 Research Article
Research Article
A Novel Theranostic Hypothesis in Ga-68 HCl for SARS-CoV-2
Mohammed Al Rowaily*1,2,3
1 Radiology Department, King Faisal Specialist Hospital & Research Centre, Riyadh, Saudi Arabia
2 Alfaisal University, 11533 Riyadh, Saudi Arabia
3 KSU, 11433 Riyadh, Saudi Arabia
Mohammed Al Rowaily, Radiology Department, King Faisal Specialist Hospital & Research Centre, Alfaisal University, KSU, Riyadh, Saudi Arabia
Received Date:February 26, 2026; Published Date:March 20, 2026
Abstract
Purpose: This study proposes a novel theranostic approach using Gallium-68 chloride (⁶⁸GaCl₃) in hydrochloric acid (HCl) as a potential
treatment for SARS-CoV-2, aiming to explore viral inactivation through targeted delivery in acidic environments.
Procedures: Drawing from existing literature on SARS-CoV-2 susceptibility to acidic environments and the chemical properties of Gallium-68,
a hypothesis was formulated. A research proposal was created to administer 5 mL of ⁶⁸Ga/0.1 M HCl to COVID-19 patients for five days, using PET
imaging for verification of targeting.
Results: A pilot investigation was conducted whereby two patients with moderate pneumonia received a single intravenous dose of 5 mL of
⁶⁸Ga-DOTATATE in a 0.1 M HCl medium. PET/CT imaging showed notable accumulation of the ⁶⁸Ga/HCl complex in the lungs, specifically in areas
of inflammation, confirming successful delivery to viral replication sites.
Conclusions: The pilot study supports the viability of the theranostic approach in targeting SARS-CoV-2. This narrative emphasizes the
importance of innovative thinking during health crises and highlights the potential of repurposing diagnostic radionuclides for therapeutic
applications against emerging viral threats.
Keywords:SARS-CoV-2; COVID-19; gallium-68; theranostic; viral inactivation; hydrochloric acid; pandemic response
Abbreviations: ACE2: Angiotensin-Converting Enzyme 2; COVID-19: Coronavirus Disease 2019; CT: Computed Tomography; Ga: Gallium; Ga³⁺: Gallium ion (trivalent); HCl: Hydrochloric Acid; M: Molar (concentration); NETs: Neuroendocrine Tumors; PET: Positron Emission Tomography; SARS-CoV-2: Severe Acute Respiratory Syndrome Coronavirus 2; TMPRSS2: Transmembrane Serine Protease 2; DOTATATE: DOTA-(Tyr3)- octreotate
The Genesis of an Idea: March 2020
In March 2020, the world held its breath. Hospitals were becoming overwhelmed, and the global scientific apparatus scram bled to understand the novel SARS-CoV-2 virus. An internal hospital email, sent to all staff, called for immediate and innovative ideas for treatments. This was a moment that demanded thinking “outside the box.” Drawing on experience in nuclear medicine and radiochemistry, a potential solution emerged from first principles. The literature was clear: the lipid envelope of viruses like SARS-CoV-2 is a critical point of vulnerability, with infectivity plummeting by over 95% after brief exposure to a pH of 4.0 [1,2]. Simultaneously, Gallium-68, a radionuclide with a well-established safety profile in diagnostic PET imaging, exists as a stable trivalent cation (Ga³⁺) precisely in low-pH, high-chloride environments [3]. Its chemical behavior and known affinity for inflamed tissues [4] suggested a unique opportunity.
The hypothesis was straightforward: Could a solution of ⁶⁸GaCl₃ in 0.1 M HCl serve as a targeted vehicle to deliver a highly acidic microenvironment directly to the viral replication sites in the lungs? This concept was formalized in a research proposal titled “Covid-19 Treatment Using Gallium-68 chloride.” The proposal outlined a study administering 5 mL of ⁶⁸Ga/0.1 M HCl daily for five days to COVID-19 patients, using PET imaging to verify targeting. The core idea was theranostic, using the radionuclide’s diagnostic capability to confirm its delivery to the therapeutic target. However, in the panic and uncertainty of the early pandemic, proposing an intravenous solution of hydrochloric acid, even in a low molarity and complexed form, was a significant conceptual hurdle. The idea, deemed too unconventional at the time, was set aside [5].
From Hypothesis to Preliminary Evidence: A Pilot Investigation
The intuitive leap of March 2020 was not forgotten. To test the fundamental premise of targeted delivery, a pilot study was later initiated. As detailed in “A Pilot Investigation into Gallium-68 as a Therapeutic Agent for SARS-CoV-2 Viral,” two confirmed COVID-19 patients with moderate pneumonia were administered a single intravenous dose of 5 mL of ⁶⁸Ga-DOTATATE in a 0.1 M HCl medium. The results were striking. PET/CT imaging revealed significant and specific accumulation of the ⁶⁸Ga/HCl complex within the pulmonary parenchyma, precisely co-localizing with regions of groundglass opacities and inflammation (Figures 1&2). This provided the first-in-human evidence that the cationic Ga³⁺ could indeed act as a vehicle, successfully delivering its acidic payload to the sites of active SARS-CoV-2 infection. The biodistribution outside the lungs was consistent with the known physiology of Ga³⁺, and no acute adverse effects were reported, underscoring the procedural safety of the approach.
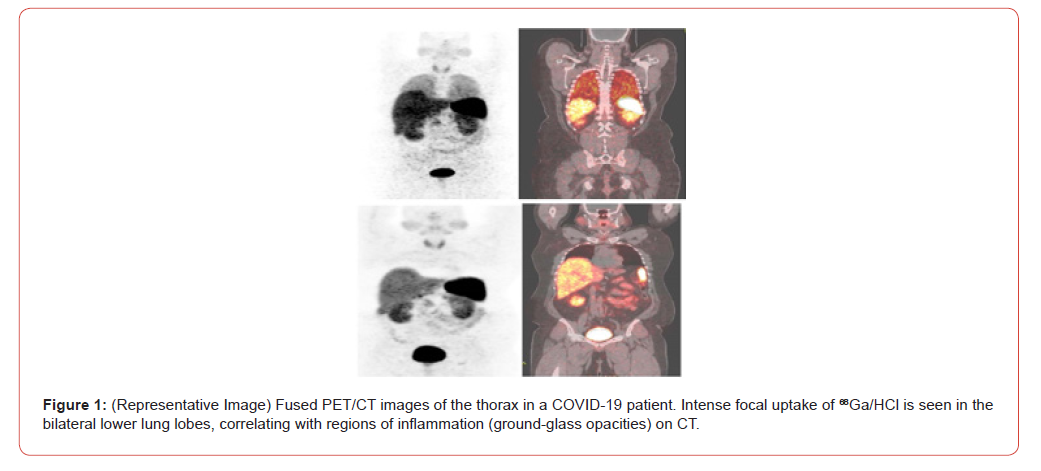
Discussion: Revisiting a Missed Opportunity?
This narrative, from initial rejection to preliminary validation, highlights both the challenges and the necessity of innovative thinking during a public health crisis. The pilot study confirmed the central tenet of the original hypothesis: targeted delivery is achievable.
Proposed Mechanism of Action: The Proposed Virucidal Mechanism is Two-Fold
a) Targeted Delivery: Inflamed tissues exhibit enhanced permeability and contain negatively charged cellular debris. The positively charged Ga³⁺ ion is extravasated and bound at these sites, co-localizing the acidic solution (pH ~1.1) with the virus.
b) Viral Inactivation: The localized, potent acidic environment disrupts the integrity of the SARS-CoV-2 lipid envelope and denatures the spike (S) glycoproteins, which are essential for host cell entry, thereby rendering the virus non-infectious.
The theranostic advantage of ⁶⁸Ga is profound. It allows for real- time, non-invasive confirmation that the proposed therapeutic agent has reached its intended target, a critical step before assessing efficacy in larger trials.
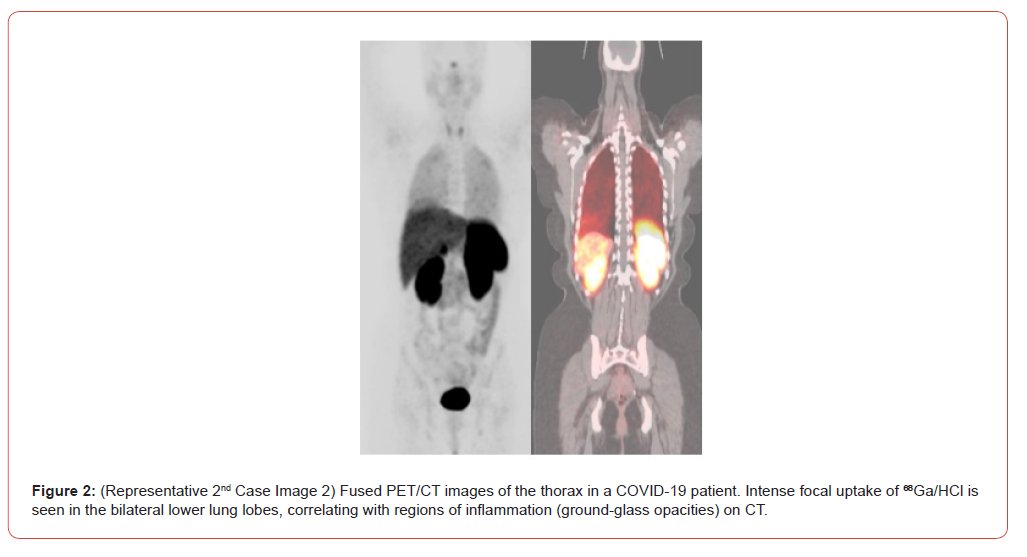
Limitations and Future Directions
The pilot study was, by design, a proof-of-concept with a very
small sample size (n=2). It did not measure the direct virological
impact, such as a reduction in viral load. The safety of repeated
doses requires rigorous investigation. The path forward, as initially
envisioned and now supported by this data, must include:
a) In vitro studies to quantitatively measure the virucidal effect of
⁶⁸Ga/HCl on live SARS-CoV-2.
b) Preclinical in vivo studies in animal models to assess efficacy
in reducing viral load and establishing a detailed safety profile.
c) Larger, controlled clinical trials to evaluate therapeutic benefit
and optimal dosing.
Conclusion
The COVID-19 pandemic taught the world a harsh lesson in preparedness. This story underscores an equally important lesson: the value of fostering and investigating unconventional ideas, even - or especially - during a crisis. The proposal to use Gallium-68/ HCl for SARS-CoV-2, born from a direct call for help in March 2020, was a theranostic hypothesis ahead of its time. While the world moved on to other solutions, the subsequent pilot data suggests the idea held merit. Repurposing well-understood diagnostic radionuclides as targeted therapeutic vehicles remains a promising, yet underexplored, avenue in our ongoing fight against current and future enveloped viral pathogens.
References
- Kohn A (1979) Early interactions of viruses with cellular membranes. Adv Virus Res 24: 223-276.
- Ye K, Harpreet K Dhiman, Joseph Suhan, Jerome S Schultz (2003) Effect of pH on Infectivity and Morphology of Ecotropic Moloney Murine Leukemia Virus. Biotechnol Prog 19(2): 538-543.
- Maecke HR, André JP (2007) 68Ga-PET Radiopharmacy: A Generator-Based Alternative to 18F-Radiopharmacy. In Radiochemical Synthesis for Positron Emission Tomography 62: 215-242.
- Weiner RE (1996) The mechanism of 67Ga localization in malignant disease. Nuclear Medicine and Biology 23(6): 745-751.
- Hoffmann M, Hannah Kleine-Weber, Simon Schroeder, Nadine Krüger, Tanja Herrler, et al. (2020). SARS-CoV-2 Cell Entry Depends on ACE2 and TMPRSS2 and Is Blocked by a Clinically Proven Protease Inhibitor. Cell 181(2): 271-280.e8.
-
Mohammed Al Rowaily*. A Novel Theranostic Hypothesis in Ga-68 HCl for SARS-CoV-2. Adv Can Res & Clinical Imag. 5(2): 2026. ACRCI.MS.ID.000605.
-
SARS-CoV-2; COVID-19; gallium-68; theragnostic; viral inactivation; hydrochloric acid; pandemic response; iris publishers; iris publisher’s group
-

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.






