 Review Article
Review Article
Use of Antibody/Antigen Diagnostic Testing and Face Coverings with Traditional Non-Pharmaceutical Intervention Measures to Mitigate Human-to- Human Coronavirus (COVID-19) Transmission at the Community Level
Cameron YS Lee1*, DMD, MD, PHD, MPH, MSEd and Kristin L Lee2, DDS
1Private Practice, Oral, Maxillofacial and Reconstructive Surgery, Aiea, HI. 96701, Professor of Surgery, Temple University, Kornberg School of Dentistry, Philadelphia, PA. 19140, USA
2Oral and Maxillofacial Surgery Resident, University of California at San Francisco/Highland General Hospital, Oakland, CA. 94602, USA
Cameron YS Lee, DMD, MD, PHD, MPH, MSEd, Private Practice. Oral, Maxillofacial and Reconstructive Surgery, Aiea, HI. 96701, Professor of Surgery, Temple University. Kornberg School of Dentistry, Philadelphia, PA. 19140, USA
Received Date:August 03, 2020; Published Date: August 28, 2020
Abstract
In December 2019, the Health Commission of Hubei province, China reported a cluster of new cases of viral pneumonia later identified as a highly infectious novel human coronavirus that causes the severe acute respiratory syndrome (SARS-CoV-2), renamed Covid-19. In January 2020, the first confirmed case of Covid-19 was reported in the United States. As of this writing, Covid-19 has spread globally, with greater than 700,000 fatalities and infecting over 21 million people around the world in just over 8 months. The United States is now the epicenter with over 5 million reported cases and greater than 170,000 deaths. The Covid-19 pandemic has emerged as an unprecedented global crisis overwhelming healthcare systems around the world. Public health authorities must rely on non-pharmaceutical intervention (NPIs) measures to mitigate the spread of coronavirus transmission as no vaccine or antiviral therapy is available. As the number of infectious disease cases continue to rise with increasing mortality, the authors recommend routine antibody/antigen diagnostic testing of not only symptomatic persons for the coronavirus, but asymptomatic and presymptomatic individuals as well. The universal policy of routine use of face coverings when out in public should also be implemented as it is now recognized that viral shedding can occur several days before the onset of clinical symptoms and last for days. In this article, we describe the public health strategies and the challenges public health authorities may encounter in this 21st century global pandemic to flatten the pandemic curve.
Introduction
Pandemic the word strikes fear because of the severity of the disease in terms of morbidity, mortality, social and economic turmoil. Within 24-36 hours, an infectious disease can arrive on the doorstep of any densely populated city from a remote part of the world due to air travel and globalization [1,2]. Undetected, the virulent pathogen can quickly overwhelm the healthcare system protecting its citizens. Several viruses have caused pandemics in the 20th century (1957 Asian flu; 1968 Hong Kong flu; 1981 AIDS/HIV), but all pandemics are measured against the Spanish influenza pandemic of 1918-1919. It is estimated to have infected over 500 million individuals and killed 50-100 million people around the globe due to sustained community spread [3,4]. It is estimated that 675,000 deaths occurred in the United States. Mortality was highest in young adults, aged 24-40 years.
A common characteristic of pandemics is that they are facilitated by sustained transmission between individuals in a community and can spread rapidly over a short time causing relatively high mortality [5,6]. Compared to the 2003 SARS pandemic, Covid-19 is far more lethal with greater infectivity and mortality as asymptomatic and presymptomatic individuals are capable of spreading the virus in the community 2-3 days before the onset of clinical symptoms [7-9]. Therefore, it is uncertain if traditional NPIs can effectively mitigate transmission of the coronavirus. As the Covid-19 pandemic trajectory continues to rise with increasing mortality, the authors recommend additional non-pharmaceutical intervention measures such as antibody/antigen diagnostic testing for the coronavirus of both asymptomatic and presymptomatic individuals and the routine use of face coverings (masks) out in public to flatten the pandemic curve. In this article, we describe the additional and traditional non-pharmaceutical intervention (NPI) measures to contain the transmission of the coronavirus in the community. We also describe the unique challenges public health authorities will encounter while implementing NPIs in the community.
2003 SARS and COVID-19
The 2003 coronavirus that is the etiology for the severe acute respiratory syndrome (SARS-CoV-1) in China killed 774 people and had 8,096 reported cases was the first pandemic of the 21st century [1,10,11]. With no viral vaccine, the coronavirus outbreak was successfully contained with traditional non-pharmaceutical public health interventions used to eradicate the 1918 influenza pandemic and other influenza pandemics of the 20th century.
In December 2019, multiple cases of a severe pneumonia with unknown origin were reported in Wuhan, the capital city of Hubei province China [12-15]. The pathogen was identified as a novel enveloped RNA beta-coronavirus that was designated severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), which is similar to SARS-CoV-1 but much more infectious. The virus rapidly spread throughout the world and became known as Covid-19. On March 11, 2020 the World Health Organization (WHO) declared Covid-19 a worldwide pandemic [16]. As of this writing, the infectious disease has spread globally, and greater than 21 million cases have been reported and over 700,000 people have died [17].
Patients infected with Covid-19 that are asymptomatic or mildly symptomatic and not seeking medical treatment are likely spreading the stealth virus throughout the community from respiratory droplets and direct surface contact [8,18,19]. This is why the Covid-19 pandemic has generated such urgency regarding the implementation of public health mitigation strategies. With no controlled scientific studies that document the effective use of NPI measures, public health has had to rely on historical analysis and observations of what proved successful in slowing down the spread and eventual eradication of a pandemic. An example is the use of geographic quarantine measures by a small town in Colorado and a naval station located on an island in San Francisco Bay during the 1918 influenza pandemic that resulted in minimal morbidity and mortality [20,21]. Use of simulation model studies based on contemporary public health data from the 2009 influenza pandemic were also studied to evaluate the effectiveness of NPIs in select cities of the United States [22]. The results of the simulation studies support the implementation of NPIs in controlling the spread of the contagion when an antiviral vaccine is not available.
Epidemiologic Characteristics of the Coronavirus Outbreak
The Covid-19 outbreak from Wuhan, China is similar to the 2003 coronavirus epidemic where zoonotic transmission from bats via the intermediate host palm civets in Guangdong, China is largely responsible for the 2003 global pandemic. As Covid-19 is more contagious than the 2003 SARS, and with a basic reproductive number of 2.2 to 2.5 it rapidly spread from Wuhan to the entire country in 30 days overwhelming China’s healthcare and public health system [14,23]. Such rapid disease transmission was due to shedding of high viral loads that can occur 2-3 days prior to the onset of clinical symptoms and human-to-human transmission in the community [8,18,19,23]. Studies have demonstrated that initial high viral loads are associated with death [24].
Mitigating the transmission of any infectious disease that may progress to a pandemic requires an understanding of the potential disease transmission of the pathogen [23,25,26]. The basic reproductive number (R0) and the case fatality rate (CFR) are two important epidemiological parameters that will allow clinicians and public health authorities to understand the viral dynamics and epidemiological characteristics of a pandemic [27,28]. One measure of viral infectivity is the basic reproductive number which is characteristic to a pathogen. It is the average number of secondary cases produced by a primary infectious case in a susceptible population without intervention [29]. Calculating the R0 can estimate the virulence of the infectious disease from an infected individual and helps to determine the selection of mitigation strategies to contain and slow down disease transmission. An R0 greater than 1 represents a disease that will be difficult to slow down and will spread rapidly in the community. A R0 of less than 1 will not progress to a pandemic and will decay. The median R0 of influenza has been reported to be between 1.5 to 2. Measles has an R0 of 15 and is very contagious. The R0 of SARS-CoV-1 was approximately 1.7 to 1.9, while it is estimated that the R0 of Covid-19 is between 2 and 4, with a median of 2.79 [23,26,30]. Guan et al (2020) estimates that a R0 between 2 and 3 correlates to a high pandemic potential where each infected individual can spread the virus to two or three other individuals [31]. Therefore, compared to SARS-CoV-1, Covid-19 with a much higher reproductive number is more virulent with greater potential for transmission of disease.
The case-fatality rate (CFR) is the number of deaths in persons who tested positive for the disease divided by the number of confirmed cases [32]. Public health interventions rely on the early CFR estimates to determine intensity, timing and duration of intervention strategies of outbreaks [33]. Patients infected with the 2003 SARS virus had a CFR between 7% to 17% (average 9.6%) in the most severely affected areas by the virus (774 deaths in 8, 096 reported cases) involving 26 countries [34]. Early studies regarding Covid-19 case fatality rate in the city of Wuhan varied from 4.3% to 14.6% [13,35]. The average fatality rate of persons reported by the Chinese CDC and Prevention was 2.3% (1,023 deaths among the 44, 672 confirmed cases in Wuhan, China). However, when CFR was stratified by age group, the fatality rate increased due to increasing age, the presence of comorbidities and initial high viral loads [36].
Differences in age structure of the affected population will reveal the true burden of disease. High viral load is considered an independent risk factor with a worse prognosis [37]. Adults in this age group had reported comorbidities such as cardiovascular disease, pulmonary disease, and diabetes mellitus. The CFR for adults older than 60 years was 3.6%, while adults over 70 years had a reported CFR of 8.0%. Adults older than 80 years had a high lethal CFR of 14.8% [31]. High viral loads observed in the older patient population is associated with a higher CFR.
Clinical characteristic of Covid-19
Estimates of the Covid-19 incubation period range between 1-14 days but may be as long as 24 days [38]. Incubation period is defined as the time from exposure of the pathogen to the development of signs and symptoms of disease. Although the estimated median incubation time is approximately 5.1 days for Covid-19, it is now recognized that asymptomatic or mildly symptomatic individuals are capable of spreading the virus in the community 2-3 days before the onset of clinical symptoms [8,18,19,23,39]. In comparison, infectiousness of the 2003 SARS outbreak occurred 7-10 days after onset of symptoms that resulted in patients seeking treatment before they could continue to spread the virus [1].
Patient clinical presentation in both SARS-CoV-1 and Covid-19 are similar to the influenza virus and includes the following: persistent fever, chills, fatigue, myalgia, malaise, dry non-productive cough, headache, dyspnea, sputum production, arthralgia, sore throat, rhinorrhea, diarrhea and nausea/vomiting. Such non-specific clinical findings that mimic influenza make the diagnosis difficult and may be responsible for the early spread of the Covid-19 virus [8,24,31,40]. However, new reported clinical findings that have raised alarming concern are the presence of neurologic manifestations. During the SARS-2003 pandemic, Tsai et al (2004) first reported on neurologic complications that consisted of axonal peripheral neuropathy or myopathy with elevation in creatine kinase levels in four patients three weeks after the first symptoms of SARS [41]. More recently, Mao et al. (2020), reported that 78 (36.4%) of 214 hospitalized patients diagnosed with Covid-19 had various neurologic manifestations that involved the central and peripheral nervous systems and skeletal muscles [42]. Older patients with comorbidities were more prone to present with neurological manifestations including ischemic stroke, seizures, ataxia and depressed levels of consciousness. Oxley et al (2020) reported five cases of large vessel stroke in patients under 50 years of age who were all diagnosed with Covid-19 [43].
Reports of loss of smell and taste have also been reported that occurs early during the course of Covid-19 [42,44,45]. However, anosmia with SARS-2003 was first reported by Hwang (2006) [46]. In this single case report, the loss of smell occurred about 3 weeks after the onset of clinical symptoms. With reports of new onset neurologic symptoms, Covid-19 should be included in the differential diagnosis when patients present with such neurological manifestations.
Non-Pharmaceutical Interventions (NPIs)
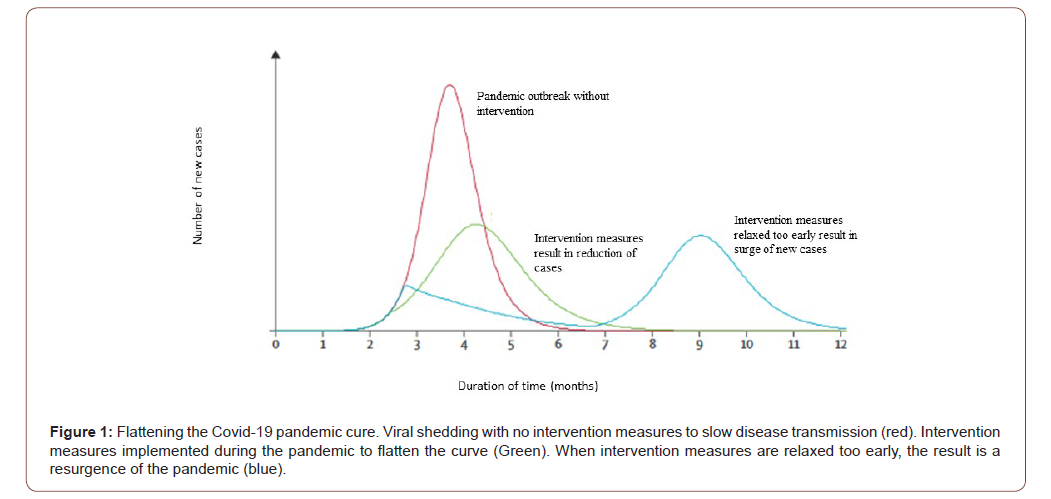
The objective of community mitigation in the face of a pandemic are to slow down the peak of the pandemic, decrease the total number of cases to decrease morbidity and mortality, control the surge capacity of the healthcare infrastructure and buy time to develop an effective vaccine or new antiviral therapies (Figure 1) [47,48]. In addition to implementing simultaneous largescale community NPIs to control viral transmission, promoting personal and environmental protective measures should not be underestimated. Household-based public health interventions include hand washing with soap and water (or alcohol-based hand sanitizers), respiratory etiquette (covering your mouth and nose when coughing or sneezing), the use of face coverings, and use of protective eyewear [18,40]. Aerosol and fomite transmission of the coronavirus is possible as the virus can remain viable and infectious in aerosols for hours and on different surface tops for days [18,24,49] Environmental cleaning of surface tops and objects is recommended as it proved effective in controlling the spread of the H1N1 influenza pandemic of 2009 along with good respiratory and hand hygiene [50,51].
Universal wearing of face coverings (Masks) in the community
The controversy and confusion of wearing face coverings by individuals when in the community is sparked by the World Health Organization (WHO) recommending against the wearing of face coverings because of the lack of evidence of protection against the coronavirus [52,53]. There is a paucity of studies that are inconclusive on the efficacy of face covering (mask) use and the ability to prevent transmission of infectious disease from respiratory droplets and aerosols in entire communities. Further, there are no studies that evaluated the effectiveness of face mask use during a pandemic.
In a meta-analysis, Xiao and colleagues (2020) concluded that there is no significant reduction in laboratory influenza transmission [54]. Other studies demonstrated limited evidence that wearing a face mask in households, in the presence of an individual suffering from the influenza virus, or in mass social gatherings will prevent the spread of the influenza virus [55,56]. In a systematic review by Brainard et al (2020) examining whether wearing a face mask with other protective measures would prevent the transmission of respiratory illness such as coronavirus, influenza and tuberculosis, the authors concluded that the evidence is not sufficiently strong to support the widespread use of face masks as a protective measure against Covid-19 [57].
Although there are no studies assessing the effectiveness of face coverings during a pandemic, the lack of protection of viral transmission remains unclear. Use of face coverings is a form of source control that may protect individuals from respiratory droplets or aerosols. The US Centers for Disease Control and Prevention now advises the wearing of cloth face coverings in public [58]. It has recently been discovered that the coronavirus sheds at high concentrations from the nasal cavity 2-3 days before the onset of symptoms and continues for at least one week after symptom onset [7,8,18,19,23]. Therefore, asymptomatic or presymptomatic individuals could be potential super spreaders of the stealth coronavirus as they are indistinguishable from healthy individuals in the community. Moreover, Booth et al (2013), demonstrated that use of surgical face masks resulted in a six-fold reduction in exposure to the influenza virus in aerosols and recommends the use of face masks [59]. Lastly, in a study by Leung et al (2020) use of surgical face masks prevented the transmission of both the coronavirus and influenza from individuals who are symptomatic [60].
Routine diagnostic testing for coronavirus antibody and antigen
The debate over what type of diagnostic test to implement and whether to test both asymptomatic and patients that exhibit signs and symptoms for the coronavirus have led to a delay in the effective response to flatten the pandemic curve and decrease the reproductive number. Further, knowledge of diagnostic tests specific for the coronavirus is still evolving and is contributing to the delay in mitigation of this novel emerging infectious disease. Currently, the gold standard for diagnosis of early active RNA viral infection is the polymerase chain reaction (PCR) test. PCR testing can identify viral genetic material in sufficient amounts from a specimen obtained from the nasopharynx or other respiratory tract specimens, including saliva [61,62]. However, PCR testing may fail to detect viral infection if insufficient amounts of the virus are not obtained and missing the critical time of viral replication may result in false negative results. A false negative result will allow infected individuals to circulate in the community spreading the virus [63
Coronavirus infection can also be detected indirectly by evaluating the host immune response to Covid-19 infection using serological assays. In a study by Zhang and colleagues (2020) they reported that seroconversion occurred 7 to 12 days after the onset of symptoms [64]. Understanding of viral dynamics is important to evaluate the penetration of the coronavirus in the community. Most important, serological testing may identify which individuals have developed herd immunity and protected from infection [65]. Detection of antibodies, especially IgM which is produced after the infection can be used concurrently with PCR testing to increase sensitivity and accuracy in detection of the coronavirus [66]. Therefore, what is urgently needed and not yet available is an portable rapid point-of care test for detection of SARS-CoV-2 specific IgM and IgG that can identify infected persons in real-time in a matter of minutes and does not need highly trained laboratory personnel to complete the testing.
Also not yet available but the need is paramount is a test to detect the presence of viral antigens that will identify which individuals are actively infected with the coronavirus and who must go into isolation to prevent viral transmission in the community. This is critical for screening individuals in the community, including healthcare staff to determine who has become actively infected [67]. The advantage of antigen detection is because coronavirus infections cannot be diagnosed strictly by clinical symptoms and it has been demonstrated that viral shedding occurs before the onset of symptoms [7,8,18,19,23]. In a study by Bruning and colleagues (2018), they were able to identify the coronavirus as the etiology for infection and antigen level in six patients who developed symptoms of respiratory disease [61]. They also observed a correlation between severity of clinical symptoms and antigen levels.
Opponents of routine diagnostic testing are concerned that testing large populations will overwhelm healthcare facilities and their services that are needed to treat more severely ill patients [68]. In addition, individuals that do not have the coronavirus will be exposed to persons that may actually harbor the coronavirus. Other concerns are misinterpretation of the testing results. As the coronavirus incubation period is 4- 5 days, such a window of infectivity may result in individuals believing they tested negative for the coronavirus. However, a positive test reflects the presence of viral RNA but does not indicate viability of the coronavirus. Such uncertainty about infectivity will still require isolation of that individual from the public [69].
But individuals that have mounted an immune response to the coronavirus that causes Covid-19 may be immune and unlikely to spread the virus to others in the community. Testing for the coronavirus to ease social distance, isolation and quarantine restrictions makes testing an integral part of public health and restoring civil liberties [70]. Surveillance is a time-tested public health strategy to protect communities from emerging infectious diseases [5,6]. Not only is surveillance important in identifying disease outbreaks that could progress to a pandemic but is also involved in containment and eradication of the threat. To monitor disease outbreaks, surveillance is utilized which relies on symptoms as described in this paper before a diagnosis is arrived at.
Social distancing is a community mitigation strategy designed to decrease non-essential gatherings between individuals to reduce viral transmission and the reproductive number of the virus [48,71,72]. Such measures may flatten the pandemic curve by increasing the physical distance between individuals and decreasing the population density. Examples of social distancing include the cancellation of musical concerts, sporting events, open food markets, and the closing of schools and businesses. Such interventions when implemented early during an outbreak were associated with lower peak death rates and viral transmission [5,73]. Because of the ability to conduct educational classes at all levels and business online over the Internet, students learning at home and employees working from home should be seriously considered to control the spread of the pandemic [51]. In a systematic review by Ahmed et al (2018), social distancing in the non-healthcare workplace has been shown to decrease the risk of influenza transmission that occurs within a distance of 6 feet or greater [74].
Social distancing is not without controversy and challenges for community acceptance as public health policy is created to protect the entire community population and not the individual. Social distancing may not be sustainable as schools and businesses close, and unemployment continues to rise [75]. Reluctance or lack of compliance with social distancing measures will increase the rate of disease transmission. This has been observed when social distancing measures were implemented after the contagion has entered the community. But, implementing social distancing measures too early may result in poor compliance due to fatigue and economic hardship [76,77]. As there is no clear criteria about the relaxation of social distancing restrictions and other NPI measures that include isolation and quarantine, social distancing must be tailored to individual-level information that relies on surveillance and antigen or antibody testing for the coronavirus. The importance of this policy is that mass testing of individuals will avoid broader community-level restrictions on civil liberties [78].
Closure of schools as an NPI have proven effective in mitigating the spread of the influenza pandemic [79,80]. It was observed that early implementation of school closures resulted in reducing the peak of the epidemic [81]. However, school closings before or after the influenza peak showed no differences in decreasing the spread of the influenza epidemic [81]. During times of an pandemic, if the R0 is low, school closings may be effective at reducing viral transmission to below 1. In contrast, if the R0 is high indicating sustained viral transmission, school closure may not have the same impact [75]. What remains unclear is the specific duration of school closure time and its effect on eradication of the disease. It was observed that the longer schools were closed, the longer the delay in the peak of the influenza outbreak. Therefore, a specific duration of school closures is not recommended.
Isolation and quarantine are two time-tested public health mitigation strategies that attempt to slow down community viral transmission while trying not to breach civil liberties and human rights [48,82,83]. Isolation is the separation and restriction of movement of individuals who have become infected with the contagious disease to prevent transmission to others. For isolation to be effective, two epidemiological factors must be considered, and they are the number of secondary infections from the index source and the degree of viral transmission that occurs before the onset of symptoms. Viral transmission before onset of symptoms can be prevented by tracing contacts of infected individuals and placing them in a 14-day quarantine [10,50,51]. Isolation procedures can be implemented at home or any special designated facility [48, 51,74].
Individuals who are asymptomatic or minimally symptomatic should remain in isolation until they are without symptoms of respiratory infection (fever and cough). However, isolation is not without personal challenges. Isolation of infected individuals to decrease the spread of viral transmission requires cooperation to adhere to this intervention. Such intervention may be inconvenient socially and could result in loss of economic viability [84]. Quarantine can be completed voluntarily, or by legal authority and refers to the restriction of movement and separation of individuals who have been exposed to an infectious disease before they become infected and transmit the disease to others [78,82,83]. Quarantine can occur in the individuals own home or in a designated facility. Although considered an extreme NPI measure, quarantine has been shown to contain the spread of secondary cases from the index case that decreases the reproductive number (R0) to less than 1 [85,86]. In many parts of the world, especially the United States restricting movement of healthy individuals for weeks or months may not be considered socially acceptable as it infringes on individual civil liberties [78,83].
With cities or communities designated “hot spots”, communitylevel quarantine, (the “cordon sanitaire”) is an extreme interventional measure that involves the closing off of an entire city, community or neighborhood from external contacts designed to increase social distancing and reducing disease transmission [2,48,87]. To control the spread of Covid-19, the Chinese government effectively used cordon sanitaire with draconian social control, armed police and electronic surveillance of its people to close off the city of Wuhan and Hubei province confining 60 million people from entering or leaving. Pan et al (2020) reported that cordon sanitaire with other NPI measures effectively decreased coronavirus transmission and the R0 to less than 1 [88]. As the Covid-19 pandemic continues to spiral out of control, it may be necessary to quarantine “hot spots” to contain disease transmission to other regions of the country. In the United States, the use of cordon sanitaire will no doubt encounter extreme opposition and resistance. Further, there are no broad national quarantine powers that would allow for the closing off of large cities all in the name of public health [82,83]. Such a mass quarantine of both infected and non-infected individuals would violate public health statutes. The more acceptable less restrictive NPI measures would be surveillance, contact tracing with isolation and social distancing while monitoring rates of infection and the reproduction number [89].
Contact tracing with isolation and quarantine has always been part of the strategy to mitigate disease transmission by identifying persons exposed to the infected individual and who may need medical evaluation [25,90]. The definition of a close contact is an individual who did not wear personal protection equipment (PPE) while in face-to-face contact with a confirmed case for 15 minutes or greater after onset of symptoms. As many individuals are harboring the coronavirus and are asymptomatic, isolation of infected individuals and contact tracing may not be possible due to the inability to identify this cohort [18]. Therefore, successful contact tracing and decreasing the delay between onset of symptoms and isolation are important as infected individuals can continue to transmit the virus while circulating in the community.
It has been shown that with airborne infections many of the network’s contacts may not be traceable [91]. If the disease is rapidly spreading in the community and the amount of contacts identified are occurring at a slow rate, contact tracing may not be able to keep pace with pandemic spread. Under these conditions, the reproductive number will be greater than 1 and the outbreak will continue to progress due to an increase in the number of infected cases [27,92]. Although non-pharmaceutical intervention strategies were effective in controlling the spread of the 1918 influenza virus, in a study by Hatchett et al (2007) reintroduction and transmission of the virus returned and extended for many months as the containment strategies were relaxed too early in many cities (Figure 1) [92]. In a study comparing 17 cities, most of the NPIs were implemented between 2 and 6 weeks. Cities that maintained their interventions beyond 6 weeks did not experience a second wave of the influenza pandemic. In contrast, cities that relaxed their interventional strategies too early experienced not only a second pandemic wave, but greater population mortality that was not observed in cities that maintained their interventions beyond six weeks [71,93]. Therefore, a critical issue that must be determined by public health authorities is when to relax the interventions implemented to control the spread of viral transmission. Without a vaccine or antivirals available to protect individuals in the community, individuals will remain vulnerable to the pandemic virus. With such uncertainty, communities may have to maintain their interventions longer that 8 weeks [92].
Conclusion
Reliance upon NPIs that include the routine use of surgical face coverings and antibody/antigen testing for both symptomatic and asymptomatic individuals should be initiated immediately to contain the spread of the novel human coronavirus. If these intervention strategies fail or relaxed too early in the course of this pandemic, the trajectory of the pandemic will continue to rise and will continue to overwhelm our health care infrastructure leading to greater mortality and economic demise in our communities.
Acknowledgement
None.
Conflict of Interest
No conflict of interest.
References
- Peiris JSM, Yuen KY, Osterhaus ADME, Klaus Stöhr (2003) The severe acute respiratory syndrome. Engl J Med 349(25): 2431-2441.
- Weinstein RA (2004) Planning for epidemics- the lessons of SARS. N Engl J Med 350(23): 2332-2334.
- Johnson NP, Mueller J (2002) Updating the accounts: global mortality of the 1918-1920 “Spanish” influenza pandemic. Bull Hist Med 76: 105-115.
- Taubenberger JK, Morens DM (2006) 1918 Influenza: the mother of all pandemics. Emerg Infect Dis 12: 15-22.
- Markel H, Lipman HB, Navarro JA, Alexandra Sloan, Joseph R Michalsen, et al. (2007) Nonpharmaceutical interventions implemented by US cities during the 1918-1919 influenza pandemic. JAMA 298(6): 644-654.
- Fineberg HV (2014) Pandemic preparedness and response-lessons from the H1N1 influenza of 2009. New Engl J Med 370(14): 1335-1342.
- He X, Lau EHY, Wu P, Xilong Deng, Jian Wang et al. (2020) Temporal dynamics in viral shedding and transmissibility of COVID-19. Nature Medicine.
- Hoehl S, Rabenau H, Berger A, Marhild Kortenbusch, Jindrich Cinatl, et al. (2020) Evidence of SARS-CoV-2 infection in returning travelers from Wuhan, China. N Engl J Med 382: 1278-1280.
- Rothe C, Schunk M, Sothmann P, Gisela Bretzel, Guenter Froeschl, et al. (2020) Transmission of 2019-nCoV infection from an symptomatic contact in Germany. N Engl J Med 382(10): 970-971.
- Twu SJ, Chen TJ, Chen CJ (2003) Control measures for severe acute respiratory syndrome (SARS) in Taiwan. Emerging Infectious Diseases 9(6): 718-720.
- Hui DSC, Chan MCH, Wu AK (2004) Severe acute respiratory syndromes (SARS): epidemiology and clinical features. Postgrad Med J 80: 373-381.
- Metlay JP, Waterer GW, Long AC, Jan Brozek, Kristina Crothers, et al. (2019) Diagnosis and treatment of adults with community acquired pneumonia: an official clinical practice guideline of the American Thoracic Society of America. Am J Respir Crit Care Med 200(7): e45-e67.
- Huang C, Wang Y, Li X (2020) Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet 395: 497-506.
- Li Q, Guan X, Wu P (2020) Early transmission dynamics in Wuhan, China of novel coronavirus- infected pneumonia. N Engl J Med. 382: 1199-1207.
- Zhu N, Zhang D, Wang W, Xingwang Li, Bo Yang, et al. (2020) A novel coronavirus from patients with pneumonia in China. New Engl J Med 382: 727-733.
- World Health Organization. Coronavirus disease 2019 (COVID-19) Situation Report-51.
- (2020) World Health Organization, Geneva, Switzerland.
- (2020) Coronavirus disease (COVID-19) situation reports. World Health Organization, Geneva, Switzerland.
- Dorenmalen N, Bushmaker T, Morris DH, Myndi G Holbrook, Amandine Gamble, et al. Aerosol and surface stability of SARS-Cov-2 as compared with SARS-Cov-1. N Engl J Med 2020 382(16): 1564-1567.
- Mills CE, Robins JM, Lipsitch M (2004) Transmissibility of 1918 pandemic influenza. Nature 432: 904-906.
- Markel H, Lipman HB, Navarro JA, Alexandra Sloan, Joseph R Michalsen, et al. (2007) Nonpharmaceutical interventions implemented by US cities during the 1918-1919 influenza pandemic. JAMA 298(6): 644-654.
- Yu D, Lin Q, C APY, Daihai He (2017) Effects of reactive social distancing on the 1918 influenza pandemic. PLoS ONE 12(7): e0180545.
- Wu JT, Leung K, Leung GM (2020) Nowcasting and forecasting the potential domestic and international spread of the 2019-nCoV outbreak originating in Wuhan, China: modeling study. The Lancet 395(10225): 689-697.
- Zou L, Ruan F, Huang M, Lijun Liang, Huitao Huang, et al. (2020) SARS-CoV-2 viral load in upper respiratory specimens of infected patients. N Engl J Med 382(12): 1177-1179.
- Lipsitch M, Cohen T, Cooper B, James M Robins, Stefan Ma, et al. (2003) Transmission dynamics and control of severe acute respiratory syndrome. Science 300(5627): 1966-1970.
- Liu Y, Gayle AA, Wilder Smith A, Joacim Rocklöv (2020) The reproductive number of COVID-19 is higher compared to SARS coronavirus. J Trav Med 27(2): taaa021.
- Donnelly CA, Ghani AC, Leung GM, Anthony J Hedley, Christophe Fraser, et al. (2003) Epidemiological determinants of spread of causal agent of severe acute respiratory syndrome in Hong Kong. Lancet 361(9371): 1761-1766.
- Jia N, Feng D, Fang LQ, Jan Hendrik Richardus, Xiao-Na Han, et al. (2009) Case fatality of SARS in mainland China and associated risk factors. Trop Med Int Health 14 (Suppl): 21-27.
- Biggerstaff M, Cauchemez S, Reed C, Manoj Gambhir, Lyn Finelli (2014) Estimates of the reproduction number for seasonal, pandemic, and zoonotic influenza: a systematic review of the literature. BMC Infect Dis 14: 480.
- Bauch CT, Lloyd-Smith JO, Coffee MP, Alison P Galvani (2005) Dynamically modeling SARS, and other newly emerging respiratory illnesses: past, present, and future. Epidemiology 16(6): 791-801.
- Guan W, Ni N, Hu Y, Wen-hua Liang, Chun quan Ou, et al. (2020) Clinical characteristics of coronavirus disease 2019 in China. N Engl J Med 382: 1708-1720.
- Ghani AC, Donnelly CA, Cox DR, J T Griffin, C Fraser, et al. (2005) Methods for estimating the case fatality ratio for a novel emerging infectious disease. Am J Epidemiol 162(5): 479-486.
- Nishiura H, Klinkenberg D, Roberts M, Johan A P Heesterbeek (2009) Early epidemiological assessment of the virulence of emerging infectious diseases: a case study of an influenza pandemic. PLoS One 4(8): e6852.
- Battegay M, Kuehl R, Tschudin Sutter S, Hans H Hirsch, Andreas F Widmer, et al. (2019) 2019-novel coronavirus (2019-nCoV): estimating the case fatality rate- a word of caution. Swiss Med Wkly 150: w20203.
- Chen N, Zhou M, Dong, X, Jieming Qu, Fengyun Gong, et al. (2020) Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: a descriptive study. Lancet 395: 507-513.
- Wang D, Hu B, Hu C, Fangfang Zhu, Xing Liu, et al. (2020) Clinical characteristics of 138 hospitalized patients with 2019 novel coronavirus-infected pneumonia in Wuhan, China. JAMA 323(11): 1061-1069.
- Chu CM, Poon LL, Cheng VC, Kin Sang Chan, Ivan F N Hung, et al. (2004) Initial viral load, and the outcomes of SARS. CMAJ 171(11): 1349-1352.
- Bai Y, Yao L, Wei T, Fei Tian, Dong Yan Jin, et al. (2020) Presumed asymptomatic carrier transmission of COVID-19. JAMA 323(14): 1406-1407.
- He X, Lau EHY, Wu P, Xilong Deng, Jian Wang, et al. (2020) Temporal dynamics in viral shedding and transmissibility of COVI-19. Nat Med 26(5): 672-675.
- Wu Z, McGoogan JM (2020) Characteristics of and important lessons from the coronavirus disease 2019: (COVID-19) outbreak in China: summary or a report of 72, 314 cases from the Chinese Center for Disease Control and Prevention. JAMA.
- Tsai LK, Hsieh ST, Chao CC (2004) Neuromuscular disorders in severe acute respiratory syndrome, Arch Neurol 61(11): 1669-1673.
- Mao L, Jin H, Wang M, Yu Hu, Shengcai Chen (2020) Neurologic manifestations of hospitalized patients with coronavirus disease 2019 in Wuhan, China. JAMA Neurol 77(6):1-9.
- Oxley TJ, Mocco J, Majidi S, Christopher P Kellner, Hazem Shoirah, et al. (2020) Large-vessel stroke as a presenting feature of covid-19 in the young. N Engl J Med 382(20): e60.
- Giacomelli A, Pezzati L, Conti F, Dario Bernacchia, Matteo Siano, et al. (2020) Self-reported olfactory and taste disorders in SARS-CoV-2 patients: a cross sectional study. Clin Infect Dis 71(15): 889-890.
- Bagheri SHR, Asghari AM, Farhadi M, Ahmad Reza Shamshiri, Seyed Kamran Kamrava, et al. (2020) Coincidence of COVID-19 epidemic and olfactory dysfunction outbreak. Otolaryngology.
- Hwang CS (2006) Olfactory neuropathy in sever acute respiratory syndrome: report of a case. Acta Neurol Taiwan. 15(1): 26-28.
- Sabrina Jones (2007) American Public Health Association. APHA Prescription for Pandemic Flu. Washington DC: American Public Health Association 7(6): 287-298.
- Wilder-Smith A, Freedman DO (2020) Isolation, quarantine, social distancing, and community containment: pivotal role for old-style public health measures in the novel coronavirus (2019-nCoV) outbreak. J Travel Med 1-4.
- Liu Y, Ning Z, Chen Y, Yingle Liu, Nirmal Kumar Gali, et al. (2020) Aerodynamic analysis of SARS-CoV-2 in two Wuhan hospitals. Nature 582(7813): 557–560.
- Wu JT, Riley S, Fraser C, (2006) Reducing the impact of the next influenza pandemic using household-based public health interventions. PLoS Med.; 3(9) (e361): 1532-1540.
- Qualls N, Levitt A, Kanade N, Narue Wright Jegede, Stephanie Dopson, et al. (2017) Community mitigation guidelines to prevent pandemic influenza- United States, 2017. MMWR 66: 1-34.
- WHO (2020) Advice on the use of masks in the community, during home care, and in healthcare settings in the context of the novel coronavirus (2019-nCoV) outbreak interim guidance.
- Chan AL, Leung CC, Lam TH (2020) To wear or not to wear: WHO’s confusing guidance on masks in the covid-19 pandemic. BMJ Blog.
- Xiao J, Shiu EYC, Gao H (2020) Nonpharmaceutical measures for pandemic influenza in nonhealthcare settings- personal protective and environmental measures. Emerg Infect Dis 26(5): 967-975.
- Aiello AE, Coulborn RM, Perez V, Rebecca M Coulborn, Brian M Davis, et al. (2010) A randomized intervention trial of mask use and hand hygiene to reduce seasonal influenza-like illness and influenza infections among young adults in a university setting. Int J Infect Dis 1(4): E320-E20.
- Suess T, Remschmidt C, Schink SB, Brunhilde Schweiger, Andreas Nitsche, et al. (2012) The role of facemasks and hand hygiene in the prevention of influenza transmission in households: results from a cluster randomized trial; Berlin, Germany, 2009-2011. BMC Infect Dis 12: 26.
- Brainard JS, Jones N, Lake I (2020) Facemasks and similar barriers to prevent respiratory illness such as COVID-19: a rapid systematic review. MedRxiv.
- (2020) US Center for Disease Control and Prevention. Recommendation regarding the use of cloth face coverings, especially in areas of significant community-based transmission.
- Booth CM, Clayton M, Crook B (2013) Effectiveness of surgical masks against influenza bioaerosols. J Hosp Infect 84: 22-26.
- Leung NHL, Chu DKW, Shiu EYC, Kwok Hung Chan, James J, et al. (2020) Respiratory virus shedding in exhaled breath and efficacy of face masks. Nat Med 26(5): 676-680.
- AHL Bruning, H Aatola, H Toivola, N Ikonen, C Savolainen Kopra, et al. (2018) Rapid detection and monitoring of human coronavirus infections. New Microbes New Infect 24: 52-55.
- Sethuraman N, Jeremiah SS, Akihide Ryo (2020) Interpreting diagnostic tests for SARS-CoV-2. JAMA Doi:10.1001/jama.2020.8259.
- Camilla Rothe, Mirjam Schunk, Peter Sothmann, Gisela Bretzel, Guenter Froeschl, et al. (2020) Transmission of 2019-nCoV infection from an asymptomatic contact in Germany. N Engl J Med 382(10): 970-971.
- Zhang J, Liu J, Li N, et al. (2020) Serological detection of 2019-nCoV respond to the epidemic: A useful complement to nucleic acid testing. mdRxiv doi:10.20030916.
- Hoffman T, Nissen K, Krambrich J, Bengt Rönnberg, Dario Akaberi, et al. (2020) Evaluation of a COVID-19 IgM and IgG rapid test: an efficient tool for assessment of past exposure to SARS-CoV-2. Infect Ecol Epidem 10(1): 1754538.
- Xin Gao, Hongli Zhou, Chao Wu, Yan Xiao, Lili Ren, et al. (2015) Antibody against nucleocapsid protein predicts susceptibility to human coronavirus infection. J Infect 71: 599-602.
- Ahmed SF, Quadeer AA, McKay MR (2020) Preliminary identification of potential vaccine targets for the COVID-19 coronavirus (SARS-CoV-2) based on SARS-CoV immunological studies. Viruses 12(3): 254.
- Sharfstein JM, Becker SJ, Mello MM (2020) Diagnostic testing for the novel coronavirus. JAMA 323(15): 1437-1438.
- Wolfel R, Corman VM, Guggemos W, Michael Seilmaier, Sabine Zange, et al. (2020) Virological assessment of hospitalized patients with COVID-2019. Nature 581(7809): 465-469.
- Studdart DM, Hall MA (2020) Disease control, civil liberties, and mass testing- calibrating restrictions during the Covid-19 pandemic. New Engl J Med 383(2):102-104.
- Bootsma MCJ, Ferguson NM (2007) The effect of public health measures on the 1918 influenza pandemic in U.S. cities. Proc Natl Acad Sci U S A 104(18): 7588-7593.
- Kelso JK, Milne GJ, Kelly H (2009) Simulation suggests that rapid activation of social distancing can arrest epidemic development due to a novel strain of influenza. BMC Public Health 9: 117.
- Inglesby TV, Nuzzo JB, O Toole T, et al. (2006) Disease mitigation measures in the control of pandemic influenza. Biosecur Bioterror 4(4): 366-375.
- Ahmed F, Zviedrite N, Uzicanin A (2018) Effectiveness of workplace social distancing measures in reducing influenza transmission: a systematic review. BMC Public Health 18(1): 518.
- Jackson C, Mangtani P, Hawker J, Babatunde Olowokure, Emilia Vynnycky, et al. (2014) The effects of school closures on influenza outbreaks and pandemics: systematic review of simulation studies. PLoS One 9(5): e97297.
- Collinson S, Khan K, Hefferman JM (2015) The effects of media reports on disease spread and important public health measurements. PLoS One 10(11): e0141423.
- Saunders Hastings P, Reisman J, Krewski D (2016) Assessing the state of knowledge regarding the effectiveness of interventions to contain pandemic influenza transmission: a systematic review and narrative synthesis. PLoS One 11(12): e0168262.
- Fong MW, Gao H, Wong JY, et al. (2020) Nonpharmaceutical measures for pandemic influenza in nonhealthcare settings- social distancing measures. Emerging Infec Dis 26(5).
- Van Tam J (2005) WHO publishes new Global Influenza Preparedness Plan. Euro Surveill Bulletin European Sur Les Maladies Transmissibles Eur Commun Dis Bull 14 (April (10)). E050414.2.
- Eames K (2018) The influence of school holiday timing on epidemic impact. Epidemiol Infect 142(9): 1963-1971.
- Sharafaldeen Bin Nafisah, Aliyah H Alamery, Aminah Al Nafesa, Bakhitah Aleid, Nora A Brazanji (2018) School closure during novel influenza: a systematic review. J Infect Public Health 11(5): 657-661.
- Cetron M, Landwirth J (2005) Public health and ethical considerations in planning for quarantine. Yale J Biol Med 78(5): 329-334.
- Parmet WE, Sinha MS (2020) Covid-19- the law and limits of quarantine. J Engl J Med 382(15): e28.
- Menickella B (2020) COVID-19 worldwide: the pandemic’s impact on the economy and markets.
- De Vlas SJ, Feng, D, Cooper BS, et al. (2009) The impact of public health control measures during the SARS epidemic in mainland China. Trop Med Int Health 14 Suppl 1:101-104.
- Wayne TA Enanoria, Fengchen Liu, Jennifer Zipprich, Kathleen Harriman, Sarah Ackley, et al. (2016) The effect of contact investigations and public health interventions in the control and prevention of measles transmission: a simulation study. PLoS One 11(12): e0167160.
- Cetrone M, Simone P (2004) Battling 21st century scourges with a 14th century toolbox. Emerg Infect Dis 10(11): 2053-2054.
- An Pan, Li Liu, Chaolong Wang, Huan Guo, Xingjie Hao, et al. (2020) Association of public health interventions with the epidemiology of the COVID-19 outbreak in Wuhan, China. JAMA 323(19): 1-9.
- Gostin LO (2020) Could- or should -the government impose a mass quarantine on an American city? Health Affairs.
- Kiss IZ, Green DM, Kao RR (2005) Disease contact tracing in random and clustered networks. Proc Biol Sci 272(1570): 1407-1414.
- Edmunds WJ, O Callaghan CJ, Nokes DJ (1997) Who mixes with whom? A method to determine the contact patterns of adults that may lead to the spread of airborne infections. Proc Biol Sci 264(1384): 949-957.
- Hatchett RJ, Mecher CE, Lipsitch M (2007) Public health interventions and epidemic intensity during the 1918 influenza pandemic. PNAS 104(18): 7582-7587.
- Murray CJL, Lopez AD, Chin B, Dennis Feehan, Kenneth H Hill (2006) Estimation of potential global pandemic influenza mortality on the basis of vital registry data from the 1918-20 pandemic: a quantitative analysis. Lancet 368(9554): 2211-2218.
-
Cameron YS Lee, Kristin L Lee. Use of Antibody/Antigen Diagnostic Testing and Face Coverings with Traditional Non-Pharmaceutical Intervention Measures to Mitigate Human-to-Human Coronavirus (COVID-19) Transmission at the Community Level. 1(2): 2020. APHE. MS.ID.000507.
-
Acute respiratory syndrome, Infectious disease, Medical treatment, Fever, chills, Fatigue, Myalgia, Viral antigens, Clinical symptoms.
-

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.






