related to protein losing enteropathy from food hypersensitivities.
Other very common manifestations were Raynaud’s (37%), migraine headaches (54%) and anti-phospholipid antibodies (15%). Many of the patients with migraine headaches responded to centrally acting vasodilators, so they probably had reversible central nervous system vasospasm, or Raynaud’s of the brain [54-56]. Surprisingly, many of the clinical features described with particular metabolic disorders were not common in this cohort: diabetes (8%), hearing loss (5%), visual loss (3%) and peripheral neuropathy (6%). Many patients did have heat intolerance and had had reactions to propofol, statins or metformin, drugs known to be metabolic poisons [8,10,11].
Other notable clinical findings in these patients included various autoimmune diseases: Sjogren’s syndrome (14%), inflammatory arthritis (10%), scleroderma (2%) and SLE (2%). All of these disorders are present at a much lower frequency in the general population [57-61].
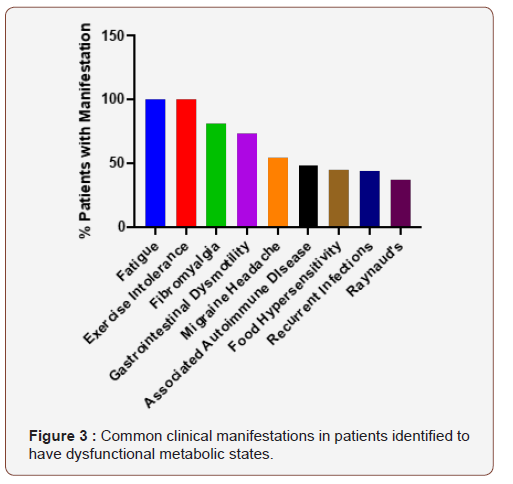
Figure 3 demonstrates the overall clinical manifestations that were most common in this cohort of patients. It provides an important listing of problems that should alert a physician that a metabolic work up is indicated. Furthermore, it provides an important list of problems that go along with metabolic disorders, each of which has to be properly addressed if patients are going to improve symptomatically with their therapy.
Discussion
This manuscript describes a cohort of patients that was referred to a University Immunology clinic because of idiopathic life altering fatigue and exercise intolerance. Additional clinical symptoms, such as gastrointestinal dysmotility, recurrent infections and accelerated osteoarthritis, suggested that there was an underlying systemic problem responsible for the clinical picture. Using a combination of laboratory studies, biochemical studies from muscle biopsies and genetic studies 95% of patients were identified to have a form of metabolic dysfunction that could be improved with available therapies. Treatment was life altering for many of these patients, although improvements in treatments for these disorders is clearly needed.
The recognition of metabolic disorders in adults is relatively recent [45,62-65]. A burgeoning literature identifies metabolic dysfunction as part of the normal aging process [66-68]. At the same time, however, many patients with inherited metabolic disorders found ways to compensate for them so that they are not noticeably symptomatic unless several factors conspired together. For example, patients with inherited metabolic disorders avoided activities that require a lot of energy expenditure, such as competitive sports during school. Patients with glycogen storage diseases had already figured out that they need to avoid complex carbohydrates. Patients with mitochondrial disorders avoided hot climates. Many patients had learned to take naps during the day. When these patients develop infections or other stressors, however, their difficulty in dealing with them made it obvious that something additional was going on.
Even more recent is the appreciation that many metabolic disorders in adults are acquired and may complicate the management of other disease processes. For example, patients with hypoxia related to lung disease or congestive heart failure will develop secondary mitochondrial dysfunction because of the requirement for oxygen in oxidative phosphorylation [69- 70]. Patients with sepsis develop multiple secondary metabolic disorders that can affect cardiac, muscle and immunological function [4,71-73]. Patients with chronic inflammation will damage local mitochondria because of local cytokine and toxin production [74].
At the same time, an underlying metabolic disorder predisposes an individual to have infections because of altered immune function [75,76]. Recurrent infections can cause tissue injury that contributes to bowel dysmotility, food hypersensitivity, autoimmunity and other forms of organ dysfunction [77-79]. Infections can alter the intestinal microbiome leading to further metabolic and immune regulatory problems [80]. Underlying metabolic disorders can alter vascular function, by reducing production of mediators such as nitric oxide, leading to organ dysfunction from altered perfusion [81-84]. Mitochondrial dysfunction has been implicated in the vasculopathy associated with diabetes [81,85,86]. Mitochondrial dysfunction has been observed in migraine headache, whether or not it is primarily vascular in nature [87].
What is emerging from these and similar studies is that metabolic dysfunction is part of many disease processes and needs to be addressed as one of the factors contributing to the pathophysiology of the disorder. For example, studies have demonstrated mitochondrial dysfunction in patients with SLE and improvement in the clinical status of the patients with antioxidants [88,89]. Particular patients with neurodegenerative diseases, such as Parkinson’s disease and multiple sclerosis, have been shown to benefit from treatment of mitochondrial dysfunction [90- 92]. Particular patients with depression see resolution of their symptoms with treatment of an underlying metabolic disorder, if it is present [93,94]. This was observed in many of the patients described in these studies.
Overall, the manuscript seeks to
1) make people aware of the existence of primary and secondary metabolic disorders in adults.
2) identify clinical symptoms and factors that make it more likely that a metabolic disorder will be identified in particular patients.
3) identify strategies for the diagnosis of these disorders. As research progresses to improve treatment of these disorders, the ability to recognize their existence will become more and more important.
Figure Legends
Figure 1
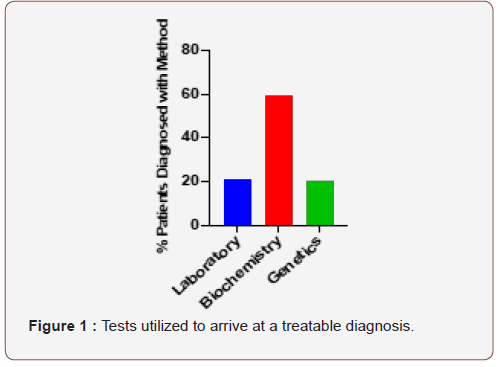
This figure demonstrates the percentage of patients who reached a treatable metabolic diagnosis based on either laboratory studies (20%), biochemical studies (59%) or genetic studies (21%) (Figure 1).
Figure 2
This figure demonstrates the number of patients who were diagnosed with the following disorders: defective glycogen storage pathway [78], defective mitochondrial respiratory chain [68], low carnitine palmitoyl transferase activit[70], mitochondrial depletion syndromes [8], mutations in other mitochondrial genes [12], congenital myopathies [11], myoadenylate deaminase deficiency [7] or no abnormality [18] (Figure 2).
Figure 3
This figure demonstrates percentage of patients expressing the common clinical manifestations of the patients evaluated in this cohort: Fatigue (100%), Exercise Intolerance (100%), Fibromyalgia (81%), Gastrointestinal Motility (73%), Migraine Headaches (54%), Associated Autoimmune Disease (48%; includes Sjogren’s syndrome-14%, Scleroderma-2%, SLE-2 %, Inflammatory arthritis-10%, Inflammatory bowel disease-3% and Antiphospholipid antibody syndrome-15%), Food hypersensitivities (45%), Recurrent infections (44%) and Raynaud’s (37%) (Figure 3).
Acknowledgement
None.
Conflicts of Interest
No conflict of interest.
References
- Perl A (2017) Metabolic Control of Immune System Activation in Rheumatic Diseases. Arthritis Rheumatol 69(12): 2259-2270.
- Zarazuela Zolkipli Cunningham, Marni J Falk (2017) Clinical effects of chemical exposures on mitochondrial function. Toxicology 391: 90-99.
- Glenn R Bantug, Lorenzo Galluzzi, Guido Kroemer, Christoph Hess (2018) The spectrum of T cell metabolism in health and disease. Nat Rev Immunol 18(1): 19-34.
- Hernando Gómez, John A Kellum, Claudio Ronco (2017) Metabolic reprogramming and tolerance during sepsis-induced AKI. Nat Reviews Nephrology 13(3): 143-151.
- Prakash YS, Pabelick CM, Sieck GC (2017) Mitochondrial Dysfunction in Airway Disease. Chest 152(23): 618-626.
- Stanzani G, Duchen MR, Singer M (2019) The role of mitochondria in sepsis-induced cardiomyopathy. Biochim Biophys Acta Mol Basis Dis 1865(4): 759-773.
- Apostolopoulou M, Corsini A, Roden M (2015) The role of mitochondria in statin-induced myopathy. Eur J Clin Invest 45(7): 745-754.
- Steven K Baker, Georgirene D Vladutiu, Wendy L Peltier, Paul J Isackson, Mark A Tarnopolsky (2008) Metabolic myopathies discovered during investigations of statin myopathy. Can J Neurol Sci 35(1): 94-97.
- Kuncl RW (2009) Agents and mechanisms of toxic myopathy. Curr Opin Neurol 22(5): 506-515.
- Anila K Madiraju, Derek M Erion, Yasmeen Rahimi, Xian Man Zhang, Demetrios T Braddock, et al. (2014) Metformin suppresses gluconeogenesis by inhibiting mitochondrial glycerophosphate dehydrogenase. Nature 510(7506): 542-546.
- Josef Finsterer, Marlies Frank (2016) Propofol Is Mitochondrion-Toxic and May Unmask a Mitochondrial Disorder. J Child Neurol 31(13): 1489-1494.
- Di Mauro S, Bonilla E, Zeviani M, Nakagawa M, De Vivo DC (1985) Mitochondrial myopathies. Ann Neurol 17(6): 521-538.
- Berardo A, Di Mauro S, Hirano M (2010) A diagnostic algorithm for metabolic myopathies. Curr Neurol Neurosci Rep 10(2): 118-126.
- Stefano Di Donato (2009) Multisystem manifestations of mitochondrial disorders. Journal of neurology 256(5): 693-710.
- Hooper RG, Thomas AR, Kearl RA (1995) Mitochondrial enzyme deficiency causing exercise limitation in normal-appearing adults. Chest 107(2): 317-322.
- Nicholas R Wawrzyniak, Anna-Maria Joseph, David G Levin, David M Gundermann, Christiaan Leeuwenburgh, et al. (2016) Idiopathic chronic fatigue in older adults is linked to impaired mitochondrial content and biogenesis signaling in skeletal muscle. Oncotarget 7(33): 52695-52709.
- Voermans NC, Snoeck M, Jungbluth H (2016) RYR1-related rhabdomyolysis: A common but probably underdiagnosed manifestation of skeletal muscle ryanodine receptor dysfunction. Rev Neurol (Paris) 172(10): 546-558.
- Damien Arnoult, Leticia Carneiro, Ivan Tattoli, Stephen E Girardin (2009) The role of mitochondria in cellular defense against microbial infection. Semin Immunol 21(4): 223-232.
- Geoffrey Burnstock, Kenneth A Jacobson, Fievos L Christofi (2017) Purinergic drug targets for gastrointestinal disorders. Curr Opin Pharmacol 37: 131-141.
- Dean Nathanial Defrees, Justin Bailey (2017) Irritable Bowel Syndrome: Epidemiology, Pathophysiology, Diagnosis, and Treatment. Prim Care 44(4): 655-671.
- Beverley Greenwood Van Meerveld, Anthony C Johnson, David Grundy (2017) Gastrointestinal Physiology and Function. Handb Exp Pharmacol 239: 1-16.
- Francisco J Blanco, Ana M Valdes, Ignacio Rego Pérez (2018) Mitochondrial DNA variation and the pathogenesis of osteoarthritis phenotypes. Nat Rev Rheumatol 14(6): 327-340.
- Gráinne S Gorman, Joanna L Elson, Jane Newman, Brendan Payne, Robert Mc Farland (2015) Perceived fatigue is highly prevalent and debilitating in patients with mitochondrial disease. Neuromuscul Disord 25(7): 563-566.
- Katrine B Norheim, Grete Jonsson, Roald Omdal (2011) Biological mechanisms of chronic fatigue. Rheumatology 50(6): 1009-1018.
- Mario D Cordero, Manuel de Miguel, Inés Carmona López, Pablo Bonal, Francisco Campa, et al. (2010) Oxidative stress and mitochondrial dysfunction in fibromyalgia. Neuro Endocrinol Lett 31(2): 169-173.
- Mishal Abdullah, Sahana Vishwanath, Amro Elbalkhi, Julian L Ambrus (2012) Mitochondrial myopathy presenting as fibromyalgia: A case report. J Med Case Rep 6: 55.
- Mario D Cordero, Elísabet Alcocer Gómez, Fabiola Marín-Aguilar, Tatyana Rybkina, David Cotán, et al. (2016) Mutation in cytochrome B gene of mitochondrial DNA in a family with fibromyalgia is associated with NLRP3-inflammasome activation. J Med Genet 53(2): 113-122.
- Robert K Naviaux, Jane C Naviaux, Kefeng Li, A Taylor Bright, William A Alaynick, et al. (2016) Metabolic features of chronic fatigue syndrome. Proc Natl Acad Sci USA 113(37): E5472-E5480.
- Cara Tomas, Audrey E Brown, Julia L Newton, Joanna L Elson (2019) Mitochondrial complex activity in permeabilised cells of chronic fatigue syndrome patients using two cell type. PeerJ 7-e6500.
- Valerie Desquiret Dumas, Geraldine Leman, Celine Wetterwald, Stephanie Chupin, Anaïs Lebert (2019) Warburg-like effect is a hallmark of complex I assembly defects. Biochim Biophys Acta Mol Basis Dis 1865(9): 2475-2489.
- Mary Ellen Harper, Erin L Seifert (2008) Thyroid hormone effects on mitochondrial energetics. Thyroid 18(2): 145-156.
- Fishbein WN (1985) Myoadenylate deaminase deficiency: inherited and acquired forms. Biochem Med 33(2): 158-169.
- Coleman RA, Stajich JM, Pact VW, Pericak Vance MA (1986) The ischemic exercise test in normal adults and in patients with weakness and cramps. Muscle Nerve 9(3): 216-221.
- Marni J Falk, Lishuang Shen, Michael Gonzalez, Jeremy Leipzig, Marie T Lott (2015) Mitochondrial Disease Sequence Data Resource (M Seq DR): a global grass-roots consortium to facilitate deposition, curation, annotation, and integrated analysis of genomic data for the mitochondrial disease clinical and research communities. Mol Genet Metab 114(3): 388-396.
- Francisca Millan, Megan T Cho, Kyle Retterer, Kristin G Monaghan, Renkui Bai (2016) Whole exome sequencing reveals de novo pathogenic variants in KAT6A as a cause of a neurodevelopmental disorder. Am J Med Genet A 170(7): 1791-1798.
- Richard H Haas (2007) The evidence basis for coenzyme Q therapy in oxidative phosphorylation disease. Mitochondrion 7 Suppl: S136-S145.
- Garth L Nicolson (2014) Mitochondrial dysfunction and chronic disease: treatment with natural supplements. Altern Ther Health Med 20 (Suppl 1): 18-25.
- Jia Sun, Haiping Zhu, Xiaorong Wang, Qiuqi Gao, Zhuoying Li, et al. (2019) CoQ10 ameliorates mitochondrial dysfunction in diabetic nephropathy through mitophagy. J Endocrinol 240: 445-465.
- Elisa I Glover, Joan Martin, Amy Maher, Rebecca E Thornhill, Gerald R Moran, et al. (2010) A Randomized Trial of Coenzyme Q10 in Mitochondrial Disorders. Muscle Nerve 42(5): 739-748.
- Rudolf A Kley, Mark A Tarnopolsky, Matthias Vorgerd (2011) Creatine for treating muscle disorders. Cochrane Database Syst Rev (2).
- Tarnopolsky MA (2008) The Mitochondrial Cocktail: Rationale for Combined Nutraceutical Therapy in Mitochondrial Cytopathies. Adv Drug Deliv Rev 60(13-14): 1561-1567.
- Ankita Bhattacharjee, Shilpi Kumari Prasad, Oly Banerjee, Siddhartha Singh, Arnab Banerjee (2018) Targeting mitochondria with folic acid and vitamin B-12 ameliorates nicotine mediated islet cell dysfunction. Environ Toxicol 33(9): 988-1000.
- Corrado Angelini, Claudio Semplicini (2010) Metabolic myopathies: the challenge of new treatments. Curr Opin Pharmacol 10(3): 338-345.
- Bax, Isackson, Moore M, Ambrus J (2020) Carnitine Palmitoyl Transferase Deficiency in a University Immunology Practice. Current Rheumatology Reports.
- Weinstein DA, Wolfsdorf JI (2002) Glycogen storage diseases: A primer for clinicians. Endocrinologist 12(6): 531-538.
- Quinlivan RM, Beynon RJ (2007) Pharmacological and nutritional treatment trials in McArdle disease. Acta Myol 26(1): 58-60.
- Sun Mi Hong, Young Kyoung Lee, Imkyong Park, So Mee Kwon, Seongki Min, et al. (2019) Lactic acidosis caused by repressed lactate dehydrogenase subunit B expression down-regulates mitochondrial oxidative phosphorylation via the pyruvate dehydrogenase (PDH)-PDH kinase axis. J Biol Chem 294(19): 7810-7820.
- Daniel A Kane (2014) Lactate oxidation at the mitochondria: a lactate-malate-aspartate shuttle at work. Front Neurosci 8: 366.
- Goebel HH, Bardosi A (1987) Myoadenylate deaminase deficiency. Klin Wochenschr 65: 1023-1033.
- Teijeira S, Millan BS, Fernandez JM, Rivas E, Vieitez I, et al. (2009) Myoadenylate deaminase deficiency: clinico-pathological and molecular study of a series of 27 Spanish cases. Clin Neuropathol 28(2): 136-142.
- Tarnopolsky MA, Parise G, Gibala MJ, Graham TE, Rush JW (2001) Myoadenylate deaminase deficiency does not affect muscle anaplerosis during exhaustive exercise in humans. J Physiol 533(Pt 3): 881-889.
- Lisa A Reynold, Brett Finlay B (2017) Early life factors that affect allergy development. Nat Rev Immunol 17(8): 518-528.
- Francisco J Blanco, Ignacio Rego, Cristina Ruiz Romero (2011) The role of mitochondria in osteoarthritis. Nat Rev Rheumatol 7(3): 161-169.
- Leonard H Calabrese, David W Dodick, Todd J Schwedt, Aneesh B Singhal (2007) Narrative Review: Reversible Cerebral Vasoconstriction Syndromes. Annals of internal medicine 146(1): 34-44.
- Sarthak Gupta, Robert Zivadinov, Deepa Ramasamy, Julian L Ambrus (2014) Reversible cerebral vasoconstriction syndrome (RCVS) in antiphospholipid antibody syndrome (APLA): the role of centrally acting vasodilators. Case series and review of literature. Clin rheumatol 33(12): 1829-1833.
- Ganne S Umamaheswara Rao, Radhakrishnan Muthuchellappan (2016) Cerebral vasospasm: current understanding. Curr Opin Anesthesiol 29(5): 544-551.
- Baodong Qin, Jiaqi Wang, Zaixing Yang, Min Yang, Ning Ma (2015) Epidemiology of primary Sjogren's syndrome: a systematic review and meta-analysis. Ann Rheum Dis 74(11): 1983-1989.
- Frederick B Vivino, Vatinee Y Bunya, Giacomina Massaro Giordano, Chadwick R Johr, Stephanie L Giattino, et al. (2019) Sjogren's syndrome: An update on disease pathogenesis, clinical manifestations and treatment. Clin Immunol 203: 81-121.
- Jammie Barnes, Maureen D Mayes (2012) Epidemiology of systemic sclerosis: incidence, prevalence, survival, risk factors, malignancy, and environmental triggers. Curr Opin Rheumatol 24(2): 165-170.
- Yannis Alamanos, Paraskevi V Voulgari, Christos Siozos, Pelagia Katsimpri, Stylianos Tsintzos (2003). Epidemiology of systemic lupus erythematosus in northwest Greece 1982-2001. J Rheumatol 30(4): 731-735.
- Maria D Alonso, Javier Llorca, Francisco Martinez Vazquez, Jose A Miranda Filloy, Teresa Diaz De Teran, et al. (2011) Systemic Lupus Erythematosus in Northwestern Spain A 20-Year Epidemiologic Study. Medicine 90(5): 350-358.
- Patrick F Chinnery (2015) Mitochondrial disease in adults: what's old and what's new? EMBO Mol Med 7(12): 1503-1512.
- Robert D S Pitceathly, Robert McFarland (2014) Mitochondrial myopathies in adults and children: management and therapy development. Curr Opin in Neurol 27(5): 576-582.
- Cenacchi G, Papa V, Costa R, Pegoraro V, Marozzo R, et al. (2019) Update on polyglucosan storage diseases. Virchows Archiv 475: 671-686.
- Margaret Adler, Perry B Shieh (2015) Metabolic Myopathies. Semin Neurol 35(4): 385-397.
- Robert S Balaban, Shino Nemoto, Toren Finkel (2005) Mitochondria, oxidants, and aging. Cell 120(4): 483-495.
- Leonard Guarente (2008) Mitochondria - A nexus for aging, calorie restriction, and sirtuins? Cell 132(2): 171-176.
- Ji Yong Jang, Arnon Blum, Jie Liu, Toren Finkel (2018) The role of mitochondria in aging. J Clin Invest 128(9): 3662-3670.
- David Bargiela, Stephen P Burr, Patrick F Chinnery (2018) Mitochondria and Hypoxia: Metabolic Crosstalk in Cell-Fate Decisions. Trends in Endocrinology and Metabolism 29(4): 249-259.
- Mc Elroy GS, Chandel NS (2017) Mitochondria control acute and chronic responses to hypoxia. Exp Cell Res 356(2): 217-222.
- María Cecilia Cimolai, Silvia Alvarez, Christoph Bode, Heiko Bugger (2015) Mitochondrial Mechanisms in Septic Cardiomyopathy. Int J Mol Sci 16(8): 17763-17778.
- Anna J Dare, Anthony RJ Phillips, Anthony JR Hickey, Anubhav Mittal, Benjamin Loveday (2009) A systematic review of experimental treatments for mitochondrial dysfunction in sepsis and multiple organ dysfunction syndrome. Free Radic Biol Med 47(11): 1517-1525.
- Clifford S Deutschman, Kevin J Tracey (2014) Sepsis: Current Dogma and New Perspectives. Immunity 40(4): 464-476.
- Jillian P Rhoads, Amy S Major, Jeffrey C Rathmell (2017) Fine tuning of immunometabolism for the treatment of rheumatic diseases. Nature Reviews Rheumatology 13(5): 313-320.
- Melissa A Walker, Stefano Volpi, Katherine B Sims, Jolan E Walter, Elisabetta Traggiai (2014) Powering the Immune System: Mitochondria in Immune Function and Deficiency. J Immunol Res 2014.
- Sarah Dimeloe, Anne Valérie Burgener, Jasmin Grählert, Christoph Hess (2017) T-cell metabolism governing activation, proliferation and differentiation; a modular view. Immunology 150(1): 35-44.
- Elizabeth A Mann, Shehzad A Saeed (2012) Gastrointestinal infection as a trigger for inflammatory bowel disease. Curr Opin Gastroenterol 28(1): 24-29.
- Lulu Sun, Gerardo M Nava, Thaddeus S Stappenbeck (2011) Host genetic susceptibility, dysbiosis, and viral triggers in inflammatory bowel disease. Curr Opin in Gastroenterol 27(4): 321-327.
- Bach JF (2005) Infections and autoimmune diseases. J Autoimmun 25: 74-80.
- Maslowski KM (2019) Metabolism at the centre of the host-microbe relationship. Clinical and Experimental Immunology 197: 193-204.
- Yu E, J Mercer, Bennett M (2012) Mitochondria in vascular disease. Cardiovasc Res 95: 173-182.
- Koga Y, N Povalko, J Nishioka, K Katayama, N Kakimoto, et al. (2010) MELAS and L-arginine therapy: pathophysiology of stroke-like episodes. Ann N Y Acad Sci 1201: 104-110.
- Paulin R, ED Michelakis (2014) The Metabolic Theory of Pulmonary Arterial Hypertension. Circ Res 115: 148-164.
- Busija, DWI Rutkai, S Dutta, PV Katakam (2016) Role of Mitochondria in Cerebral Vascular Function: Energy Production, Cellular Protection, and Regulation of Vascular Tone. Compr Physiol 6: 1529-1548.
- Iizuka T, Y Goto, S Miyakawa, M Sato, Z Wang, et al. (2009) Progressive carotid artery stenosis with a novel tRNA phenylalanine mitochondrial DNA mutation. J Neurol Sci 278: 35-40.
- Wenceslau CF, CG McCarthy, T Szasz, K Spitler, S Goulopoulou, et al. (2014) Mitochondrial damage-associated molecular patterns and vascular function. Eur Heart J 35: 1172-1177.
- Gross EC, M Lisicki, D Fischer, PS Sandor, J Schoenen (2019) The metabolic face of migraine - from pathophysiology to treatment. Nat Rev Neurol 15: 627-643.
- Oaks Z, A Perl (2014) Metabolic control of the epigenome in systemic Lupus erythematosus. Autoimmunity 47: 256-264.
- Lai ZW, R Hanczko, E Bonilla, TN Caza, B Clair, et al. (2012) N-acetylcysteine reduces disease activity by blocking mammalian target of rapamycin in T cells from systemic lupus erythematosus patients: a randomized, double-blind, placebo-controlled trial. Arthritis and rheum 64: 2937-2946.
- Area Gomez E, C Guardia Laguarta, EA Schon, S Przedborski (2019) Mitochondria, OxPhos, and neurodegeneration: cells are not just running out of gas. J Clin Invest 129: 34-45.
- Barcelos IP, RM Troxell, JS Graves (2019) Mitochondrial Dysfunction and Multiple Sclerosis. Biology (Basel) 8: E37.
- Fernandez Moriano C, E Gonzalez Burgos, MP Gomez Serranillos (2015) Mitochondria-Targeted Protective Compounds in Parkinson's and Alzheimer's Diseases. Oxid Med Cell Longev.
- Allen J, R Romay Tallon, KJ Brymer, HJ Caruncho, LE Kalynchuk (2018) Mitochondria and Mood: Mitochondrial Dysfunction as a Key Player in the Manifestation of Depression. Front Neurosci 12: 1-386.
- Caruso, GC Benatti, JMC Blom, F Caraci, F Tascedda (2019) The Many Faces of Mitochondrial Dysfunction in Depression: From Pathology to Treatment. Front Pharmacol 10: 1-995.
-
Julian L A, Paul I, Molly M, John B, Lucia B. Investigating Fatigue and Exercise Intolerance in a University Immunology Clinic. Arch Rheum & Arthritis Res. 1(1): 2020. ARAR.MS.ID.000505.
-
Carnitine palmitoyl transferase, Mitochondria, Glycogen Storage Disease, Myoadenylate Deaminase, Asthma, Sjogren’s syndrome, Raynaud’s, Gastrointestinal dysmotility, Curcumin, Arthritis, Treatment, Medications, Anti-inflammatory
-

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.