 Research Article
Research Article
Head and Neck Tumors Associated with Human Papillomavirus Infection: A Literary Review Addressing Immunotherapy for Checkpoints
Gabriel Nascimento Cerqueira Rodrigues1, Igor Henrique Silva Soares1, Marco Antônio de Melo1, Pedro Tiago Felice Vilela1, Saulo Caldas Teixeira1 and Douglas Reis Abdalla1,2*
1Medicine Course, University of Uberaba, Uberaba-Minas Gerais, Brazil
2Health Courses, Faculty of Human Talents, Uberaba-Minas Gerais, Brazil
Douglas Reis Abdalla, Tonico dos Santos, 333- Jardim Induberaba Uberaba, Minas Gerais, Brazil.
Received Date: April 06, 2021; Published Date: April 22, 2021
Abstract
Introduction: The squamous cell carcinoma is the most common head and neck cancer, representing 90% of the cases. Among the risk factors, smoking and alcoholism can be highlighted, but the influence of the human papilloma virus on the development of head and neck squamous cell cancer is also evident. HPV+ have more favorable prognosis in comparison with HPV- cases. There are several treatments involving surgery, chemotherapy, radiotherapy. In addition, the associated immunotherapy has been studied to become another therapeutic alternative.
Aim: To evidence the main findings in the literature regarding the use of immunotherapy for checkpoints in head and neck tumors related to HPV infection.
Methodology: Systematic search of scientific articles published between 2010 and 2020 in the PubMed database, using the descriptors: head and neck cancer, HPV, immune system, immunotherapy. After complete analysis of the publications, six articles met all inclusion criteria.
Result: Tumors presented infiltration of myeloid and Treg suppressive cells related to tumor growth. The immunotherapy strategy associated to checkpoint inhibitors, being them anti-PD-1 and anti-CTLA-4, resulted in increased CD8+ infiltration and reduction of tumor cells, reverting the tumor growth. In addition, it led to the decrease of suppressive immune cells (MDSCs and Treg).
Conclusion: Based on the reviewed studies, head and neck neoplasms associated with human papilloma virus infection present an effective therapeutic response with immunotherapy using checkpoint inhibitors in experimental models, therefore further clinical studies involving HPV+ tumor suppression strategies with checkpoint inhibitors are needed.
Keywords: Human papilloma virus; Head and neck cancer; Immunotherapy
Introduction
The term head and neck cancer is used to refer to malignant neoplasms that originate in the regions of the upper digestive tract, such as the oral cavity, oropharynx, hypopharynx, larynx and paranasal sinuses [1-3]. Among all tumors that include the aerodigestive tract, squamous cell carcinoma (SCC) is the common one, representing 90% of all cases [2,4]. The SCC is a neoplasm that has multiple causes, resulting from the interaction of environmental and genetic factors [4]. The number of new cancer cases grows every year. For 2018/2019, the estimate of the National Cancer Institute José Alencar Gomes da Silva (INCA) is the occurrence of around 600 thousand new cancer cases in Brazil for each one of these years [5]. According to INCA, the most recent world evaluation, of 2018, points out 17 million new cancer cases in the world being 9,5 million deaths [5]. Head and neck cancer (HNC), including carcinomas of the oral cavity and oropharynx, is the sixth most common type of cancer in the world, with approximately 600,000 new cases per year and 355,000 deaths [6,7]. Brazil is one of the countries with the highest incidence of squamous head and neck carcinoma, according to INCA [6]. Oral cavity carcinoma in Brazil ranks fifth in incidence in men, presenting 5% of the total (11,200 new cases) [6].
Among the risk factors, Galbiatti, et al. [4] states that alcoholism and smoking have an additive factor and are the main factors associated with the development of lesions. However, there are other factors that also influence the development of head and neck SCC, such as HPV infections, diet, exposure to carcinogens, oral health conditions, infectious agents, family history, nutritional deficiencies, exposure to ultraviolet light, chronic irritation of the mouth lining and formation of dental plaque, pre-existing pathologies and demographic variations. According to Krüger et al. [10], the diagnosis of head and neck cancer is made through biopsy under anesthesia for anatopathological analysis of the suspected lesion. In case of neoplastic evidence, the tumor staging should be performed through imaging and complementary exams; to evaluate the possibilities of prognosis and treatment. The treatment for head and neck cancers are varied, but the main ones are radiotherapy exclusive or combined with surgery, surgery exclusive or radiotherapy combined with chemotherapy. However, these methods are very aggressive for the patient11. According to Krüger et al [10], the morbidity associated with each treatment approach is an important factor in the decision making of this one. In addition, oropharyngeal squamous cell carcinoma can be treated with primary surgery or with definitive radiation therapy (RT) as a single modality [11], but according to University of Pennsylvania data [12], patients treated in this way have loco-regional recurrence (27%) and distance metastasis (31%). In addition, about 60% have an unstable restoration [13], and may also have xerostomia, odinophagia, weight loss and the need for alternative feeding pathways [14]. On the other hand, the use of chemotherapy associated with radiotherapy presents good results in survival, recurrence and cancer control [15], besides, the effect is even greater when there is the substitution of cisplatin by cetuximab, in chemotherapy treatment, since this substitution causes less toxicity in the patient [16]. In the case of HPV positive head and neck cancer, the treatment is the same as in other carcinomas, but has a better prognosis because it has a more favorable epidemiology, since they are younger patients with fewer risk factors [16]. Therefore, less aggressive alternatives are sought, with lower morbidity, fewer sequelae and consequently better quality of life. Then, evidence shows that cisplatin replacement by cetuximab, in chemotherapy (as previously mentioned), in addition to reducing the dose in radiotherapy and also the use of minimally invasive transoral approaches (both in transoral laser microsurgery-TLM and in transoral robotic surgery-TORS), proved promising in the removal and treatment of these tumors [17,18]. According to Farkona and Collaborators [8]. immunotherapies are designed to increase the activity of the immune system to eradicate cancerous cells. These include vaccines using tumor peptide antigens or vectors based on viruses, bacteria and DNA, as well as monoclonal antibodies with specific tumor antigens (moAbs). In addition, immunological checkpoint inhibitors (ICIs) are a widely effective class of immunotherapies that block the pathways of inhibitory immune control points in order to reactivate immune responses against head and neck cancer. These include anti-CTLA-4 and anti-PD-1 [9]. Thus, the present work aims to evidence the main findings in the literature regarding the use of immunotherapy for checkpoints in head and neck tumors related to HPV infection.
Methodology
In the present study, an integrative review was conducted, which consists of a research that allows from evidence the evaluation, synthesis and knowledge about a phenomenon, aiming to produce an overview of complex concepts, theories or relevant health problems from pre-existing studies, enabling the proposition of intervention. [19,20] For the selection of articles, 6 methodological steps were conducted, which are: 1. elaboration of the guiding question or hypothesis of the research, that is, the problem was identified, the search engine and the descriptors or key words were presented; 2. establishment of the inclusion and exclusion criteria of the articles to be selected for sample composition; 3. exploratory reading of the titles and abstracts of the articles for pre-selection; 4. analytical reading of the articles in order to compile, analyze and categorize the information; 5. interpretation of the results. 6. synthesis followed by presentation of the results identified, which permeate the guiding question. [21] Therefore, in this study we chose to perform a search on the concepts: head and neck cancer, HPV, immune system and immunotherapy. Based on these concepts, the guiding question was defined: What are the main findings in the literature regarding the use of immunotherapy for checkpoints in head and neck tumors related to HPV infection.
After formulating the question to be researched, a bibliographic survey was conducted on the PubMed platform. The survey of the study took place between September and October 2020. And the selection of texts proceeded with the searches in the platform, using the filters available for texts published between 2010 and 2020. For the selection of publications, the following inclusion criteria were adopted: scientific articles, published in English, between 2010 and 2020, available online and free of charge in full. Articles without abstract in the database or incomplete, editorials, letters to the editor, reflective studies, systematic or integrative literature reviews were excluded. After defining the guiding issue, location and selection of articles, 81 publications were identified as potentially eligible to be included in this review. After applying the inclusion and exclusion criteria, the sample consisted of 73 publications, the abstracts of 37 records were analyzed to verify if they would meet the eligibility criteria and if they would answer the question that guides this review. Thus, 31 records were excluded and only 6 were analyzed in full to confirm eligibility for quantitative synthesis and data analysis according to the selection flowchart (Figure 1).
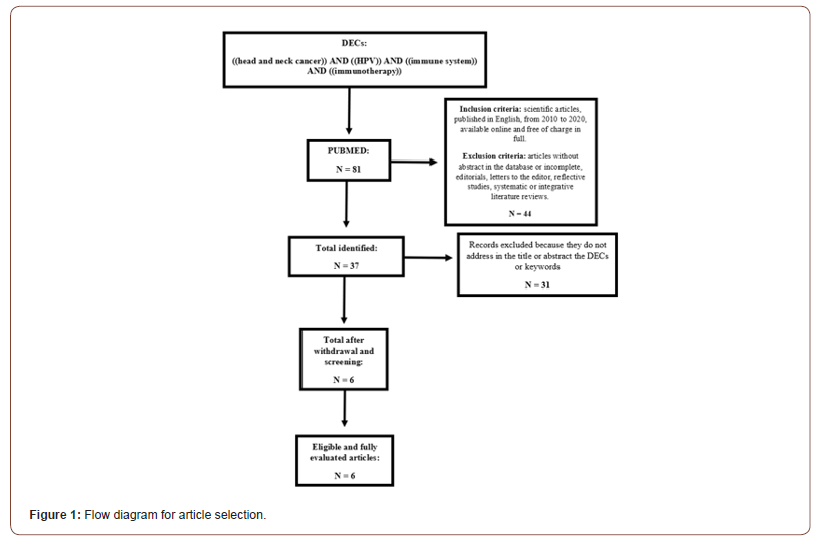
Result and Discussion
In the time frame set for this study (2010-2020) 6 publications were found and analyzed. In 2017 and 2019 2 articles (66.7%) were published each year respectively, already in 2015 and 2018 there is only 1 publication in each year (33.3%). According to the methodology of the selected papers the types of study were pre-clinical research using mice and cell lines of dependent HPV tumors. The publications resulted from different magazines being: Cancer Research, Clinical Cancer Research, JCI Insight, Journal for ImmunoTherapy of Cancer and Oncotarget. Analyzing the study sites 5 articles were conducted in the United States (83.3%) and the other article developed in China (16.7%). According to Aggarwal; Cohen; Morrow et al (2019)22, the development of head and neck squamous cell carcinomas (HNSCCa), represent significant numbers when associated with human papillomavirus (HPV). Checkpoint points assist in the creation of an immunoprivileged site for HPV infection, thus, the tumor develops adaptive resistance against the host immune system. There are already immunotherapies with checkpoint inhibitors, which bring temporary secondary benefits, however only a portion of patients (15 to 20%) remain with survival gain and lasting benefits. The status of the tumor’s HPV does not seem to be the main responsible for the result with this new therapy. Combination strategies of immunotherapies can be applied in cases of patients who respond well to PD-1 inhibition. And an interesting immunological approach is to target “non-own” antigens for HPV that will activate the immune system in a more potentiated way [22]. According to information from the articles analyzed, in cases of HNSCCa associated with HPV infection the prognosis is more favorable in comparison with cases without HPV infection [23-27]. Among the existing subtypes, HPV16 constitutes the majority of cases of HNSCCa [22,25].
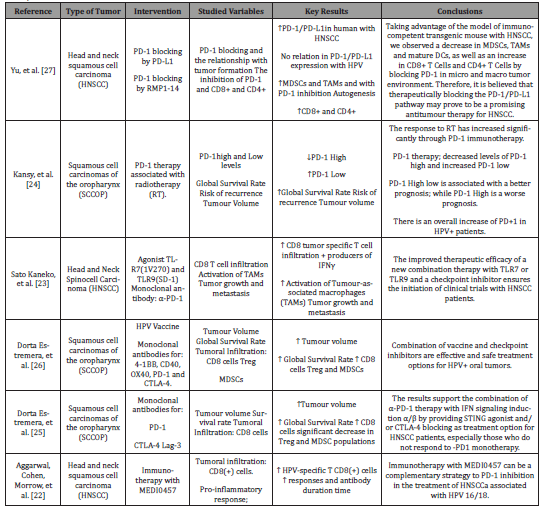
According to Yu; Bu; Huang, et al. [27], a significant increase in PD-1 / PD-L1 was observed in HNSCCa, which is closely correlated with important immunosuppressive cells, myeloid origin suppressive cells (MDSCs) and tumor associated macrophages (TAMs). The authors took advantage of the immunocompetent transgenic mouse model with HNSCCa and identified a decrease in MDSCs, TAMs and mature Dendritic Cells (DCDs), as well as an increase in CD8+ T cells and CD4+ T cells by blocking PD-1 in micro and macro tumor environment. Moreover, according to Kansy et al. [24]; the expression of PD-1low was significantly higher in HPV+ than in samples of HPV- which had in turn more PD-1high. The predominance of PD-1high cells was associated with worse survival, larger tumor size and increased chance of tumor recurrence, while the predominance of PD-1high had better prognosis in the factors mentioned above. Moreover, it was shown through this study that the use of radiotherapy associated with anti-PD-1 immunotherapy showed better results in the decrease of tumors, increased PD-1low and decreased PD-1high; presenting, therefore, better results than radiotherapy alone.
In addition, Sato-Kaneko; Yao; Ahmadi et al. [23], in their combined therapy of TLR (Toll-like sReceptor) agonists with checkpoint inhibitors produced activation of tumor associated macrophages (TAMs) and induced tumor-specific adaptive immune responses, with infiltration of tumor-specific CD8 T cells and producing IFN-γ. In addition, they led to suppression of primary tumor growth and prevention of metastases. In a study conducted by Dorta-Estremera and Cols [25], monotherapies were made in mice with anti-PD-1, anti-CTLA-4 and anti-Lag 3, not presenting satisfactory results, but, on the other hand, the association of anti-PD-1 with anti-CTLA-4 or anti-PD-1 with anti-Lag3, showed an increase in overall survival and infiltration of TCD8+ cells, in addition to a significant decrease in immunosuppressive populations, such as Treg and MDSCs, and tumor volume compared to other control groups and those that were made other combinations of immunotherapies. Another study conducted by Dorta-Estremera and Cols [26], used an experimental model with C57BL/6 mice, with mEER inoculation in the nasal cavity, and thus, performed therapeutic groups in monotherapy or polytherapy, with HPV vaccine (inhalation), monoclonal antibodies for 4-1BB, CD40, OX40, PD-1 and CTLA-4 and the combination of the intranasal HPV vaccine and the antibodies α-4-1BB and α-CTLA-4 were shown to produce a curative efficacy and improved safety profile against orally implanted tumors and larger CD8+ T-cell tumor infiltrate and smaller Treg and MDSC cell infiltrates. Thus, they concluded that the combination of vaccine and checkpoint inhibitors are effective and safe treatment options for HPV+ oral tumors.
Finally, Aggarwal et al [22], sought combination strategies of immunotherapies that can be applied in cases of patients who respond well to inhibition of PD-1, thus, verified about the inhibition of HPV 16/18 proteins E6 and E7 oncogens in being the complementary immunotherapy strategy to the inhibition of PD- 1, and was found increase in the number of specific CD8+ T cells for HPV. Thus, Figure 2 summarizes the findings of the literature in which in the left part, we observe a tumor focus infected by HPV, presenting a predominant infiltrate of Treg cells and MDSCs and in a lower proportion of CD8+ T cells. In the image on the right this same tumor focus in detriment of anti-PD-1 and Anti-CTLA-4 therapy, it is possible to verify that the inhibition of the checkpoints facilitates the recognition of the tumor cell by CD8+ T cells, by means of MHC-I and TCR molecules respectively; resulting in a lytic pathway that causes the destruction of the tumor cell (Figure 2).
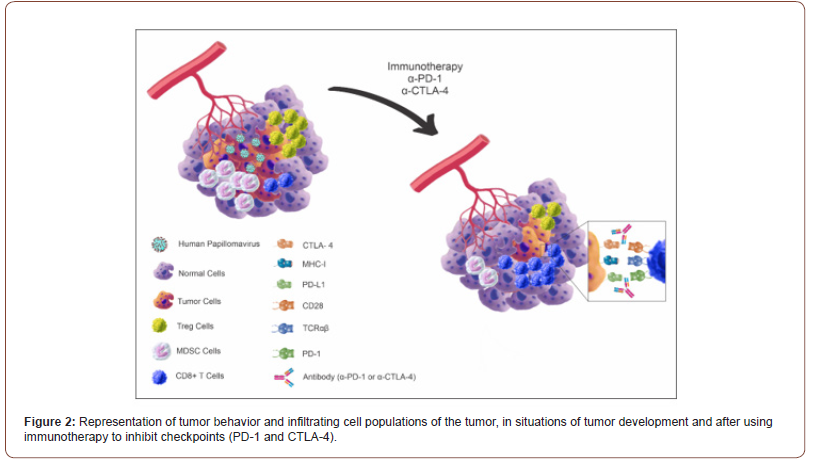
Table 1: List of articles that investigated in experimental models of head and neck tumors the influence of immunotherapy with immunological checkpoint inhibitors.
Conclusion
The study of HPV+ related head and neck tumors has become possible in experimental models. The cancer presented an infiltration of suppressive immune cells (MDSCs and Treg) related to HPV infection. Immunotherapy, anti-PD1 and CTLA- 4, predominantly reverted the tumor growth, due to the larger infiltration of CD8+ T lymphocytes and decrease of MDSCs and Treg. Therefore, the development of clinical studies is necessary for a more comprehensive knowledge about the use of checkpoint inhibitors.
Acknowledgement
None.
Conflict of Interest
No Conflict of Interest.
References
- Mouth and Throat Cancer Association (2018) Types and Cancer: All about head and neck cancer.
- Keys ALF, Oliveira TB, Santos M, Soares JMA, Kowalski LP (2017) Guidelines for Head and Neck Cancer of the Brazilian Society of Clinical Oncology.
- Keys ALF, Santos M, Quintanilha MA, Barreira CESR (2019) Cancer guidelines 2: Head and Neck Cancer. São Paulo: Doctor Press Ed Scientific Cap 4: 53-70.
- Galbiatti ALS, Padovani JA, Maníglia JV, Rodrigues CDS, Pavarino EC, et al. (2013) Head and neck cancer: causes, prevention and treatment. Braz J otorhinolaryngol 79(2): 239-247.
- Ministry of Health (2020) National Cancer Institute. Estimate: Incidence of cancer in Brazil, Rio de Janeiro: INCA, Brazil.
- Ministry of Health (2020) National Cancer Institute. Cancer statistics, Rio de Janeiro: INCA, Brazil.
- Petito G, Carneiro MAS, Santos Sílvia HR, Silva AMTC, Alencar RC, et al (2017) Human papillomavirus (HPV) in oral cavity and oropharynx carcinomas in central Brazil. Braz J otorhinolaryngol 83(1): 38-44.
- Farkona S, Diamandis EP, Blasutig IM (2016) Cancer immunotherapy: the beginning of the end of cancer? BMC Med 14: 73.
- Ferris RL (2015) Immunology and Immunotherapy of Head and Neck Cancer. J Clin Oncol 33(29): 3293-3304.
- Krüger CP, Poletto E, Souza GACD, Azevedo RSD, Steffen N (2017) Neoplasms of oropharynx. Minutes méd (Porto Alegre) 7-7.
- Kowalski LP (2000) Head and neck cancer In: Carrara-de Angelis E, Furia CLB, Mourão LF, Kowalski LP. The performance of speech therapy in head and neck cancer. São Paulo: Lovise, Brazil, p.23.
- Machtay M, Rosenthal DI, Hershock D, Jones H, Williamson S, et al. (2002) Organ preservation therapy using induction plus concurrent chemoradiation for advanced resectable oropharyngeal carcinoma: a University of Pennsylvania Phase II Trial. J Clin Oncol 20(19): 3964-3971.
- Denittis AS, Machtay M, Rosenthal DI, Sanfilippo NJ, Lee JH, et al. (2001). Advanced oropharyngeal carcinoma treated with surgery and radiotherapy: oncologic outcome and functional assessment. Am J Otolaryngol 22(5): 329-335.
- Novaes PERS (2000) Radiotherapy in head and neck neoplasms. In: Carrara-de Angelis E, Furia CLB, Mourão LF, Kowalski, LP. The performance of speech therapy in head and neck cancer. São Paulo: Lovise; Brazil, p: 33-41.
- Machtay M, Rosenthal DI, Hershock D, Jones H, Williamson S, et al. (2002) Organ preservation therapy using induction plus concurrent chemoradiation for advanced resectable oropharyngeal carcinoma: a University of Pennsylvania Phase II Trial. J Clin Oncol 20(19): 3964-3971.
- Mirghani H, Blanchard P (2018) Treatment de-escalation for HPV-driven oropharyngeal cancer: Where do we stand? CT Radiation Oncology 8: 4-11.
- Smith RV, Schiff BA, Garg M, Haigentz M (2015) The Impact of Transoral Robotic Surgery on the Overall Treatment of Oropharyngeal Cancer Patients. Laryngoscope 125(10): S1-S15
- Kelly K, Johnson-Obaseki S, Lumingu J, Corsten M (2014) Oncologic, functional and surgical outcomes of primary Transoral Robotic Surgery for early squamous cell cancer of the oropharynx: A systematic review. Oral Oncology 50(8): 696–703.
- Whittemore R, Knafl K (2005) The integrative review: updated methodology. J Adv Nurs 52(5): 546-553.
- Galvão CM, Sawada NO, Trevizan MA (2004) Systematic review: a resource that allows the incorporation of evidence into nursing practice. Rev Latino Am Enfermagem 12(3): 549-556.
- De Sousa LD, Lunardi Filho WD, Lunardi VL, Santos SS, Dos Santos CP (2011) The scientific nursing production about the clinic: an integrativa review. Rev Esc Enferm USP 45(2): 494-500.
- Aggarwal C, Cohen RB, Morrow MP, Kraynyak KA, Sylvester AJ, et al. (2019) Immunotherapy targeting HPV16/18 generates potent immune responses in HPV-associated head and neck cancer. Clinical Cancer Res 25(1): 110-124.
- Sato-Kaneko F, Yao S, Ahmadi A, Zhang SS, Hosoya T, et al. (2017) Combination immunotherapy with TLR agonists and checkpoint inhibitors suppresses head and neck cancer. JCI insight 2(18).
- Kansy BA, Concha Benavente F, Srivastava RM, Jie HB, Shayan G, et al. (2017) PD-1 status in CD8+ T cells associates with survival and anti-PD-1 therapeutic outcomes in head and neck cancer. Cancer Res 77(22): 6353-6364.
- Dorta Estremera S, Hegde VL, Slay RB, Sun R, Yanamandra AV, et al. (2019) Targeting interferon signaling and CTLA-4 enhance the therapeutic efficacy of anti-PD-1 immunotherapy in preclinical model of HPV+ oral cancer. J Immunother Cancer 7(1): 252.
- Dorta Estremera S, Chin, RL, Sierra G, Nicholas C, Yanamandra AV, et al. (2018) Mucosal HPV E6/E7 peptide vaccination in combination with immune checkpoint modulation induces regression of HPV+ oral cancers. Cancer Res 78(18): 5327-5339.
- Yu GT, Bu LL, Huang CF, Zhang WF, Chen WJ, et al. (2015) PD-1 blockade attenuates immunosuppressive myeloid cells due to inhibition of CD47/SIRPα axis in HPV negative head and neck squamous cell carcinoma. Oncotarget 6(39): 42067-42080.
-
Marco Antônio de Melo, Pedro Tiago Felice Vilela, Saulo Caldas Teixeira, Douglas Reis Abdalla. Head and Neck Tumors Associated with Human Papillomavirus Infection: A Literary Review Addressing Immunotherapy for Checkpoints. Adv Can Res & Clinical Imag. 3(2): 2021. ACRCI.MS.ID.000559.
-
Cancer, Human papilloma virus, Head and neck cancer, Immunotherapy, Malignant neoplasms, Tumor
-

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.






