Mini Review
Mini Review
Cranial Nerve Manifestations in Sarcoidosis: A National Retrospective Analysis
Mary Salim*, Sankeerth Kondapalli, Emily Kokus, Evelyne Kal Youssef, Eugenio Capitle and Reena Khianey
University of Rutgers, New Jersey Medical School, New Jersey
Mary Salim, University of Rutgers, New Jersey Medical School, New Jersey
Received Date:February 27, 2024; Published Date:March 14, 2024
Introduction
Sarcoidosis is a systemic granulomatous inflammatory disease that can involve multiple organ systems, including the lungs, skin, skeletal, and nervous system. Cranial neuropathies (CN) in patients with sarcoidosis is a well-known clinical entity and neuropathies of the optic, facial, and oculovestibular nerves can be an isolated manifestations in 10-38% of sarcoidosis patients [1].
The effects of sarcoidosis on the nervous system are less apparent, partly due to its prevalence of less than 10% of all sarcoidosis patients, but also because it often appears very early in the disease course, even before any other systemic symptoms present [2,3]. Complicating the diagnosis is the presence of other symptoms that can be attributed to a variety of other medical conditions. Epileptic seizures, meningitis, mass lesions, and cranial neuropathies can all present as part of the presentation of neurosarcoidosis in individuals, with varying degrees of likelihood [3].
Given that University Hospital, Rutgers in Newark, New Jersey is a sarcoidosis specialized center, we were interested in performing a statistical, retrospective analysis of sarcoidosis patients with and without CN. We aimed to identify comorbidities in this subset of sarcoidosis patients to raise a higher clinical suspicion for this diagnosis in a higher risk population; thereby, avoiding a delay in appropriate management. We used the National Inpatient Sample (NIS) from 2003 to 2014 was queried to identify patients diagnosed with sarcoidosis (ICD9: 135). Patients with sarcoidosis were categorized into two groups based on whether they had comorbid CN palsies and disorders (CN I-XII palsy, multiple CN palsies, optic neuritis, trigeminal neuralgia, and sensorineural hearing loss). Univariate and multivariate analyses were conducted to compare patient and hospital characteristics as well as Elixhauser comorbidities between the two groups.
We found that approximately 0.8% (n=1,314) of patients with sarcoidosis (n=173,236) had cranial neuropathies. Patients with CN were significantly younger (52.62 years ± 15.19 vs 56.15 ± 13.85, p<0.001) and female (70.1% vs 65.9%, p=0.002) compared to their non-CN counterparts. Although length of hospital stay (5.91days ± 6.56 vs 5.50 ± 6.82, p=0.030) was significantly longer for sarcoidosis with CN, total hospital charges ($41,511 ± $55,206 vs $38,150 ± $62,345, p=0.053) did not significantly differ between the groups. Furthermore, patients were at significantly lower odds to develop cranial neuropathies if they had comorbid chronic pulmonary disease (OR [95% CI]: 0.634 [0.544-0.738], p<0.001), congestive heart failure (0.528 [0.401-0.697], p<0.001), pulmonary circulation disorders (0.609 [0.423-0.876], p=0.008), or renal failure (0.649 [0.514-0.818], p<0.001). However, patients with lymphoma (2.271 [1.518-3.398], p<0.001), other neurologic disorders (1.425 [1.155- 1.758], p=0.001), and non-facial paralysis (2.682 [2.037-3.531], p<0.001) had increased likelihood of developing CN. The presence of comorbid coagulopathy, iron deficiency anemia, diabetes, liver disease, or fluid/electrolyte disorders did not significantly impact the likelihood of developing CN (table 1).
Table 1:
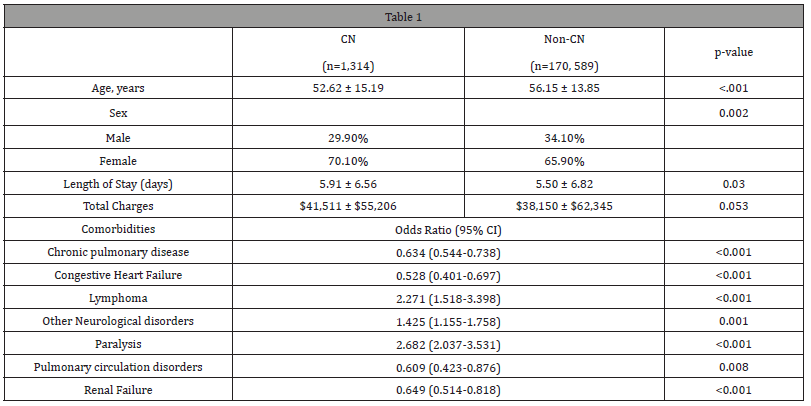
Our analysis demonstrates greater odds of CN in sarcoidosis patients with comorbid lymphoma, other neurologic disorders, or non-facial paralysis. However, sarcoidosis patients with cardiopulmonary comorbidities or renal failure were less likely to have CN, suggesting that sarcoidosis patients with CN may somewhat spare other organs. Sarcoidosis patients who are relatively young, or have other neurological disorders, non-facial paralysis, or lymphomas, require a higher index of suspicion for CN. Goel et al. identified the importance of identifying manifestations for prognosticating patients, as they noted poor prognosis in patients with bilateral optic neuritis, myelopathy, and imaging evidence of hydrocephalus or leptomeninges’s [4]. Prognosis is an important facet of patient care, so obtaining improved prognosis measures would allow physicians to better care for their patients.
Identifying manifestations early in the course of disease is not only important for prognosticating patients, but also for treating them. Arun and Palace noted that early immunosuppressive treatment significantly improved clinical outcomes for patients with neurosarcoidosis. In particular, they found multi-drug therapy with azathioprine, methotrexate, and infliximab to be more effective than treatment solely with steroids [5]. This example demonstrates the importance of identifying risk of developing neurosarcoidosis to not only improve patient care but also improve patient outcomes.
Acknowledgements
None.
Conflict of interest
No conflict of Interest.
References
- Barreras, Paula, Barney J Stern (2022) Clinical features and diagnosis of neurosarcoidosis-review article. Journal of neuroimmunology 368: 577871.
- Joseph FG, Scolding NJ (2007) Sarcoidosis of the nervous system. Practical Neurology 7: 234-244.
- Joseph FG, Scolding NJ (2009) Neurosarcoidosis: a study of 30 new cases. Journal of Neurology, Neurosurgery & Psychiatry 80(3): 297-304.
- Goel J, Anadure R, Gupta S, Vinny Wilson, Rajeev Saxena, Samaresh Sahu, et al. (2020) A Study of the Clinical Profile, Radiologic Features, and Therapeutic Outcomes in Neurosarcoidosis from Two Tertiary Care Centers in Southern India. Neurology India 68(3): 609-616.
- Arun T, Palace J (2021) Effects of immunotherapies and clinical outcomes in neurosarcoidosis: a retrospective cohort study. Journal of Neurology 268(7): 2466-2472.
-
Mary Salim*, Sankeerth Kondapalli, Emily Kokus, Evelyne Kal Youssef, Eugenio Capitle and Reena Khianey. Cranial Nerve Manifestations in Sarcoidosis: A National Retrospective Analysis. Arch Rheum & Arthritis Res. 3(1): 2024. ARAR.MS.ID.000555.
-
Postural orthostatic tachycardia syndrome, Neuropathy, Autoimmune diseases, Inflammation, Rheumatoid arthritis, chronic fatigue syndrome, Immunizations.
-

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.