 Case Report
Case Report
Intrasellar Hemangioblastoma in Von Hippel-Lindau Disease Treated With HIF-2α Inhibitor
Natacha Driessens1, France Devuyst1, Chiara Mabiglia2, Christiane Jungels3, Myriam Delhaye4, Florence Lefranc5*
1Department of Endocrinology, Brussels, Belgium
2Department of Radiology, Brussels, Belgium
3Department of Oncology, Brussels, Belgium
4Department of Gastroenterology, Brussels, Belgium
5Department of Neurosurgery, Université Libre de Bruxelles (ULB), Hôpital Universitaire de Bruxelles (H.U.B), Brussels, Belgium
Florence Lefranc, Department of Neurosurgery, Université Libre de Bruxelles (ULB), Hôpital Universitaire de Bruxelles (H.U.B), Brussels, Belgium
Received Date: April 8, 2026; Published Date: April 21, 2026
Introduction
Von Hippel-Lindau (VHL) disease is a rare autosomal dominant inherited syndrome due to a mutation in the tumor suppressor gene VHL on chromosome 3p, which results in the stabilization of hypoxia-inducible factor (HIF)-1α and -2α [1]. VHL disease therefore predisposes patients to the development of highly vascularized tumors such as renal cell carcinomas, paragangliomas, pancreatic neuroendocrine tumors and the most common ones hemangioblastomas of the retina, the brain and the spinal cord. Central Nervous System (CNS) hemangioblastomas, the most common cause of mortality [2,3], occur in up to 80% of patients with VHL disease throughout their lifetime, with up to 40% of cases having hemangioblastomas as their presenting features [4].
The standard treatment for CNS hemangioblastomas is neurosurgical resection frequently associated with highrisk depending on the location of the lesion [5]. Stereotactic radiosurgery is an option for difficult-to-resect tumors [6,7]. Recently, belzutifan (Welireg), a small molecule first-in-class HIF- 2α inhibitor, has demonstrated benefit in VHL-associated tumors [8] including disease-associated CNS hemangioblastomas [9,10]. On August, 2021, the Food and Drug Administration approved belzutifan for adult patients with VHL disease who require therapy for associated renal cell carcinoma, CNS hemangioblastomas, or pancreatic neuroendocrine tumors, and for whom localized procedures are unsuitable [8].
Thirty percent of patients with CNS hemangioblastomas (n=50) who received belzutifan responded to the treatment, the hemangioblastomas decreased by 30%–50%, and 6% of patients had complete response [9].
We report, to our knowledge, the first case of a non-invasive therapeutic approach using belzutifan for a hemangioblastoma located in the intrasellar region.
Case Report
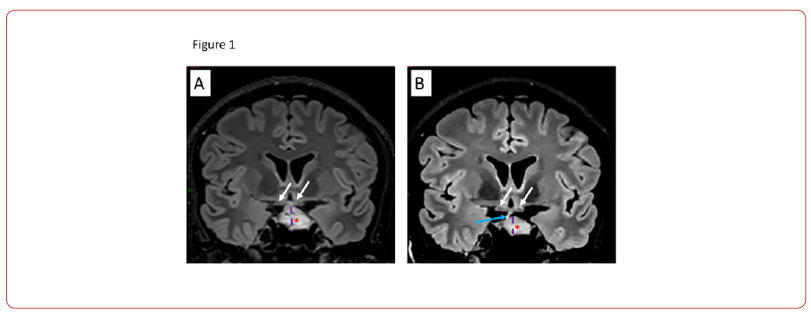
A 27-year-old man with VHL disease, previously treated with neurosurgery and radiosurgery for symptomatic cerebral and spinal cord hemangioblastomas, presented with a slowly growing asymptomatic intrasellar hemangioblastoma (typical vascular pedicle) dangerously reaching the optic nerfs (white arrows in Figure 1A and Figure 1B), thereby threatening his vision. Given the high risk of complications associated with microneurosurgery and/or radiosurgery for this asymptomatic lesion, treatment with hypoxia-inducible factor-2-alpha (HIF-2α) inhibitor was initiated.
Belzutifan was provided by MERCK-MSD through a compassionate use program (Belgium, ref CUP-202422). Therapy (orally at a dose of 120mg daily) was well tolerated. Magnetic Resonance Imaging (MRI) performed one month after the treatment initiation revealed significant shrinkage of the lesion (19mm (in Figure 1A) to 15mm (in Figure 1B); dotted lines).
Conclusion
This case highlights belzutifan as a novel targeted therapy for neurosurgically challenging progressive hemangioblastomas even without symptoms in patients with VHL disease, offering a noninvasive treatment alternative.
Conflict of Interest
We declare no competing interest.
References
- Latif F, Tory K, Gnarra J, Yao M, Duh FM, Orcutt ML, et al. (1993) Identification of the von Hippel-Lindau disease tumor suppressor gene. Science 260(5112): 1317-20.
- Binderup ML, Jensen AM, Budtz-Jørgensen E, Bisgaard ML (2017) Survival and causes of death in patients with von Hippel-Lindau disease. J Med Genet 54(1): 11-18.
- Zhou B, Wang J, Liu S, Peng X, Hong B, Zhou J, et al. (2019) Hemangioblastoma Instead of Renal Cell Carcinoma Plays a Major Role in the Unfavorable Overall Survival of Von Hippel-Lindau Disease Patients. Front Oncol 9: 1037.
- Maher ER, Yates JR, Harries R, Benjamin C, Harris R, (1990) Clinical features and natural history of von Hippel-Lindau disease. Q J Med 77(283): 1151-1163.
- Hodgson TS, Nielsen SM, Lesniak MS, Lukas RV (2016) Neurological Management of Von Hippel-Lindau Disease. Neurologist 21(5): 73-78.
- Pan J, Jabarkheel R, Huang Y, Ho A, Chang SD (2018) Stereotactic radiosurgery for central nervous system hemangioblastoma: systematic review and meta-analysis. J Neurooncol 137(1): 11-22.
- Qiu J, Cai D, Yang F, Zhou J, Gong Y, et al. (2020) Stereotactic radiosurgery for central nervous system hemangioblastoma in von Hippel-Lindau disease: A systematic review and meta-analysis. Clin Neurol Neurosurg 195: 105912.
- Fallah J, Brave MH, Weinstock C, Mehta GU, Bradford D, Gittleman H, et al. (2022) FDA approval summary: belzutifan for von Hippel-Lindau disease associated tumors. Clin Cancer Res 28: 4843-4848.
- Jonasch E, Donskov F, Iliopoulos O, Rathmell WK, Narayan VK, (2021) MK-6482-004 Investigators. Belzutifan for Renal Cell Carcinoma in von Hippel-Lindau Disease. N Engl J Med 385(22): 2036-2046.
- Iliopoulos O, Iversen AB, Narayan V, Maughan BL, Beckermann KE, et al. (2024) Belzutifan for patients with von Hippel-Lindau disease-associated CNS haemangioblastomas (LITESPARK-004): a multicentre, single-arm, phase 2 study. Lancet Oncol 25(10): 1325-1336.
-
Natacha Driessens, France Devuyst, Chiara Mabiglia, Christiane Jungels, Myriam Delhaye, Florence Lefranc*. Contrast-Intrasellar Hemangioblastoma in Von Hippel-Lindau Disease Treated With HIF-2α Inhibitor. Adv Can Res & Clinical Imag. 5(1): 2026. ACRCI.MS.ID.000608.
-
HIF-2α Inhibitor; Central Nervous System; Hemangioblastomas
-

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.






