 Review Article
Review Article
Cenobamate: New Antiseizure Medication with High Efficacy
Steve Chung*
Professor of Neurology, Banner University Medical Center-Phoenix University of Arizona-Phoenix, USA
Steve Chung MD, Professor of Neurology, Banner University Medical Center-Phoenix University of Arizona-Phoenix, USA.
Received Date: October 18, 2021; Published Date: November 17, 2021
Introduction
Approximately 30% of epilepsy patients experience recurrent seizures despite many new antiseizure medications (ASMs) became available over the past 25 years. Cenobamate is a newest FDA approved ASM for the treatment of focal onset seizures in adults as both monotherapy and adjunctive therapy in November 2019 [1]. Pivotal clinical trials reported superior seizure improvement in patients with focal epilepsy.
Mechanisms of Action
Cenobamate has anti-seizure effects on voltage-gated Na+ channels and GABAA receptors in preclinical studies. Cenobamate delayed the time to recovery from fast and slow inactivation of voltage-dependent Na channels in depolarized neurons. In addition, cenobamate is also a positive allosteric modulator of GABAA receptors in hippocampal neurons, enhancing GABA inhibition of neurons [2].
Clinical Pharmacokinetics
Cenobamate was well absorbed after oral ingestion, and not influenced by food. The half-life for cenobamate is approximately 50 to 60 hours after single doses of 100 to 400 mg. Plasma binding of cenobamate was about 60% in healthy subjects. Cenobamate is metabolized by hepatic glucuronidation and then oxidation. It inhibits hepatic isoenzyme CYP2C19 (potentially increases phenytoin and phenobarbital serum level) and induces CYP3A4 (potentially reduces midazolam and simvastatin serum level). Therefore, modification of certain medications may be necessary when co-administering cenobamate. There were no significant influences of cenobamate on warfarin concentrations [3,4].
Efficacy
The efficacy of cenobamate was established by the results from two double-blind, randomized, placebo-controlled, parallel-group, multicenter, multinational studies. Both studies were performed in adult patients with medically refractory focal epilepsy to evaluate the efficacy, safety, and tolerability of cenobamate. These studies had 8-week baseline period to establish patients’ baseline 28-day seizure frequency. Patients were required to have at least 3-4 focal seizures per 28 days and be stable on treatment with 1-3 ASMs. Patients had a mean duration of epilepsy of approximately 22 years and median baseline seizure frequency of 8.5 seizures per 28 days. The first study had a single target dose of 200 mg/day. A statistically significant improvement was observed in cenobamate arm with 55.6% (n=113, p<0.0001) median percent seizure reductions from baseline, compared to 21.5% in the placebo arm [5]. In addition, a significant difference in the 50% responder rates (patients with ≥50% reduction in seizure frequency from baseline to maintenance) were also observed in the cenobamate treatment group (50%, p<0.0001), compared to placebo (22%). Seizure freedom was achieved in 28% (p=0.0001) among cenobamate group, compared to 1% for placebo. The second study had multiple cenobamate doses of 100 mg, 200 mg, or 400 mg per day and placebo. The median percent reductions from baseline in seizure frequency per 28 days were 36.3%, 55.2%, and 55.3% for those patients who received cenobamate at 100 mg, 200 mg, 400 mg, respectively, compare to 24.3% in placebo arm [6]. Higher seizure freedom was achieved in cenobamate ranged from 4 to 21%. In addition, cenobamate showed statistically significant reductions in seizure frequency across all types of focal seizures including focal aware motor, focal impaired awareness, focal to bilateral tonicclonic seizures in both studies. A post hoc analysis of these two studies revealed that cenobamate treated seizure improvements were observed regardless of baseline seizure frequency, disease duration, concomitant number or kind of baseline ASMs [5,6].
Safety
Cenobamate was generally well tolerated, with a consistent safety profile in clinical studies. The most frequent Treatment- Emergent Adverse Effects (TEAEs) reported with cenobamate were somnolence and fatigue-related adverse reactions (somnolence, fatigue, asthenia, malaise, hypersomnia, sedation, and lethargy). The second most common cenobamate related TEAEs were dizziness and disturbance in gait and coordination (dizziness, vertigo, balance disorder, ataxia, nystagmus, gait disturbance, and abnormal coordination). Cognitive dysfunction related events (i.e., memory impairment, disturbance in attention, amnesia, confusion, aphasia, speech disorder, slowness of thought, disorientation, and psychomotor retardation) were infrequent. Formal cardiac ECG studies demonstrated shortening of the QT interval (mean = 16 msec, for doses greater than 400 mg daily) with cenobamate. Patients with Familial Short QT syndrome should not be treated with cenobamate [1]. There were no other clinically significant changes from the baseline in hematology, clinical chemistry, and other laboratory tests.
Table 1: Cenobamate Summary
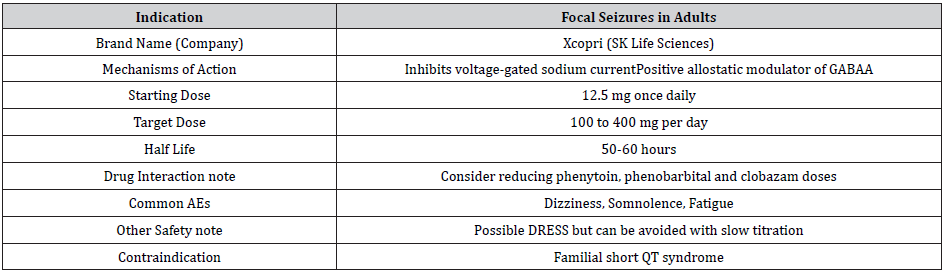
DRESS: Drug rash with eosinophilia and systemic symptoms
AEs: Adverse effects
During the clinical development of cenobamate, three cases of drug rash with eosinophilia and systemic symptoms (DRESS) were reported among 2,528 study patients. DRESS is a rare systemic drug reaction and may present with fever, rash, lymphadenopathy, and facial swelling, in association with other organ system involvement. In all three cases, cenobamate was initiated at a higher starting dose and titrated faster. To further evaluate the safety of cenobamate another large study (n=1,339) was conducted at the lower starting dose with slower titration rate. Patients in this study began cenobamate treatment with a low starting dose of 12.5 mg/ day and titrated up slowly every 2 weeks to a target dose of 200 mg/day and then up to a possible maximum dose of 400 mg/day as tolerated. With this slow titration of cenobamate, no cases of DRESS were reported [7]. Therefore, cenobamate should be initiated at 12.5 mg once daily and titrated every two weeks.
Summary
Cenobamate is approved in the US for the treatment of focal seizures in adult patients (>18 years) as monotherapy and adjunctive therapy. Cenobamate was highly effective in two pivotal trials for reducing focal seizures, and seizure freedom was achieved in about 20% of refractory epilepsy patients, which was higher than other newly approved ASMs. Common AEs include dose-dependent dizziness and somnolence related symptoms. In order to minimize risks for DRESS, cenobamate should be titrated gradually with initial dose of 12.5 mg once a day followed by increases every two weeks to a maintenance dose of 100 to 400 mg/day.
Acknowledgment
None.
Conflict of Interest
No conflict
References
- Prescribing information (2019) Paramus, NJ: SK Life Science. Available at: https://www.accessdata.fda.gov/drugsatfda_docs/label/2019/212839s000lbl.pdf. Accessed October 12, 2021
- Sharma R, Nakamura M, Neupane C, Byeong Hwa Jeon, Hyewon Shin, et al. (2020) Positive allosteric modulation of GABAA receptors by a novel antiepileptic drug cenobamate. European Journal of Pharmacology 879: 173117.
- Hansten P, Horn J (2019) The Top 100 Drug Interactions: A Guide to Patient Management. H & H Publications.
- Greene SA, Kwak C, Kamin M (2019) The Effect of Cenobamate On The Single-Dose Pharmacokinetics Of Multiple Cytochrome P450 Probes Using A Cocktail Approach In Healthy Subjects [Data on File] SK Life Science.
- Chung SS, French JA, Kowalski J, Gregory L Krauss, Sang Kun Lee, et al. (2020) Randomized phase 2 study of adjunctive cenobamate in patients with uncontrolled focal seizures. Neurology 94(22): e2311–e2322.
- Krauss GL, Klein P, Brandt C, Sang Kun Lee, Ivan Milanov, et al. (2020) Safety and efficacy of adjunctive cenobamate (YKP3089) in patients with uncontrolled focal seizures a multicentre, double-blind, randomised, placebo-controlled, dose-response trial. The Lancet Neurology 19(1): 38-48.
- Sperling MR, Klein P, Aboumatar S, Michael Gelfand, Jonathan J Halford, et al. (2020) Cenobamate (YKP3089) as adjunctive treatment for uncontrolled focal seizures in a large phase 3, multicenter, open-label safety study. Epilepsia 61(6): 1099-1108.






