 Research Article
Research Article
Parents and Care Givers Radiation Exposure from High Dose 131I-MIBG Therapy in Pediatrics With NBL; A KFSHRC - Saudi Arabia Initial Experience
Mohammed Al Rowaily*1,2,3
1 Radiology Department, King Faisal Specialist Hospital & Research Centre, Riyadh, Saudi Arabia
2 Alfaisal University, 11533 Riyadh, Saudi Arabia
3 KSU, 11433 Riyadh, Saudi Arabia
Mohammed Al Rowaily, Radiology Department, King Faisal Specialist Hospital & Research Centre, Alfaisal University, KSU, Riyadh, Saudi Arabia
Received Date:February 26, 2026; Published Date:March 20, 2026
Abstract
To our knowledge, there are no curative salvage treatments available to date. It is in this scenario that 131I-MIBG has been widely investigated
as a potential therapeutic radiopharmaceutical. This high avidity of MIBG for NBL makes it an ideal radiopharmaceutical for therapy as well as
imaging. KFSHRC as a major centre in Middle East provide this treatment and capable of using High Dose 131I-MIBG Therapy because of availability
autologous stem cell. Radiation exposure to parents and caregiver was investigated to provide high safety level.
Background: High-risk and relapsed neuroblastoma (NBL) presents a significant therapeutic challenge, with a pronounced lack of curative
salvage regimens. 131I-Metaiodobenzylguanidine (131I-MIBG) therapy, leveraging the high avidity of neuroblastoma cells for the MIBG molecule, has
emerged as a potent targeted radiopharmaceutical option. As a major referral centre, King Faisal Specialist Hospital and Research Centre (KFSHRC)
is uniquely positioned in the Middle East to deliver high-dose 131I-MIBG therapy, supported by its robust autologous stem cell transplantation
program. This study reports our initial experience, with a specific focus on quantifying radiation exposure to parents and caregivers to validate
established safety protocols.
Methods: We analysed data from three pediatric patients who received a total of five therapeutic doses. The first two doses were administered
at 444 MBq kg−1 (12 mCi kg-1), and the subsequent three at an escalated dose of 666 MBq kg-1 (18 mCi kg-1). Radiation exposure rates (in mSv/
hour) were meticulously measured for a representative administered activity of 200 mCi (7400 MBq) at varying distances (0.5 to 5 meters) over a
four-day post-therapy period. Urinary excretion rates of the radiopharmaceutical were also tracked.
Results: Monitoring revealed a rapid clearance of 131I-MIBG, with over 50% of the injected activity excreted in the urine within the first 24
hours, over 75% by 48 hours, and over 90% by 72 hours. Correspondingly, radiation exposure rates demonstrated a rapid decline, consistent with
this pharmacokinetic profile. All measured exposure levels for caregivers, even at close proximity (0.5 m) during necessary interactions, were well
below international safety limits.
Conclusion: High-dose 131I-MIBG therapy for neuroblastoma can be safely and effectively administered at KFSHRC. The comprehensive
radiation safety data confirm that with proper protocols, the exposure to parents and caregivers is minimal and within internationally mandated
constraints, thereby affirming the feasibility of this advanced treatment in the region.
Keywords:131I-MIBG therapy high-dose radiopharmaceutical; radiation safety; pediatric oncology; autologous stem cell transplantation; caregiver exposure; radiation dosimetry; middle east
Introduction
KFSHRC one of few if not the only centre in Middle East provides this treatment and capable of using High Dose 131I-MIBG Therapy because of availability autologous stem cell. 3 Pediatrics Patients (5 Doses) was administered systemically, first tow doses were treated with dose 444 Mbq kg-1 (12 mCi kg-1) the other three doses were 666 MBq kg−1 (18 mCi kg-1). Neuroblastoma (NBL), a malignancy arising from the sympathetic nervous system, is the most common extracranial solid tumour in children. Despite multimodal treatment approaches incorporating chemotherapy, surgery, and radiotherapy, a substantial proportion of patients with high-risk disease experience relapse, facing a dismal prognosis with few curative options. The urgent need for effective salvage therapies has driven the investigation of targeted molecular treatments. Among the most promising of these is 131I-Metaiodobenzylguanidine (131I-MIBG). The MIBG analogue is actively taken up by neuroblastoma cells via the norepinephrine transporter, and when labelled with the betaemitting radioisotope Iodine-131, it delivers a potent, localized cytotoxic radiation dose to tumour cells. This specific avidity makes 131I-MIBG an ideal theragnostic agent, useful for both diagnostic imaging and targeted radiotherapy [1].
The administration of high-dose 131I-MIBG, capable of delivering ablative radiation to tumours, is a complex procedure that necessitates stringent radiation safety measures and the support of an autologous stem cell transplant (ASCT) service to rescue the patient from inevitable myelosuppression. King Faisal Specialist Hospital and Research Centre (KFSHRC) have established itself as a pioneer and a leading centre in the Middle East, being one of the very few, if not the only, institution capable of providing this advanced level of care due to its integrated oncology and transplant programs. This article outlines the initial experience of KFSHRC with high-dose 131I-MIBG therapy. Beyond demonstrating clinical feasibility, a primary objective of this report is to provide a detailed quantitative analysis of the radiation exposure to parents and caregivers, a critical aspect of patient management that ensures the safety of all involved and maintains public confidence in nuclear medicine therapies. Furthermore, this research is addressed specifically to clinical centres that recognize the need for this lifesaving treatment but may perceive its implementation as prohibitive due to limited resources or infrastructure. By detailing our practical experience, safety data, and patient management protocols, we aim to provide a realistic and actionable framework that can demystify the process and guide other institutions in the region and beyond in developing their own safe and effective 131I-MIBG therapy programs, thereby expanding access to this critical modality for a vulnerable pediatric population [2].
Methods
Patient Cohort and Treatment Protocol
A cohort of three pediatric patients with relapsed or refractory neuroblastoma was treated with a total of five cycles of high-dose 131I-MIBG therapy. The treatment was conducted in dedicated radiation isolation rooms. The first two doses were administered at a level of 444 MBq/kg (12 mCi/kg). Based on initial tolerance and safety, the protocol was escalated for the subsequent three doses to 666 MBq /kg (18 mCi/kg). All patients had previously undergone harvesting and cryopreservation of autologous stem cells, which were reinfused approximately two weeks post-therapy to mitigate severe haematological toxicity [3].
Radiation Safety and Exposure Monitoring
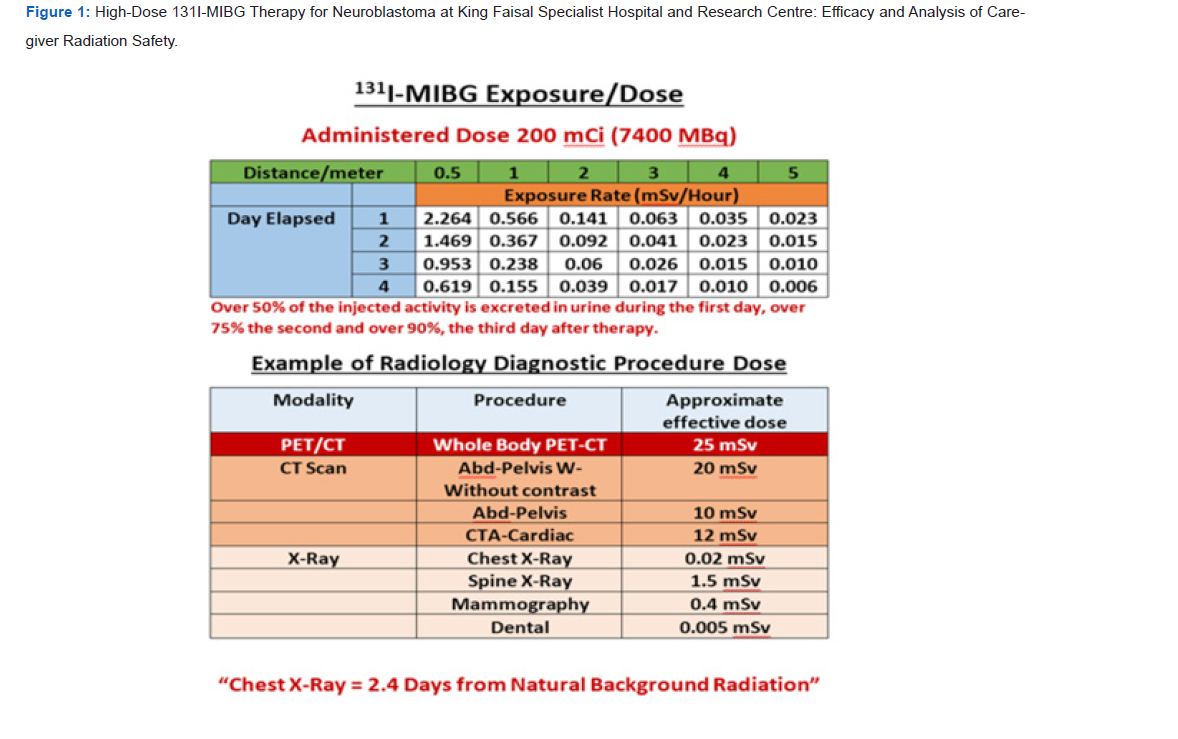
A critical component of the therapy protocol was the continuous monitoring of radiation exposure. For the purpose of standardized analysis, exposure rates were modelled for a typical administered activity of 200 mCi (7400 MBq). Measurements of the exposure rate (in mSv/hour) were taken at the patient’s bedside and at increasing distances (0.5 m, 1 m, 2 m, and 5 m) from the patient. This monitoring was conducted frequently over the first 96 hours (four days) post-administration.
Assessment of Radiopharmaceutical Clearance
The pharmacokinetic profile of 131I-MIBG was assessed by measuring the cumulative urinary excretion of the radioisotope. All patient urine was collected and assayed for radioactivity to determine the fraction of the administered dose excreted over time [4].
Results and Findings
Pharmacokinetics and Clearance of 131I-MIBG
The data confirmed the rapid systemic clearance of 131I-MIBG,
primarily through renal excretion.
a) Day 1: Over 50% of the injected radioactive activity was
excreted in the urine.
b) Day 2: Cumulative excretion exceeded 75% of the initial dose.
c) Day 3: By the end of the third day, over 90% of the
radiopharmaceutical had been cleared from the body.
This rapid decline in body burden is a key factor in reducing the duration of significant radiation exposure [5].
Radiation Exposure Analysis
The radiation exposure rates correlated directly with the
measured clearance kinetics. The highest exposure rates were
recorded immediately after infusion and decreased exponentially
over time. The data, summarized for a 200 mCi dose, showed that:
a) Exposure rates were highest at the closest distance (0.5 m) but
fell rapidly within the first 24 hours.
b) The inverse square law was clearly demonstrated, with
exposure rates dropping significantly with increasing distance
(e.g., exposure at 2 m was a fraction of that at 0.5 m).
c) By the fourth day, exposure rates at all measured distances
were negligible (Figure 1).
Most importantly, when these exposure rates were integrated over the isolation period and combined with strict time-limiting protocols for caregiver proximity, the calculated total effective radiation dose to parents and caregivers was consistently and significantly below the annual public dose limit of 1 mSv as recommended by the International Commission on Radiological Protection (ICRP) [6].
Discussion
The successful implementation of high-dose 131I-MIBG therapy represents a significant advancement in the regional management of high-risk neuroblastoma. The program at KFSHRC effectively bridges a critical gap in available salvage therapies, offering a viable and potent option for a patient population with otherwise limited prospects. The findings of this study underscore two pivotal points. First, the pharmacokinetic data align with global literature, confirming the rapid urinary excretion of 131I-MIBG, which inherently limits the duration of radiation hazard. Second, and crucially for clinical practice, the rigorous radiation safety monitoring provides empirical evidence that caregiver exposure can be managed effectively. The measured exposure levels, when adhered to institutional safety protocols (including distance maintenance, limited contact time, and strict hygiene measures), are well within international safety limits [7].
Conclusion
In conclusion, the high-dose 131I-MIBG therapy program at KFSHRC is not only clinically feasible but is also conducted with a high degree of safety for healthcare workers, parents, and the public. This establishes a new benchmark for specialized pediatric nuclear oncology in the Middle East and provides a replicable model for other centres aspiring to develop similar capabilities. Continued patient enrolment and long-term follow-up will further define the efficacy and refine the protocols of this critical treatment modality.
References
- Laqua R, McCarville MB (2021) Ionizing Radiation Exposure in Pediatric Patients: The Importance of Safety Protocols. Medical Physics 48(7): 3925-3934.
- Ranjan A, Smith MA (2020) The Role of 131I-MIBG in the Management of Pediatric Neuroblastoma. Journal of Nuclear Medicine 61(5): 697-704.
- van der Hiel B, van der Laan K (2020) The Pharmacokinetics of MIBG: Implications for Therapy. European Journal of Nuclear Medicine and Molecular Imaging 47(12): 2825-2834.
- Kiser SE, Hoffer MM (2020) Outcomes of High-Dose 131I-MIBG Treatment in Neuroblastoma: A Multicenter Study. Journal of Clinical Oncology 38(15 suppl): 19023-19023.
- Marzi S, Macchini M (2019) Safety and Efficacy of High-Dose 131I-MIBG Therapy in Pediatric Patients: A Review. Cancer Treatment Reviews 74: 45-54.
- Kuo HT, Wu WH (2018) Advances in Neuroblastoma Treatment: The Role of 131I-MIBG Therapy. Pediatric Blood & Cancer 65(10): e27297.
- Zanzonico P, O’Donoghue J (2017) Practical Considerations in the Administration of 131I-MIBG Therapy. Seminars in Nuclear Medicine 47(5): 462-474.
-
Mohammed Al Rowaily*. Parents and Care Givers Radiation Exposure from High Dose 131I-MIBG Therapy in Pediatrics With NBL; A KFSHRC - Saudi Arabia Initial Experience. Adv Can Res & Clinical Imag. 5(1): 2026. ACRCI.MS.ID.000604
-
131I-MIBG therapy high-dose radiopharmaceutical; radiation safety; pediatric oncology; autologous stem cell transplantation; caregiver exposure; radiation dosimetry; middle east; iris publishers; iris publisher’s group
-

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.






