 Case Report
Case Report
De-Escalation Treatment (DET) as a Personalized Pharmacological Intervention in Two Airline Pilots with Type 2 Diabetes Mellitus
Andreas Pfützner1,2*, Julia Jantz1, and Dirk M Rose3
1 Pfützner Science & Health Institute, Mainz, Germany
2 Department of Internal Medicine & Laboratory Medicine, University for Digital Technologies in Medicine & Dentistry, Wiltz, Luxembourg
3 FlyMed Medical Aviation Center, Frankfurt Airport, Frankfurt, Germany
Andreas Pfützner, Pfützner Science & Health Institute, Mainz, Germany
Received Date: June 25, 2025; Published Date: July 01, 2025
Abstract
Background: Personalized multi-pharmacological approaches are being developed to improve blood glucose control in patients with type 2
diabetes mellitus as an alternative to the drug escalation programs that are suggested by current treatment guidelines. De-Escalation Treatment
(DET) is a temporary, multi-drug intervention to target and improve pancreatic beta-cell function. It includes administration for three-month of a
personalized drug combination determined based on a novel biomarker panel. This report is about the results with two airline pilots with type 2
diabetes mellitus who underwent DET.
Case 1: A 48-year-old airline pilot with progressive type 2 diabetes (on metformin and glimepiride treatment) showed further impairment of
glycaemic control, and was escalated to insulin treatment. This would have terminated his professional career. He was treated with DET composed
of four anti-diabetic drugs, selected based on his biomarker profile. After 3 months, DET was stopped, and an oral glucose challenge indicated no
remaining prevalence of diabetes. Subsequently, diabetes management was successfully continued with lifestyle measures only. When diabetes
started to return after 2 years, as shown by ß-cell biomarkers, DET was repeated with similar success. Over the next eight years, DET was repeated
four times. The patient always had stable glucose control (HbA1c < 6 %).
Case 2: A second patient case was treated also with DET and with similar result.
Conclusions: DET was employed to restore and maintain airworthiness of two airline pilots for meanwhile up to eight years. Appropriately
designed prospective randomised clinical studies are required to confirm the applicability of DET in larger patient populations.
Keywords: Type 2 diabetes; remission phase; airline pilots; ß-cell regeneration; de-escalation therapy
Introduction
According to international air traffic regulations, professional airline pilots have to pass medical examinations on a regular basis to keep their license [1]. Commercial pilots working in the passenger transportation business require a stringent class 1 medical approval, and theias a general treatment alternative r professional career is dependent on successfully passing these procedures. It has been observed that pilots therefore generally have a better health status as compared to the overall population, which is certainly supported by their high socio-economic standards [2]. In addition, physicians performing these medical examinations usually also consider more frequently additional preventive and labour-related aspects [3]. The future working perspective of pilots, who are developing type 2 diabetes, is dependent on the actually performed therapy, with particular focus on the potential risk for hypoglycemia, and the resulting metabolic control. Pilots who can successfully control their metabolic condition by lifestyle (i.e. diet and exercise) only, have no flight restrictions in Europe and in the United States [1,4].
In case of oral anti-diabetic treatment, an international evidence-based risk evaluation is employed [5], and the responsible air traffic control agency has to approve the flight permit on an individual basis. Acceptance criteria are a good glycemic control (hemoglobin A1c (HbA1c) < 7.5 %) achieved by oral drugs with low risk of hypoglycemia, and regularly performed and documented self-measurement of blood glucose by the individual pilot [1,6]. If the pilot requires a continuous insulin treatment in the further course of the disease, he/she is not allowed to fly passenger plane anymore and is usually forever grounded [1,4]. These regulations are effective in the US and in Europe and have substantial financial consequences for both the affected pilot and the employer. Exceptions for this strict regulation exist in Europe only in England, Ireland, and Austria, which are the first countries to introduce the possibility to acquire and maintain the flying license for passenger aircrafts for insulin-treated pilots with type 1 or type 2 diabetes under certain conditions in 2012, 2015, and 2019, respectively.
The encouraging results of this approach from a cohort of 26 insulin-treated pilots have been published recently [7] and give room for the hope that this more liberal approach may be introduced in other countries as well. However, in the United States, in Germany, and in the other European countries, insulin-treated pilots cannot obtain the flight permit for professional passenger transportation at this point of time. In recent years, we have therefore investigated an alternative therapeutic approach to the current guidelines for diabetes therapy with several pilots in order to avoid long-term insulin therapy and to maintain the pilot’s career prospects. The aim of our intensive temporary polypharmaceutical intervention was firstly, to achieve a comprehensive remission of the disease by actively protecting the ß-cells, and secondly, to stabilise this remission for as long as possible through lifestyle measures. If necessary, the treatment was repeated at regular intervals.
De-escalation therapy (DET) is a personalized therapeutic approach for type 2 diabetes mellitus, in which a special diagnostic procedure is performed to determine the phenotype of the disease [8,9]. In addition to the measurement of classical disease markers (HbA1c, fasting glucose, lipids, kidney values), various other specific laboratory parameters (DET root cause parameters) are determined, e.g. insulin, intact proinsulin, total adiponectin and high sensitivity C-reactive protein (hsCRP) [10], to determine the extent and severity of the basic pathophysiological disorders of type 2 diabetes (ß-cell dysfunction, insulin resistance, chronic systemic inflammation). On the basis of the results, we select a lowdose combined pharmacotherapy with a total of four components together with the patient. The individual drugs are selected from a range of options to target ß-cell dysfunction (basal insulin or shortacting insulin), insulin resistance (plioglitazone or rosiglitazone), and the increased hormonal activity of the visceral lipid tissue (Glucagon-like peptide 1(GLP1) agonists, Sodium-Glucose-Co- Transporter-2 (SGLT2) inhibitors, metformin).
The individual selection of the drugs is based in particular on patient preference, and the doses are selected based on the biomarker result distribution. As a general rule, each drug is provided at low dose, but the drug targeting the most dominant deterioration is given at a higher dose. This approach requires extensive information of the patient regarding the planned therapy measure. Furthermore, the consent of the patient should be documented. Subsequently, the patient is trained with regard to simple nutritional recommendations (calorie-reduced, low carb), possible options for exercise, and the usefulness of regular blood glucose determinations. The patient then applies the therapy as consistently as possible for three months, whereby in our practice the laboratory determinations are repeated after 6 weeks to determine the intermediate therapy result. In this phase, the pilot cannot fly because of the insulin therapy, which is why a close interdisciplinary coordination between pilot physician and diabetologist is necessary.
The patient may be motivated by the fact that after just a few days of therapy, he/she is practically only measuring normal blood glucose levels. It is therefore of importance to remind the patient that the treatment target is to improve ß-cell function, which can only be achieved if the drugs are taken during the entire treatment period. After 12 weeks, all medication is discontinued, and after a further week, the patient undergoes an oral glucose tolerance test (OGTT) with 75g of Glucose. Depending on the OGTT results and the other laboratory parameters, further treatment is determined. In the majority of cases a consistent continuation of the lifestyle measures is initially sufficient for further therapy after this extensive pharmacological intervention. The DET biomarkers and HbA1c are then measured every 6 months. The OGTT is repeated at annual intervals. In this phase, the patients can continue their work as traffic pilots without further restrictions.
The recurrence of diabetes is most likely to be detected by an increase in the fasting-intact proinsulin levels long before the blood glucose levels rise again. The significance of intact proinsulin as a predictive diabetes marker and indicator of progressive insulin resistance in general has been described earlier and has also been recently reviewed in the international literature [11-13]. According to our experience, the duration of the phase of freedom from therapy, which can last several years, depends in particular on the adherence of the pilots to the recommended lifestyle interventions. This report is of two airline pilots with type 2 diabetes mellitus who underwent DET and describes the use of an individualized approach based on a classic and novel biomarker panel.
Case Reports
Case 1
Patient W.A., male, 48 years old at first presentation in 2012, commercial pilot (long-haul, Boeing 747, 777 and in the course also A380). At the first presentation in September 2012, this patient (1.70 m tall, 82 kg body weight, body mass index (BMI): 26.4 kg/ m²) had already been diagnosed with type 2 diabetes for 4 years. Under the current therapy with metformin 1000 mg (morning and evening) and glimepiride 3 mg in the morning, he recently showed an HbA1c value of 7.3 % with a tendency towards further deterioration, which is why the aeromedical service and the family doctor considered the additional administration of long-term insulin. For the pilot, who actually wanted to train as an A380 pilot, this change in therapy would have meant the end of his career as a commercial traffic pilot. In our laboratory determinisations. The patient showed an HbA1c of 7.2 % (normal range: < 6.4 %), a significant increase in intact proinsulin (13.2 pmmol/L, normal range < 7 pmol/L), a pronounced suppression of total adiponectin (1.7 mg/dL, normal range for men: > 6 mg/dL). With 1.5 mg/dL his hsCRP value was in the moderate coronary heart disease risk range according to Ridker (>1 - 3 mg/dL [14]).
Our personalized diagnosis was accordingly: ß-cell dysfunction in stage III [12] with pronounced hormonal activity of the visceral fat tissue and with moderate cardiovascular risk. For his DET, a combination consisting of moderate dose insulin glargine (10 Usc. at night), low dose liraglutide (0.6 mg s.c. in the morning), moderate dose pioglitazone (30 mg p.o. in the morning) and low dose metformin (500 mg p.o. in the morning) was selected together with the patient. For the implementation of this therapy, the patient was released from active flight duty by the aviation physician. At the return after three months of therapy (November 2012 - January 2013), and one week after discontinuation of all medication, the patient showed a normal OGTT result (0h: 99 mg/dL, 2h: 126 mg/ dL) and an HbA1c of 5.7 %. All other parameters also had returned to normal. The patient was subsequently trained in helpful lifestyle measures to prevent the return of the disease and subsequently carried out the therapy without diabetes medication and exclusively with diet and exercise. After two years, a renewed increase in intact fasting proinsulin indicated the impending recurrence of type 2 diabetes.
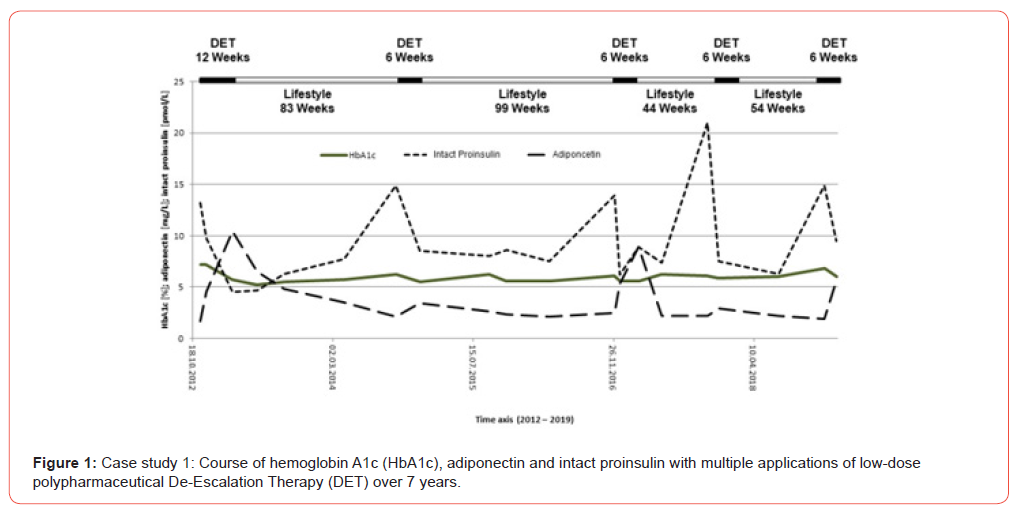
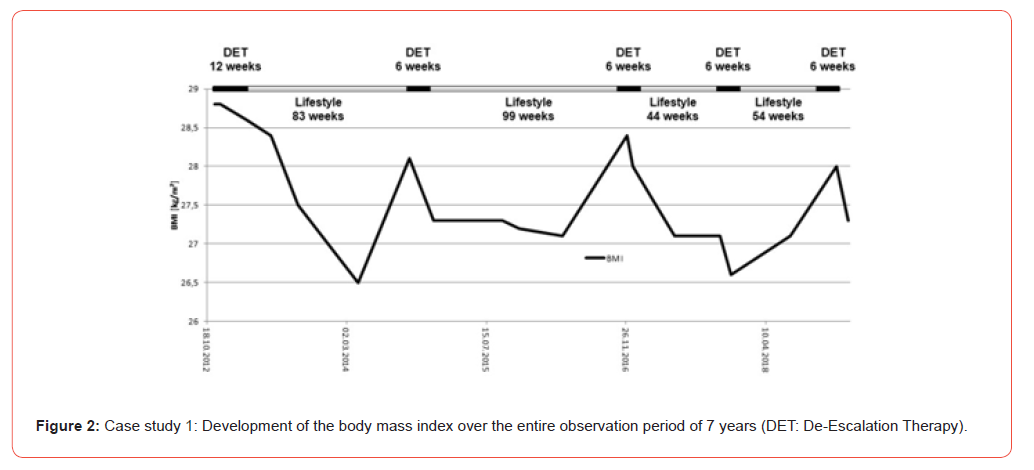
In this patient, it was not possible to perform the DET for more than 6 weeks at a time due to occupational reasons. Therefore, it was not possible to return all parameters to the normal range, as we have seen with this same patient in the first DET conduct and with many other patients. Overall, it can be said that this pilot has a stable blood glucose control despite high occupational stress by employing the temporary intermittent DET over 8 years now and is expected to be able to work actively as a traffic pilot in the next few years until retirement.
Case 2
Patient C.R., male, 57 years old at first presentation 02/2017 57, diabetes mellitus type 2 known for 9 years, commercial German airline pilot (short-haul A320/A321). Treatment at first presentation: Repaglinide (1 x 5 mg), metformin (2 x 1000 mg) and sitaglipin (2 x 50 mg), resulting in HbA1c: 7.4%. The blood tests showed a suppression of adiponectin to 3.9 mg/L with moderately elevated hsCRP (1.41 mg/L), and with intact proinsulin values in the upper normal range (6.9 pmol/L). We subsequently diagnosed stage II ß-cell dysfunction [10] with pronounced hormonal activity of visceral fat tissue and moderate cardiovascular risk.
DET was performed in this patient in consultation with the Aeromedical Service for three months with daily morning doses of 0.6 mg liraglutide (low dose), 30 mg pioglitazone (moderate dose), 10 mg empagliflozine (low dose), and 8 U of insulin glargine at night (low dose).
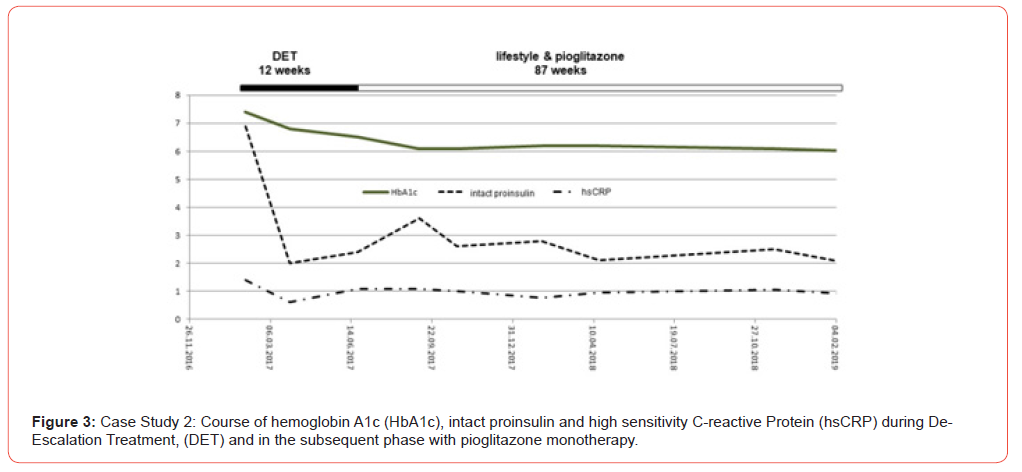
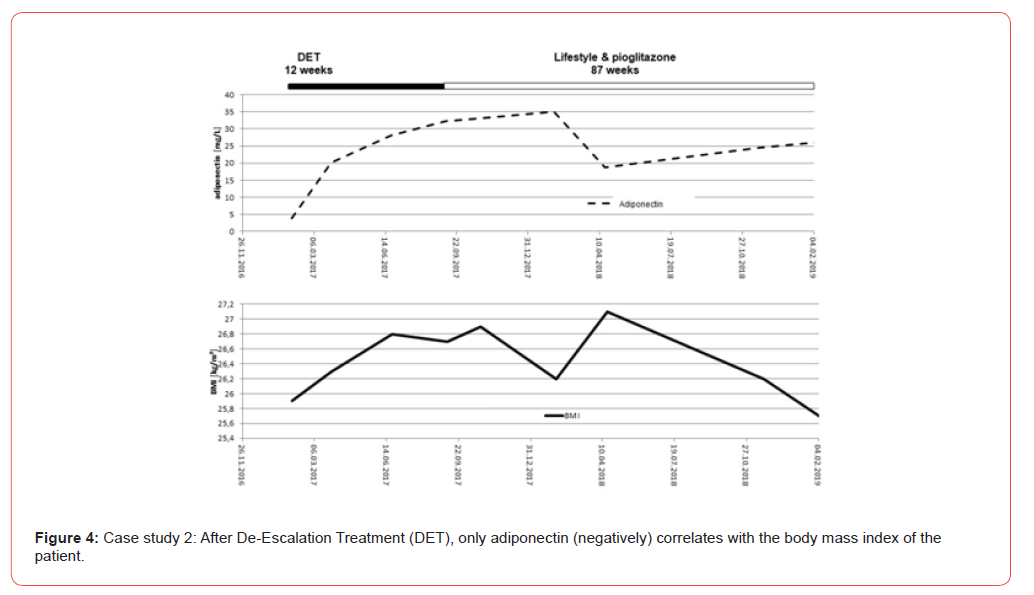
Under this therapy, HbA1c initially decreased to 6.5%, intact proinsulin to 2.1 pmol/L, hsCRP to 0.6 mg/L and adiponectin to 20.1 mg/L. As this patient had already received triple pharmacotherapy prior to DET, it was decided to continue therapy with the most appropriate monotherapy. This case had a diabetes phenotype in which insulin resistance was the most pronounced disorder as determined by the laboratory biomarker panel. Therapy was therefore continued with pioglitazone monotherapy (30 mg). As a result, HbA1c and intact proinsulin remained within the normal range for more than 1.5 years (Figure 3). Fluctuations in adiponectin levels were negatively associated with changes in BMI at the end of DET (Figure 4). Therefore, the course of the disease can so far be evaluated as extremely stable and without a current tendency towards chronic progression (Figures 3&4).
Discussion
The pathophysiology of type 2 diabetes is a complex event in which several fundamental disorders interact with each other. A genetic ß-cell secretion disorder leads to a temporal, quantitative and qualitative secretion disorder, which is accompanied by increased hormonal activity of the visceral fat tissue. This dysfunction is further aggravated by consecutive chronic systemic inflammation and insulin resistance with resulting further increased insulin requirements. Recent studies based on the comprehensive bioinformatic analysis of large patient populations have identified different subgroups or clusters in diabetes, which could be related to different severities of these underlying deteriorations, and which have been suggested by the respective authors to support targeted prevention and treatment [15,16]. However, the extent of the underlying disorders can also easily be determined by several biomarkers (e.g. HbA1c, insulin, intact proinsulin, adiponectin, hsCRP), which can also be used to detect normoglycemic precursor stages of type 2 diabetes (e.g. cardiodiabetes or prediabetes) and to treat them [10-14,17].
Our current approach with the aim of protecting and regenerating ß-cells was inspired by studies in China and other Asian countries. In these studies, a substantial remission phase was described after early use of insulin in type 2 diabetes. This phenomenon appears to be similar to the known “honeymoon” period in type 1 diabetes after start of insulin therapy [18,19]. In our opinion, the described effects of a short-term early insulin therapy can be substantially increased in their extent and prolonged in their temporal duration by the additional use of further pathophsiologically oriented therapeutics. The administration of pioglitazone already improves insulin resistance as monotherapy, has strong anti-inflammatory effects in combination therapy and reduces the cardiovascular risk [20,21]. The use of GLP-1 analogues (e.g. liraglutide) also helps in low-dose weight loss [22], reduces the amount and activity of visceral fat [23], and also reduces cardiovascular risk [24]. SGLT-II inhibitors also help with weight loss [25] and, according to current data, can normalize blood glucose levels and reduce cardiovascular risk [26].
The combination therefore appears to be sensible and, according to our observations, also shows clear positive effects. With our low-dose temporary combination therapy with the selected drugs, we did not observe any hypoglycaemia or other substantial side effects in our pilots or in numerous other patients, which would alter the benefit-risk ratio of the individual substances or that could have been additionally induced by the combination of the drugs. Only the administration of insulin could theoretically lead to a hypoglycaemia risk. However, with a maximum insulin dose of 8-12 U of the basal analog insulins used and insulin resistance present at the same time, hypoglycaemia is unlikely to happen even in combination with the other substances, and we have not seen any case so far. Our observations could possibly lead to a rethinking of diabetes therapy, at least in certain occupational groups. With DET, we have successfully tried to stop and even reverse the progression of diabetes in the presented cases but also in several additional professional pilots, at least during the active professional phase.
So far, we have succeeded in achieving this goal in practically all cases, provided the patients cooperated and really had type 2 diabetes, and no late manifesting type 1 diabetes. Due to the professional situation, the motivation of the affected pilots was very high. The subsequent drug-free phase following DET was of varying length and correlated with adherence to the recommended lifestyle measures (mean duration: 2-3 years). A repetition of DET is possible with comparable results. So far none of our more than ten pilots treated by this method has needed a permanent insulin therapy and all DET-treated Type 2 pilots are still in active service in commercial aviation. We are therefore encouraged to further investigate the DET approach as a possible additional treatment option, e.g. for patients in special professional situations. The patients can also initiate or resume treatment according to guidelines at any time. In summary, we would like to state that through a pathophysiological, personalized and time-limited deescalation therapy, a substantial remission of the disease was achieved in the described two pilots with type 2 diabetes, whereby the airworthiness could be completely and unrestrictedly restored for several years. If necessary, the therapy was repeated with reproducible success.
Conclusion
Phenotyping and DET represents a possible alternative to traditional diabetes therapy, which could be used in particular if the professional life of the patients could be significantly impaired by the progressive guideline-oriented treatment intensification. In our experience, DET is superior to traditional therapy in these cases at least initially and in terms of short-term prognosis until retirement, and probably also in terms of quality of life. Further studies and documented patient experiences are now necessary to build the scientific evidence of our anecdotal observations.
Conflict of Interest
None to report.
References
- Siedenburg J, Küpper Th (2015) Moderne Flugmedizin; Handbuch für Ärzte, Piloten, Fluglehrer, Flugschüler, Flugbegleiter und Flugreisende (Modern flight medicine-handbook for physicians, pilots, instructors, students, flight attendants and passengers). V. Gentner Verlag, Stuttgart.
- Pizzi C, Evans SA, De Stavola BL, Evans A, Clemens, et al. (2008) Lifestyle of UK commercial aircrews relative to air traffic controllers and the general population. ASEM 79(10): 964-974.
- Dunn JR (2010) Health behaviour vs the stress of low socioeconomic status and health outcome. JAMA 303(12): 1199-1200.
- Navathe P, Drane M, Preitner C (2014) Aeromedical decision making: From principles to practice. ASEM 85(5): 576-580.
- Mitchell SJ, Hine J, Vening J, Montague J, Evans S, et al. (2017) A UK Civil Aviation Authority protocol to allow pilots with insulin-treated diabetes to fly commercial aircraft. Lancet Diabetes Endocrinol 5(9): 677-679.
- Tajima N, Yamada C, Asukata I, Yamamoto K, Hokari M, et al. (1989) Pilots with non-insulin-dependent diabetes mellitus can self-monitor their blood glucose. Aviat Space Environ Med 60(5): 457-459.
- Pfützner A, Jantz J (2025) Glucose Cosmetics vs. Personalized Diabetes. Therapy-Part 1: Functional Phenotyping may Provide the Basis for an Individual Diabetes Treatment, Diabetologia (epub ahead of print).
- Pfützner A (2023) Glukosekosmetik vs. Personalisierte Diabetestherapie Teil 1: Funktionelle Phänotypisierung als Grundlage einer individuellen Typ 2 Diabetes Therapie. Diabetes Stoffw. Herz 32: 299-306.
- Pfützner A, Weber MM, Forst T (2008) A Biomarker Concept for Assessment of Insulin resistance, ß-Cell Function and Chronic Systemic Inflammation in Type 2 Diabetes mellitus. Clin Lab 54(11-12): 485-490.
- Pfützner A, Kunt T, Mondok A, Pahler S, Konrad T, et al. (2004) Fasting Intact Proinsulin is a Highly Specific Predictor of Insulin Resistance in Type 2 Diabetes. Diabetes Care 27(3): 682-687.
- Pfützner A, Pfützner AH, Larbig M, Forst T (2004) Role of intact proinsulin in diagnosis and treatment of type 2 diabetes mellitus. Diab Technol Ther 6(3): 405-412.
- Pfützner A, Sachsenheimer D, Lier A (2018) Erhöhtes Proinsulin als früher Hinweis auf einen zukünftigen Typ 2 Diabetes (Elevated proinsulin as early indicator for future type 2 diabetes). Diabetes Stoffw. Herz 27: 69-73.
- Pfützner A, Forst T (2006) HsCRP as cardiovascular risk marker in patients with diabetes mellitus. Diab Technol Ther 8(1): 28-36.
- Zaharia OP, Strassburger K, Strom A, Bönhof GJ, Karusheva Y, et al. (2019) German Diabetes Study Group. Risk of diabetes-associated diseases in subgroups of patients with recent-onset diabetes: a 5-year follow-up study. Lancet Diabetes Endocrinol 7: 684-694.
- Ahlqvist E, Storm P, Käräjämäki A, Martinell M, Dorkhan M, et al. (2018) Novel subgroups of adult-onset diabetes and their association with outcomes: a data-driven cluster analysis of six variables. Lancet Diabetes Endocrinol 6(5): 361-369.
- Kintscher U, Marx N, Koenig W, Pfützner A, Forst T, et al. (2006) Kardiodiabetologie: Der aktuelle Stand-Epidemiologie, Pathophysiologie, Therapie, Klinik und Praxis. (Cardiodiabetology: actual status – epidemiology pathophysiology, clinic, and practice) Diabetes Stoffw. Herz 15: 31-45.
- Weng J, Li Y, Xu W, Shi L, Zhang Q, et al. (2008) Effect of intensive insulin therapy on beta-cell function and glycaemic control in patients with newly diagnosed type 2 diabetes: a multicentre randomised parallel-group trial. Lancet 371(9626): 1753-1760.
- Kramer CK, Zinman B, Retnakaran R (2013) Short-term intensive insulin therapy in type 2 diabetes mellitus: a systematic review and metaanalysis. Lancet Diabetes Endocrinol 1(1): 28-34.
- Pfützner A, Marx N, Lübben G, Langenfeld M, Walcher D, et al. (2005) Improvement of Cardiovascular Risk Markers by Pioglitazone is Independent from Glycemic Control - Results from the Pioneer Study. J Am Coll Card 45(12): 1925-1931.
- Dormandy JA, Charbonnel B, Eckland DJ, Erdmann E, Massi-Benedetti M, et al. (2005) PROactive Investigators. Secondary prevention of macrovascular events in patients with type 2 diabetes in the PROactive Study (PROspective pioglitAzone Clinical Trial In macroVascular Events): a randomised controlled trial. Lancet 366(9493): 1279-1289.
- Jendle J, Nauck MA, Matthews DR, Frid A, Hermansen K, (2009) LEAD-2 and LEAD-3 Study Groups. Weight loss with liraglutide, a once-daily human glucagon-like peptide-1 analogue for type 2 diabetes treatment as monotherapy or added to metformin, is primarily as a result of a reduction in fat tissue. Diabetes Obes Metab 11(2): 1163-1172.
- Santilli F, Simeone PG, Guagnano MT, Leo M, Maccarone MT, et al. (2017) Effects of Liraglutide on Weight Loss, Fat Distribution, and β-Cell Function in Obese Subjects with Prediabetes or Early Type 2 Diabetes. Diabetes Care 40(11): 1556-1564.
- Marso SP, Daniels GH, Brown-Frandsen K, Kristensen P, Mann JF, (2016) LEADER Steering Committee; LEADER Trial Investigators. Liraglutide and Cardiovascular Outcomes in Type 2 Diabetes. N Engl J Med 375(4): 311-322.
- Ribola FA, Cançado FB, Schoueri JH, De Toni VF, Medeiros VH, et al. (2017) Effects of SGLT2 inhibitors on weight loss in patients with type 2 diabetes mellitus. Eur Rev Med Pharmacol Sci 21(1): 199-211.
- Zinman B, Wanner C, Lachin JM, Fitchett D, Bluhmki E, et al. (2015) EMPA-REG OUTCOME Investigators. Empagliflozin, Cardiovascular Outcomes, and Mortality in Type 2 Diabetes. N Engl J Med 373(22): 2117-2128.
- Pfützner A, Lewin J, Do L, Liu J, Demircik F, et al. (2020) De-Escalation Therapy (DET)-An intensive short-term temporary pharmacological intervention to stop disease progression in patients with type 2 diabetes. Abstract presented at the Diabetes Technology Meeting 2019. J Diabetes Sci Technol 14: 391-462.
-
Andreas Pfützner*, Julia Jantz, and Dirk M Rose. De-Escalation Treatment (DET) as a Personalized Pharmacological Intervention in Two Airline Pilots with Type 2 Diabetes Mellitus. Case Report. 4(5): 2025. ACCS.MS.ID.000597.
-
Type 2 diabetes; remission phase; airline pilots; ß-cell regeneration; de-escalation therapy; iris publishers; iris publisher’s group
-

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.






