 Review Article
Review Article
Catalytic Role of Antioxidants in Mitigating the Physiological Consequences of High-Altitude- Associated Environmental Stressors
Poornima Sharma and Yasmin Ahmad*
Defence Institute of Physiology & Allied Sciences (DIPAS), Defence R&D Organization (DRDO), Timarpur, New Delhi-110054, India
Yasmin Ahmad, DDT Division, DIPAS, DRDO, New Delhi-110054, India.
Received Date:October 27, 2025; Published Date:November 19, 2025
Abstract
Despite the allure of high-altitude environments, they present a myriad of challenges, including reduced barometric pressure, heightened ultraviolet radiation, severe cold temperatures, and arid climates. These adversities, ranging from scorching days to freezing nights, coupled with hypobaric hypoxic conditions, impact an individual’s ability to tolerate hypoxia. Every year, millions of individuals venture to high-altitudes for various purposes, ranging from employment and sports to leisure activities and professional obligations. High-altitude-associated environmental stressors prompt an elevation in free radical generation, which reduces the effectiveness of the antioxidant system, resulting in increased reactive nitrogen and oxygen species production at high-altitude, resulting in oxidative damage to macromolecules. Anti-oxidant supplementation demonstrated a promising approach to oxidative damage prevention by preserving a balanced ratio of pro- and antioxidants and by maintaining cellular homeostasis. This article addresses the convoluted relationship between oxidative stress and high-altitude-associated environmental stressors, coupled with the safeguarding advantages associated with antioxidant supplementation. It investigates the process of ROS generation and cellular ROS-associated proteomic disruptions driven by high-altitude hypobaric hypoxia. In conclusion, this article strengthens comprehension of the efficacy of the antioxidant strategies contributing in lowering the oxidative damage whilst improving general wellbeing in high-altitude habitats.
Keywords:High-altitude; Environmental Stressors; Hypoxia; ROS; Anti-Oxidant Supplementation
Background
Multiple high-altitude (>2500m) destinations are present worldwide that captivate millions of travelers and adventurers each year, such as Mount Elbrus, Mont Blanc, Aconcagua, and Mount Kilimanjaro. Through the lens of ecology, high-altitude habitats are more vulnerable to external changes as opposed to lower-altitude habitats. Rapid high-altitude ascent prompts multitudinous physiological changes in a very short time due to the drop in atmospheric pressure and declining oxygen partial pressure caused by the lowering of the density of air molecules. This condition is typified by hypobaric hypoxia. Speedy ascent to high-altitude leads to maladaptation to the high-altitude environment, which enhances ROS generation and causes oxidative stress [1]. Hypoxia-dependent oxidative stress is determined by two factors, termed as exposure dose and exposure time. The elevated catecholamine generation [2], induction of the xanthine oxidation pathway [3], and diminished mitochondrial redox potential [4,5] accompanies the primary mechanism employed for enhanced ROS production. High-altitude induced oxidative stress, resulting in significant alterations to the proteome such as the formation of carbonyl groups on proteins, altered protein structure and function, formation of nitrotyrosine, protein aggregation, protein degradation, induction of generation of inflammatory responses, and inflammatory cytokines [6,7] Cognizance of these changes is pivotal for evolving antioxidant strategies to mitigate the high-altitude stress and its effect on the proteome. Thus, the antioxidant supplement should be specifically designed for oxidative stress at high-altitude. In this article, we will explore the various mechanisms through which high-altitude exposure leads to oxidative stress and its impact on the body [8,9]. Throughout this article, we aim to comprehend the oxidative stress due to high-altitude exposure, ROS production at high-altitude, the high-altitude effect on the antioxidant system, proteomic disruptions caused by ROS, and the development of targeting antioxidant strategies, their efficacy in mitigating the high-altitudeinduced stress, and case studies.
High-altitude Environment Stressors
Yearly, Plentiful hordes are immersed in high-altitude travel with the aim of professional endeavors, vocational activities, employment, pleasure, trekking, national security, and professional duty. Majorly high-altitude environmental stressors are characterized by declining barometric pressure, peak ultraviolet radiation exposure, severe frigid conditions, parched climates, and hypoxia [8].
Declined Atmospheric Pressure and Altitude Hypoxia
Oxygen deficiency in mountainous terrain is a principal ramification of the subdued partial pressure of oxygen reduced atmospheric pressure [9]. Generally, high elevation refers to a rise exceeding 1500 meters above sea level. An In-depth categorization of high-altitudes is provided in Table 1.
Table 1: High-altitude categorization (Ali, et al. 2023).
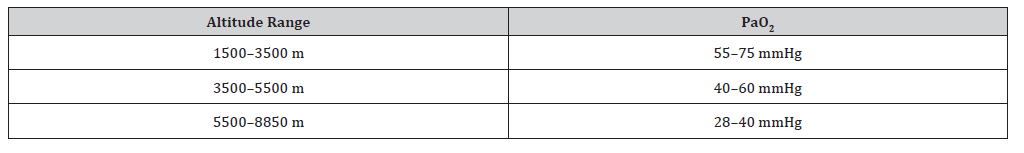
The troposphere, the lowermost region of the air envelope, comprises nearly 20.93% of oxygen. The complex interaction between topography and meteorology allows us to derive the partial pressure of oxygen (pO2) in ambient air by multiplying 0.2093 by the barometric pressure at a specific elevation. At sea level, this calculation yields a pO2 of 0.2093 x 760 mmHg, equating to 159.1 mmHg. Contrastingly, at the crest of Mount Everest, the pO2 steeply descends to 52.9 mmHg. The steep decline in barometric pressure with mountain terrain is the predominant cause of highaltitude hypoxia, which leads to Altitude Mountain Sickness (AMS) [10,11]. AMS-associated clinical symptoms are characterized as vertigo, fatigue, insomnia, and anorexia. Acclimatization consists of multiple complex physiological modifications that promote highaltitude adaptation and reduce the risk of AMS development [12].
Neurological manifestations at high-altitude encompass a spectrum of conditions, most prominently High-Altitude Cerebral Edema (HACE), which displays numerous diagnostic symptoms, namely behavioral incongruities, cognitive dysfunction, ataxia, papilledema, and radiological evidence of cerebral swelling [13,14], as well as findings that seem reminiscent of the organic brain [13,15]. HACE may promptly progress into a vegetative state with catastrophic consequences if it fails to receive medical attention within an appropriate time frame [16].
In conclusion, mild to moderate AMS symptoms often worsen over the course of 24 to 72 hours following the ascent. Notably, the time window for the onset of HACE symptoms usually coincides with the peak AMS presentation period, suggesting a possible transition between AMS and HACE [17,18] The diagnosis of HACE is not ruled out in the absence of preceding or present HAH, which is an additional hallmark of AMS. As a result, in certain instances, deterioration may seem to happen quickly— a HACE-induced coma may develop in the time frame of 12 hours [13]. This could lead to a physiological or anatomical predisposition, or even a failure to recognize important prodromal signs in time. The existence or absence of concomitant high-altitude pulmonary edema (HAPE) is probably an important component in the development and course of HACE [19,20]. Remarkably, around 15% of individuals with HAPE diagnoses also have HACE [21]. Simultaneously, pulmonary edema has been reported to be widespread in the few hospitalized cases of severe HACE; around 85–100% of patients suffering HACE were diagnosed with HAPE [22,23]. It is also like that endothelial dysfunction can be the cause of elevated circulating inflammation [24] or higher resistance in the pulmonary artery, which raises venous pressure from the right heart to the brain.
Heightened Ultraviolet Radiation
The sun emits ultraviolet radiation, which is a component of the electromagnetic spectrum, which has seven different wavelengths: X-rays, microwaves, radio waves, visible light, ultraviolet, infrared, and gamma rays. Ultraviolet radiation is “beyond violet,” or beyond visible light [25]. German scientist Johann Wilhelm Ritter discovered UV radiation in 1801. Ritter found that invisible light that fell outside of the visible spectrum’s violet region caused silver chloride-treated paper to discolor faster than violet light. Since this invisible light was chemically reactive, he first referred to it as “oxidizing rays.” Before the late 19th century, “thermal radiation” was recognized as infrared radiation, and “actinic rays” were reclassified as UV radiation. The ultraviolet spectrum intersects between visible light and X-rays in the electromagnetic spectrum. Its wavelengths range from approximately 380 nanometers (nm) to 10 nm, and its frequencies range from around 8 × 10¹⁴ to 3 × 10¹⁶ hertz [26]. Generally, there are three sub-bands of UV radiation, as shown in Figure 1.
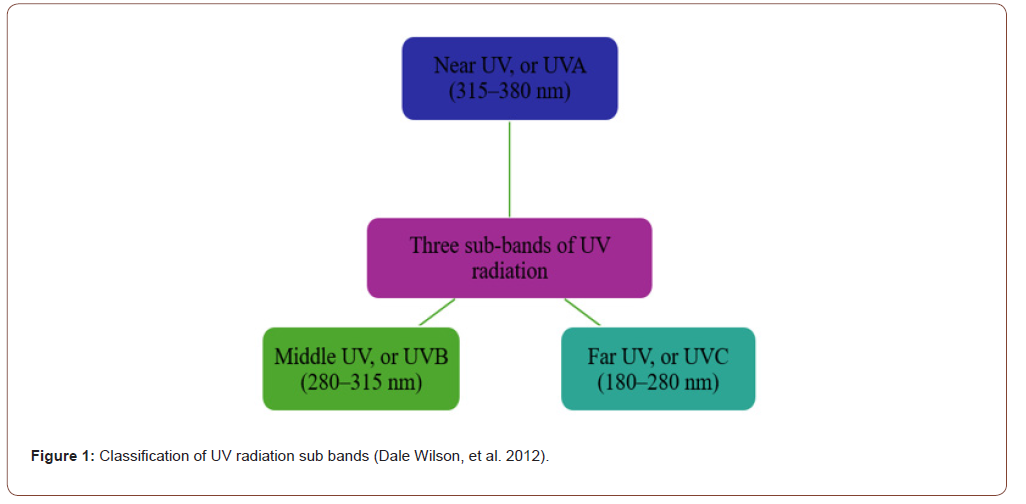
Radiations with a wavelength between 10 and 180 nm are often known as “vacuum or extreme UV” since they can only travel in a vacuum and are typically absorbed by the Earth’s atmosphere. Besides the sun, numerous celestial sources release UV radiation. High-altitude balloons and space-based telescopes equipped with specialized imaging sensors and filters are employed to estimate wavelengths < 280 nm absorbed by the Earth’s surface. Moreover, scientific inquiries into the effect of evaluating the surface temperature of the hottest stars and detecting the presence of interstellar gas clouds between the Earth and quasars [27].
Potential Consequences of Ultraviolet Radiation at High Altitude
Solar radiation is the main source of natural ultraviolet light that humans encounter. Approximately 10% of solar radiation comprises UV wavelengths; however, only one-third of this spectrum can be absorbed in the atmosphere, affecting the Earth’s crust. In equatorial regions, UVA comprises 95% of the solar UV radiation, while UVB constitutes the remaining 5%. Remarkably, solar UVC is productively neutralized by water vapor, ozone, and molecular oxygen in the mountainous atmosphere, preventing any substantial influence on the terrestrial surface. However, wide-range ultraviolet radiation (UVB and UVA) continues to be the most potent and environmentally destructive element for terrestrial life. The Alps are one of the regions in Europe exhibiting the most heightened ultraviolet (UV) radiation level, which may be attributed to an aggregation of snow-covered surfaces, elevated altitudes, and snow-covered surfaces. The thin air, cloud cover, and ozone contribute to the elevated solar UV radiation in mountainous terrain. Research has found that UV radiation dynamics in mountainous terrain are significantly impacted by altitude effects. In mountainous environments, the altitude effect has a significant impact on UV radiation dynamics. Quantification of erythemal UV radiation in the Swiss Alps has shown a rise of 17.4% per 1000 meters under unobstructed skies [28]. Thus, the high ultraviolet radiation at high-altitude is sensitive to the photochemical injury of the ocular space. It is associated with the clinical symptoms such as ocular pain, tearing, blepharospasm, deterioration of vision. The acute UVR effect cause a condition called photokeratitis. It is a temporary, painful eye damage. It may also refer to the drying and freezing of corneal surface, due to the severe dry air at high-altitude [29].
Dry air, lower relative humidity, high wind velocity and low temperature cause a decrease in water content of skin. Tanning, acute and chronic sunburn, photomelanosis, actinic dermatitis, actinic cheilitis are the complications caused by abnormal cutaneous reactions to solar radiation [30]. UVA contributes to the generation of reactive oxygen species and subsequently induces inflammatory signaling pathways. Free radicals induced by UV radiation activates the function of various signaling molecules such as AP-1 (Activator protein), NF-κB (nuclear factor kappa-lightchain- enhancer of activated B cells), STAT (signal transducer and activator of transcription) and COX-2 [cyclooxygenase 2], which leads to inflammation upregulation, which in turns activates the generation of reactive oxygen species. These reactive oxygen species are responsible for the DNA lesions and increase the chances of mutation [31-32].
Oxidizing Mechanism Associated with Ultraviolet Radiation
UVR is another important high-altitude environmental stressor that mediates oxidative imbalance by photooxidation. It induces a series of redox reactions leading to the generation of free radicals and causing oxidative imbalances in the cell [33]. The UV radiation induced oxidizing mechanism is accompanied by direct photochemical reactions and indirect photosensitized process, which significantly enhances the ROS generation within the cells which involving singlet oxygen, hydroxyl radicals, and hydrogen peroxide through multiple mechanisms. Lower levels of ROS can cause mutations, medium levels of ROS can trigger the mechanisms involved in senescence, and higher ROS levels may lead to cell death, called necrosis and apoptosis. These reactive oxygen species excessively damage the nucleic acid, proteins, and lipids, specifically via lipid peroxidation and generation of reactive aldehydes such as 4-hydroxynonenal and malondialdehyde [33]. Photooxidation is one of the principal oxidizing mechanisms. It involves the direct activation of chromophores such as porphyrins, tryptophan, and riboflavin by UV photons, which causes the formation of highenergy intermediates that transfer the energy to molecular oxygen. This interaction will lead to the production of reactive oxygen species such as superoxide anions and hydroxyl radicals, which oxidize lipids, proteins, and nucleic acids. Photooxidation increases the activity of mitogen-activated protein kinase (MAPK) and regulates the expression of NF-κB and activator protein-1 (AP-1) by elevating ROS production [34]. In tandem, photosensitization plays a significant role, specifically in the presence of a photosensitizer (light-absorbing molecule). These molecules absorb the UV radiation and undergo excitation, contributing to the electron transfer mechanism called Type I reactions or the energy transfer mechanism called Type II reactions. Collectively, these mechanisms elicit the ROS production in response to UV radiation [35].
Severe Cold Temperatures and Arid Climates
High-altitude regions are areas > 2500 m, which encompass elevations reaching up to 5500 m, as exemplified by the Siachen Glacier and regions within the Leh-Ladakh. Factors like low oxygen sensing, frigid temperature, pervasive snow squalls, and a scarce supply of flora and food contribute to the deterioration of natural settings at elevation. High-altitude indigenous populations, called highlanders, show substantial adaptation to these harsh environments; however, they still continue to confront considerable difficulties in their daily lives. Military deployment in such an inclement habitat presents a variety of grave concerns, ranging from minor exertional dyspnea, erythema, pernio, and prolonged sleep latency to potentially fatal ailments like photokeratitis, myocardial infarction, and HAPE. Limb endangering injuries like frostbite, pernio, and trench foot may impair the body’s functionality, and severe circumstances may lead to amputation [36]. Extremities are most susceptible to cold injuries, such as frostbite, in alpine and subzero environments. Lower extremities are more affected than upper extremities. Damage caused by frostbite displays immediate functional impairment and long-term sequelae such as persistent pain, cold sensitivity, and regional osteoporosis, that remain for a prolonged duration of time following the initial injury [37-38].
Oxidative strain and Elevation exposure
A steady-state balance between the antioxidant and prooxidant is essential for oxygen-requiring existence. It is universally recognized that hilly elevations are responsible for hastening the free radical generation and leading to serious health emergencies. The scholarly inquiry emphasized the effect of mountainous terrain on oxidative stress. Elevations above 3000 m are considered dangerous for unacclimatized individuals, and elevations exceeding 5500 m are fatal and pose a life-threatening hazard to individuals. These risks come with cardiopulmonary alterations that are induced by hypoxia and impair the normal functioning of the body. Adaptive responses from the human body are required in response to the reduced atmospheric oxygen partial pressure at high-altitude, which may result in subcellular physiological imbalances and irreversible or reversible injury [Sharma et al., 2023]. Some of the most immediate cellular responses to hypoxia provoked by altitude include elevated ROS production. Although cells can regulate their ROS levels, irreparable damage to proteins, lipids, and other biomolecules can result from a failure to promptly mitigate ROS. Cells enhance their antioxidant defenses and adjust the redox environment in response. In clinical settings, dietary interventions that employ antioxidants have proven efficacy [39].
Free Radical Generation at High-altitude
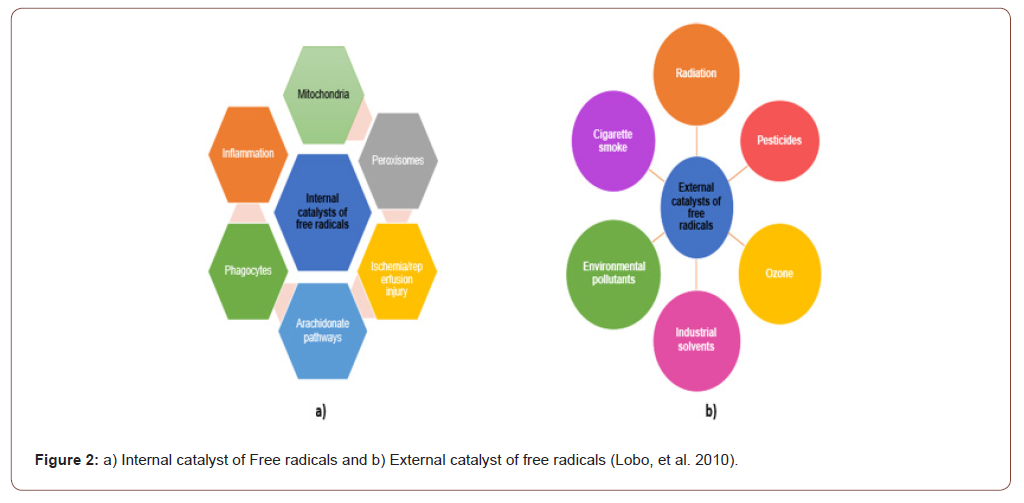
High-altitude environmental conditions are accompanied by lower oxygen pressure and declined barometric pressure, which leads to the enhanced generation of reactive oxygen species due to the hypoxia. The presence of an unpaired electron in an atomic orbital defines a free radical, which is any chemical species capable of existing independently. Most radicals have several similar features due to the presence of an unpaired electron. Many radicals exhibit high levels of reactivity and instability. They may act as oxidants or reductants, depending on whether they provide or receive an electron from other molecules [11]. The most important oxygencontaining free radicals in many diseases are the hydroxyl radical, the superoxide anion radical, the oxygen singlet, the hypochlorite radical, the nitric oxide radical, and the peroxynitrite radical. These are very reactive species that may harm physiologically significant components, including DNA, proteins, carbohydrates, and lipids in the cell’s membranes and nucleus [12]. Free radicals injure cells and disturb homeostasis by attacking crucial macromolecules. All molecules in the body are targets for free radicals [13-40] Some internal and external catalysts for free radicals are shown in Figure 2.
ROS-Associated Signaling Pathways in Response to Hypobaric Hypoxia
Reactive oxygen species are oxygen-based compounds and are characterized by their high reactivity and instability. These molecules can be produced from different signaling pathways, such as xanthine oxidase, mitochondrial electron transport chain, the NADPH oxidase complex, and uncoupled nitric oxide synthase (NOS). The most studied ROS molecules are superoxide (O2•−), peroxide radicals (ROO•), hydroxyl radicals (•OH), hydrogen peroxide (H2O2), singlet oxygen (1 O2), and peroxynitrite (ONOO−) [41]. Oxidative stress caused by hypoxia exposure is responsible for the endoplasmic reticulum dysfunction. It may lead to the aggregation of misfolded/unfolded proteins, which in turn activate the various processes associated with the inflammatory pathways. Among these, HIF-1α, NRF2, and eNOS play a significant role in regulating inflammation and antioxidant defense.
Hypoxia-inducible factors such as HIF-1α and HIF-2α are the primary effectors of cellular hypoxic response. These transcription factors regulate the expression of an array of genes associated with enhanced oxygen supply [42]. Under normoxic conditions, prolyl hydroxylases (PHDs) hydroxylate HIF-1α, leading to its recognition and degradation via the VHL (von Hippel–Lindau) proteasome pathway. Hypoxic conditions impair the activity of prolyl hydroxylase. After stabilization, it dimerizes with HIF-1β in the nucleus to trigger the transcription of genes associated with angiogenesis, ROS detoxification, and glucose metabolism [43]. Its downstream signaling includes the regulation of the expression of endothelial nitric oxide synthase activity [44]. The eNOS produces Nitric oxide (NO), which is required for vascular homeostasis. eNOS is exclusively present in endothelial cells. Studies found that hypoxia-mediated expression of eNOS is ambiguous and complex. eNOS was found to be inhibited in in-vitro hypoxia studies of cultured human umbilical vein endothelial cells, human coronary artery endothelial cells (HCAECs), bovine pulmonary artery endothelial cells (PAECs), human saphenous vein endothelial cells, as well as in vivo in the lungs of patients with pulmonary hypertension. Studies concluded that the effect of hypoxia on eNOS depends on whether it is a venous or arterial endothelium [45]. A study shows the upregulation of eNOS expression in the endothelium from the uterine of pregnant sheep in response of chronic hypoxia. Hypoxia and ischemia decrease the endothelial eNOS expression via a posttranscriptional mechanism [46]. HIF-1α stabilization in hypoxic conditions mediates the cascade of gene expression; its downstream effect intersects with the NRF2-controlled antioxidant mechanism which induces cellular adaptation to limited oxygen supply. NRF2 is considered as a master transcriptional factor controlling antioxidative defense mechanisms. On activation, NRF2 mediates the expression of various genes involved in cell proliferation, antioxidant and survival. In normoxic condition, NRF2 is found to be associated with KEAP1 (Kelch-like ECH-associated protein 1) in the cytoplasm which facilitates the NRF2 ubiquitination and proteasomal degradation and sustains the NRF2 levels low. In hypoxic conditions, enhanced mitochondrial ROS production oxidizes cysteine residues on KEAP1, which prevents KEAP1 from targeting NRF2 for degradation. Then, NRF2 aggregates and moves into the nucleus and get binds to the antioxidant response elements (AREs) which mediates the upregulation of genes involved in ROS detoxification (SODs, Prdxs), Glutathione metabolism (GCLC, GCLM), NADPH regeneration (NQO1, G6PD) in response to oxidative stress [47].
Impact of high-altitude on antioxidant systems
Aerobes have evolved intricate enzymatic and non-enzymatic mechanisms for antioxidants to combat the effects of reactive oxygen and nitrogen species. The enzymatic system consists of mitochondrial manganese superoxide dimutase (Mn-SOD), extracellular SOD, and cytosolic copper zinc superoxide dimutase, which converts the reactive superoxide radicals into less hazardous hydrogen peroxide. Subsequently, glutathione peroxidase (GPX) and catalase facilitate the decomposition of hydrogen peroxide into water. Sparse literature has investigated the protective effects of antioxidant enzymes at high-altitudes. Early studies have documented that rats’ skeletal muscle showed reduced activity and protein levels of mitochondrial SOD after six months of sporadic exposure to high-altitude (4000 m). Another corroborated study indicated that 5500 m of simulated altitude enhanced immunoreactive Mn-SOD levels in serum but lowered it in the animals’ liver and lungs [48]. Additionally, the decreased activity of the liver’s GPX suggests a greater susceptibility to high-altitudeinduced oxidative stress. Conversely, in another study, no discernible impacts were found on antioxidant enzyme activity during a fourweek exposure at 4000m [49]. Through comparative evaluation, it was established that GPX activity in the serum of native highlanders (4000 m) and sea-level subjects was lower than in high-altitude residents [50]. The glutamyl cycle continuously generates glutamylcysteinyl- glycine, which is an essential thiol/antioxidant in cells. Exposure to high altitudes lowers reduced glutathione (GSH) levels and raises oxidized glutathione concentrations.
The thiol system state has a significant impact on GPX’s activity and efficacy. The glutamyl cycle generates glutamyl-cysteinyl-glycine constantly, which is an essential thiol/antioxidant in cells. Exposure to high altitudes lowers reduced glutathione (GSH) levels and raises oxidized glutathione concentrations [51]. Thus, it seems that both enzymatic and non-enzymatic antioxidant mechanisms are slightly less effective at high-altitude. High-altitude has prompted the use of antioxidant supplements to mitigate its oxidative damage. Empirical research highlighted that an antioxidant combination that included ascorbic acid, catechin, selenium, lutein, alpha-lipoic acid, and N-acetyl cysteine was found to decrease the oxidative stress caused by a high-altitude exposure [52]. A brief simulated hypobaric hypoxia exposure of rats to an altitude of 8000 m resulted in increased blood melatonin levels [53]. Melatonin, which has a wide range of effects, also functions as an antioxidant. After the first four days following exposure, the number of mitochondria and lipid droplets in pinealocytes decreased compared to control rats, suggesting an additional source of melatonin beyond pinealocytes. In summary, high-altitude exposure appears to reduce the activity and content of certain antioxidant enzymes and diminish the effectiveness of the thiol system. There is some evidence that antioxidant supplementation can mitigate or prevent high-altitudeinduced oxidative damage to macromolecules [48].
Antioxidants and their protective role
An antioxidant is characterized as a stable molecule that has the ability to donate an electron to encounter and nullify a reactive free radical, thereby lowering the cause of potential damage. Therefore, antioxidants effectively prevent or hinder cellular damage due to their capacity to neutralize free radicals [54]. Antioxidants with reduced molecular weight may interact cautiously with oxidative stressors to halt their damaging chain reactions before they damage crucial molecules. Some antioxidants are produced naturally in the body during normal metabolic processes, for instance, ubiquinol, glutathione, and uric acid [55]. Others are obtained from dietary sources. Although the body has numerous enzymatic systems to encounter free radicals, for instance, vitamin C (ascorbic acid), vitamin E (α-tocopherol), and β-carotene [56], Since the body cannot synthesize certain micronutrients independently, they must be obtained through dietary intake.
Levels of Antioxidant Action
In the world of antioxidant defense systems, there are different levels of action, each with its own specific purpose. These levels include radical scavenging, protective measures, repair and synthesis (de novo), and adaptation—the last line of defense. The first line of defense is associated with the preventive antioxidants, which prevent the formation of free radicals. While the precise mechanism and site of ROS formation in the living system remain partially obscure, metals are considered significant sources of ROS generation due to their facilitation of the breakdown of hydroperoxides and hydrogen peroxide. To stifle this reaction, certain antioxidants are anticipatorily transformed from hydroperoxide into alcohols and hydrogen peroxide into water without producing free radicals. It is known that glutathione peroxidase, glutathione-S-transferase, phospholipid hydroperoxide glutathione peroxidase (PHGPX), and peroxidase enzymes can change lipid hydroperoxides into alcohols [57].
PHGPX is particularly noteworthy for its potential to diminish phospholipid-bound hydroperoxides within the bio membrane. Furthermore, glutathione peroxidase and catalase are essential for the conversion of hydrogen peroxide into innocuous water molecules. The second line of defense entails neutralizing active radicals, thereby inhibiting the chain reaction and disrupting the propagation of oxidative processes. Based on solubility, a variety of endogenous radical-scavenging antioxidants are likely watersoluble (hydrophilic), such as vitamin C [58], uric acid [59], bilirubin [60], albumin, and thiols [61], while others are lipidsoluble (lipophilic), such as vitamin E and ubiquinol [62]. Notably, scientists recognize Vitamin E as the most potent lipophilic antioxidant for radical scavenging. Repair and de novo antioxidants accompany the tertiary defense system. There are peptidases, proteases, and proteinases in the cytosol and mitochondria of mammalian cells. This helps to find, break down, and remove oxidatively modified proteins, which stops them from building up. Similarly, the total defense system against oxidative damage relies heavily on genome maintenance systems, while enzymes such as nucleases and glycosylases play a crucial role in restoring damaged DNA strands. Ultimately, another important function of adaptation serves a critical role, where the signals for the generation and reaction of free radicals induce the synthesis and targeted delivery of antioxidants to their specific sites of action [40] (Figure 3).

Reactive Oxygen Species (ROS) Generation and Cellular ROS-Associated Proteome Disruptions Driven by High-Altitude Hypobaric Hypoxia
High-altitude habitation triggers multifaceted physiological changes, predominantly due to fluctuating oxygen. The lung is the primary organ to confront fluctuating oxygen levels and plays a fundamental role in mediating and counterbalancing these alterations. The extant literature encompasses a multitude of comprehensive and astonishing studies that scrutinize proteomic alterations consequent to hypobaric hypoxia exposure. These investigations elucidate a plethora of biochemical pathways and responses elicited upon immediate exposure, as well as the mechanisms underlying the resolution of these effects amidst the challenges posed by hypobaric hypoxia. A study identified significant pulmonary proteomic modifications in response to varying time durations of hypobaric hypoxia using 2-DE/MS. Their findings suggest that SULT1A1 may serve as a promising biomarker for detecting pulmonary edema (HAPE), with elevated SULT1A1 levels found in both rodents and humans exposed to elevations, in contrast to controls.
Using 2-DE/MS, a study demonstrated that the lungs’ proteome changes significantly when exposed to variable durations of hypobaric hypoxia. Researchers found that SULT1A1 may serve as a promising biomarker for diagnosing high-altitude pulmonary edema (HAPE), with elevated levels of SULT1A1 observed in both rats and humans exposed to high altitudes compared to sea-level controls. It is a phase II detoxification enzyme which catalyses the sulfonation of hydroxyl–containing compounds. Its upregulation in HAPE indicates a protective detoxification response to lipid peroxidation byproducts elicited by reactive oxygen species under hypoxic conditions [63].
A study was performed on the analysis of the platelet proteome to investigate the prothrombotic phenotype induced by acute hypoxia. This research showed rats subjected to acute simulated hypoxia (282 torr/8% oxygen) exhibited a declining hemorrhagic propensity and heightened platelet reactivity. Proteomic analysis revealed 27 differentially expressed proteins, including those involved in coagulation processes. Notably, calpain small subunit 1, a 28-kDa regulatory component crucial for calpain function, showed significant upregulation under hypoxic conditions. They found a correlation between calpain small subunit 1 expression and intracellular Ca2+ levels within platelets and platelet calpain activity. Inhibition of calpain activity effectively reversed hypoxiainduced platelet hyperreactivity.
Further validation using an in vivo model of hypoxia-induced thrombosis confirmed calpain’s prothrombotic role, with patients developing thrombosis at extreme altitudes showing elevated plasma calpain activities and increased soluble P-selectin levels. This study suggests a close link between heightened calpain activity and increased thrombotic risk under hypoxic conditions. Overactivation of calpain participates in cytoskeletal proteolysis and hyperreactivity of platelets, which creates a prothrombotic state under reactive oxygen-rich hypoxia [64] Studies found that ROS upregulates calpain in smooth muscle cells. The calcium dysregulation triggered by the reactive oxygen species plays a crucial role in upregulating the calpains. Calpain activation leads to the degradation of β2 integrin in endosomes [65].
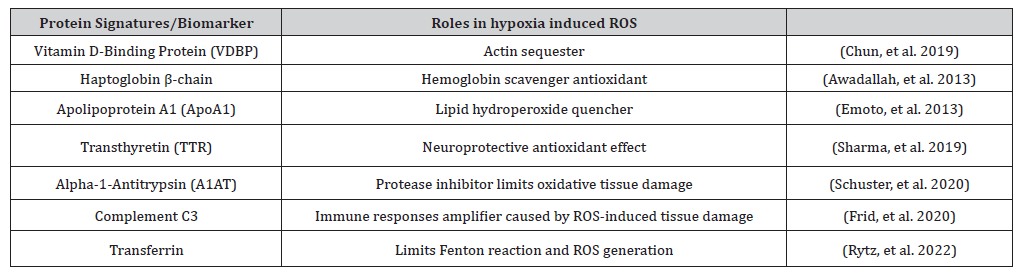
In a study, human subjects exposed to periodic sessions of 12% and 13.5% fractions of oxygen were consequently airlifted to an elevation of 3500m. Plasma proteome analysis was conducted on the 4th and 7th days of high-altitude residency, unveiling oxidative stress and accompanied by perturbed lipid metabolism, post-translational modifications, and inflammatory signaling induced by periodic hypoxic training at baseline. It was concluded that these conditions were found to activate antioxidant mechanisms, maintain bioenergetic stability, and elicit antiinflammatory responses during successive high-altitude exposures [66]. Comparative proteome analysis between the control group and high-altitude residents identified numerous proteins with substantial alterations. Observed upregulated proteins include hemopexin, vitamin D-binding protein, haptoglobin β-chain, apolipoprotein A1, transthyretin, hemoglobin beta chain, and alpha-1-antitrypsin, whereas downregulated proteins encompass complement C3, transferrin, plasma retinol-binding protein, and serum amyloid [67].
Among these upregulated proteins, Hemopexin is a hemebinding protein that functions as an effective antagonist against heme toxicity caused by acute and chronichemolysis [68]. A study found that reactive oxygen species and nitrogen species inactivate the apo-hemopexin [69]. Vitamin D-Binding Protein majorly participates in Vitamin D regulation. It plays a significant role in immune regulation and the antioxidant defense mechanism. It acts as an actin scavenger and eliminates the extracellular actin released due to cell injury. By eliminating actin, VDBP inhibits the pro-thrombotic and inflammatory pathways induced by ROS and actin accumulation [70]. Haptoglobin β-chain mitigates the oxidative damage by impairing heme-driven Fenton chemistry. Studies demonstrated that Haptoglobin counteracts heme toxicity and facilitates oxidative protection during systemic hemolysis [71]. Apolipoprotein A1 (ApoA1) is an HDL protein that facilitates the protection of lipoproteins against oxidative damage [72]. Transthyretin (TTR) shows antioxidant properties by inhibiting protein accumulation under stress. A study illustrated that Transthyretin (TTR) protects neuronal cells against oxidative damage [73]. Alpha-1-antitryFFpsin (A1AT) exhibits both antiinflammatory and antioxidant properties. Janciauskiene, et al. found that A1AT suppresses neutrophil-derived ROS and modulates inflammatory responses [74].
In a quantitative proteomics study, 100% of the subjects died when directly exposed to extreme altitude (24 hours, 3049 m; pO2: 71 kPa, 4573 m; pO2: 59 kPa, and 7620 m; pO2: 40 kPa). However, after pre-exposure to 59 kPa, the subjects’ survival rate significantly increased. Molecular investigations revealed that, via modifying the STAT3-RXR-Nrf2 axis, the pO2 of 59 kPa (extremely highaltitude zone) activated systemic energy and redox homeostatic mechanisms. It was posited that various downstream processes of STAT3-RXR-Nrf2 and plasma proteins could serve as biomarkers for assessing individual redox status [75] (Table 2).
Table 2: Role of protein signatures/biomarkers in response to hypobaric hypoxia-induced ROS generation.
Implications for Tailoring High-altitude specific Antioxidant interventions:
The redox hypothesis postulated that perturbations in thiol redox stimuli lead to oxidative stress [76]. Increased oxidative stress leads to an imbalance between pro-oxidants and antioxidants, resulting in molecular damage and the destruction of redox signaling. This, in turn, precipitates chronic diseases by increasing the production of ROS and RNS. ROS generation in response to environmental hypoxia is determined by the intensity and duration of the hypoxia [4]. The mitochondrial electron transfer chain, xanthine oxidase activity, hemoglobin (HB) autooxidation are the numerous reported sources of hypoxia at high-altitude. Studies conducted on the isolated mitochondria have reported that lower oxygen availability for mitochondrial respiration is responsible for the downregulation of the respiratory chain and enhanced proton leak [77].
Hypobaric hypoxia encountered at high-altitude is due to the lower barometric pressure and reduced partial pressure of oxygen [78; Sharma et al., 2023]. While in pathological conditions like cancer, hypoxia is considered a non-physiological level of oxygen tension. In Tumors, hypoxia, dysfunctional vascularization, and acquisition of epithelial to mesenchymal transition phenotype led to cell mobility and metastasis [Sharma et al., 2023]
Efficacy of Antioxidants in Mitigating the High- Altitude Induced Stress
Numerous studies have demonstrated the effects of antioxidant drugs on stress and maladies caused by high-altitude hypoxia. Following are the numerous examples of antioxidants that research has proven effective in mitigating high-altitude stress induced by hypoxia.
Solnatide and Hypoxic Conditions
A study revealed the potency of Solnatide in alleviating lung fluid accumulation and protein levels in bronchoalveolar fluids in rodents exposed to hypoxic hypobaric conditions and exhaustive exercise. The findings substantiate compelling evidence that the anti-inflammatory and anti-edema characteristics of solnatides make them high-performance drugs in comparison to the currently prescribed drugs, such as aminophylline and dexamethasone. Inhibition of the proinflammatory pathway by solantide decreases the generation of cytokines and deregulates the inflammatory response. Furthermore, studies identified that enhanced expression of a tight junction protein called Occludin due to solantide, facilitates the maintenance of alveolar capillary barriers, which in turn helps to inhibit the leakage of protein in alveolar fluid. These observations indicate the positive therapeutic implications of solantide for treating HAPE in humans [79,80].
Root Extract (RCE) of Rhodiola crenulata
Findings suggest that the root extract of Rhodiola crenulata inhibits biomarkers of oxidative stress, such as malondialdehyde and reactive oxygen species. Studies have shown that it possesses cardioprotective effects orchestrated by phosphorylation of endothelial nitric oxide synthase (eNOS) in cardiomyocytes and by the inhibition of arginase-1 under hypoxic conditions, via the PI3K/AKT signaling pathway [81]. Furthermore, RCE suppresses the overexpression of vascular endothelial growth factor (VEGF) and endothelin-1 (ET-1), thereby preserving the normal function of the alveolar-capillary barrier, which is beneficial for relieving HAPE [82,83].
Salidroside
Salidroside, an active ingredient of Rhodiola rosea, resists oxidation and protects the heart while also inhibiting the production of inflammatory factors and pulmonary vascular remodeling, potentially benefiting high-altitude pulmonary hypertension (HAPH) [84]. In an experiment on rats with pulmonary arterial hypertension under hypoxia, RCE reduced RBC count, hemoglobin, hematocrit, right ventricular hypertrophy index, and mean pulmonary artery pressure, while inhibiting vascular smooth muscle thickening and pulmonary vascular remodeling [85].
Tibetan Turnip (Brassica rapa L.)
Brassica rapa L., commonly known as Tibetan Turnip, is a cruciferous plant species from the genus Brassica. It is specifically used in Tibetan traditional medicine as a natural remedy to alleviate mild symptoms associated with Chronic Mountain Sickness (CMS). The root vegetable has been found to possess anti-hypoxia, antifatigue, analgesic, anti-inflammatory, and neuroprotective properties. The deliquescent broth of turnip root has been found to exhibit strong free radical scavenging properties, and various studies have confirmed that it is composed of phenols and flavonoids, such as kaempferol, quercetin, and isorhamnetin [86,87].
Senecio Nutans (Chachacoma)
Senecio nutans, predominantly found in the Andes region, belongs to the Asteraceae family and is commonly referred to as “Chachacoma.” Its leaves are frequently utilized to alleviate symptoms of Acute Mountain Sickness (AMS), including headaches, dizziness, nausea, and fatigue. Sesquiterpenes, monoterpenes, and p-hydroxyacetophenone are the bioactive parts of S. nutans [88]. The efficacy of S. nutans is comparable to that of nifedipine, as it effectively lowers myocardial oxygen consumption and induces vasodilation [89]. The antifatigue and antioxidant effects of S. Nutans facilitate the sojourners’ acclimatization to elevated regions. Its antioxidant and antifatigue effects can assist sojourners in a relatively easy adaptation to high-altitude conditions. Further research is required to gain a comprehensive understanding of the efficacy and potency of S. nutans in addressing AMS.
Coca Leaf
Coca, falling within the Erythroxylaceae family, is employed in South America for the management of gastrointestinal disorders, cerebral disorders, pharyngeal discomfort, and acute mountain sickness. In Andean communities, the most effective remedy to treat clinical symptoms of acute mountain sickness, headaches, nausea, and dizziness, is steeping Coca leaves in warm water. Cocaine is found to be the principal alkaloid derived from coca leaves, which manifests neuroendocrine effects and stimulates organs such as the heart, lungs, and brain, along with the sympathetic nervous system. Previous studies demonstrate that masticating the coca leaf, which contains 0.5% cocaine, steadily elevates blood levels. Literature highlights that resisting the feedback vicious cycle between the central nervous system and the stomach or intestines by coca leaf helps to mitigate acute mountain sickness. Moreover, evidence points out that the anti-hypoxic effect of coca leaves might reduce the extensive production of RBCs caused by chronic hypoxia exposure, which is required to be proven with further experimental validation. With the expanding demand for natural therapeutic agents exhibiting lower adverse effects, coca leaves can be a beneficial inclusion to current medical care. However, further validations are required for the establishment of treatment [90,91].
Superior Anti-hypoxic Activity of Salvia przewalskii Maxim (Labiatae)
Salvia przewalskii Maxim (Labiatae) (SPM) is a traditional Chinese herbal medicine and has known antibacterial, antiviral, antioxidant, anti-thrombotic, and anti-depressant activities. In vivo, SPM downregulates the proteins, Khk and AldoB, and functions as an antioxidant by suppressing the oxidation-reduction system and fructolysis. Thus, facilitating adaptation to hypoxia. In acute hypoxic environments, SPM extracts have been found to enhance the activities of lactate dehydrogenase and superoxide dismutase.
Additionally, these extracts inhibit the regulation of proliferating cell nuclear antigen, hypoxia-inducible factor-1α, cyclin-dependent kinase 4, Bcl-2, P27Kip1, and CyclinD1, downregulate the monocyte nuclear factor-kappa B and chemoattractant protein-1, and regulate the RhoA-Rho-associated protein kinase signalling pathway, facilitating the restoration of lung injury induced by chronic hypoxia. More clinical evidence is required to validate its anti-hypoxic property [92,93].
Lepidium meyenii (Maca)
Commonly, Lepidium meyenii is referred to as Maca. This Peruvian plant belongs to the Cruciferous family. Its roots, which are comprised of fatty acids and polysaccharides, are commonly indigestible as vegetables. It is extensively deployed to combat inflammation, memory loss, sexual dysfunction, anxiety, and tumors. Choline present in red Maca is found to enhance neurocognitive function. Due to its antioxidant properties, it is implemented as a dietary supplement to mitigate the CMS in high-altitude inhabitants.
Compelling evidence from a study reported that 12-week consumption of spray-processed maca extracts in highlanders enhanced their mood, health, and energy relatively to the placebo group. Remarkably, both red and black Maca were shown to reduce CMS scores at the onset of the 4th week of intervention and the onset of the 8th week, respectively [94-100]. Fascinatingly, a combination of little red maca and black maca declined the blood hemoglobin levels by the 4th week of intervention. Despite this, further investigations are needed to establish the mechanisms and effects of maca for CMS treatment [40].
Conclusion
Conclusively, the findings discussed in this article highlight the possibility of antioxidant supplementation serving as a useful intervention against high-altitude-induced oxidative stress. Further studies should be conducted to enhance these approaches, considering the unpredictable nature of the high elevation environment. By employing improved antioxidant strategies, it is feasible to safeguard the well-being and performance of individuals subjected to the harsh environments of high altitudes.
Declaration of competing interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Acknowledgement
Author contributions: Conceived and designed by YA. The manuscript was written by PS. The manuscript was critically evaluated by YA.
References
- Strapazzon G, Malacrida S, Vezzoli A, Dal Cappello T, Falla M, et al. (2016) Oxidative stress response to acute hypobaric hypoxia and its association with indirect measurement of increased intracranial pressure: a field study. Sci Rep 6: 32426.
- Cassera E, Ferrari E, Vignati DAL, Capucciati A (2025) The interaction between metals and catecholamines: oxidative stress DNA damage and implications for human health. Brain Res Bull 226: 111366.
- Di Meo S, Reed TT, Venditti P, Victor VM (2016) Role of ROS and RNS Sources in Physiological and Pathological Conditions. Oxid Med Cell Longev 1245049.
- Debevec T, Millet GP, Pialoux V (2017) a. Hypoxia-Induced Oxidative Stress Modulation with Physical Activity. Front Physiol 8: 84.
- Vanden Hoek TL, Becker LB, Shao Z, Li C, Schumacker PT (1998) Reactive Oxygen Species Released from Mitochondria during Brief Hypoxia Induce Preconditioning in Cardiomyocytes. J Biol Chem 273(29): 18092-18098.
- Gangwar A, Paul S, Arya A, Ahmad Y, Bhargava K (2022) Altitude acclimatization via hypoxia-mediated oxidative eustress involves interplay of protein nitrosylation and carbonylation: A redoxomics perspective. Life Sci 296: 120021.
- Mrakic-Sposta S, Biagini D, Bondi D, Pietrangelo T, Vezzoli A (2022) OxInflammation at High-altitudes: A Proof of Concept from the Himalayas. Antioxidants 11(2): 368.
- Ali M, Choudhary R, Rabyang S, Thinlas T, Mishra A (2023) Harsh environmental stressors of high-altitude on pathogens susceptibility in: Genomic Surveillance and Pandemic Preparedness. Elsevier pp. 357-373.
- Ch MR, Castillo A, Huicho L (2008) Hypoxia and other environmental factors at high-altitude. International Journal of Environment and Health 2(1): 92.
- Gallagher SA, Hackett PH (2004) High-altitude illness. Emerg Med Clin North Am 22: 329-355.
- Savioli G, Ceresa IF, Gori G, Fumoso F, Gri N, et al. (2022) Pathophysiology and Therapy of High-Altitude Sickness: Practical Approach in Emergency and Critical Care. J Clin Med 11(14): 3937.
- Cobb AB, Levett DZH, Mitchell Kay, Aveling W, Hurlbut Daniel (2021) Physiological responses during ascent to high-altitude and the incidence of acute mountain sickness. Physiol Rep 9(7): e14809.
- Hackett PH, Roach RC (2004) High-altitude Cerebral Edema. High Alt Med Biol 5: 136-146.
- Wu T, Ding S, Liu J, Jia J, Dai, R et al. (2006) Ataxia: An Early Indicator in High-altitude Cerebral Edema. High Alt Med Biol 7(4): 275-280.
- Hüfner K, Falla M, Brugger H, Gatterer H, Strapazzon G, et al. (2023) Isolated high-altitude psychosis, delirium at high-altitude, and high-altitude cerebral edema: are these diagnoses valid? Front Psychiatry 14: 1221047.
- Hüfner K, Brugger H, Kuster E, Dünsser F, Stawinoga AE, et al. (2018) Isolated psychosis during exposure to very high and extreme altitude – characterisation of a new medical entity. Psychol Med 48(11): 1872-1879.
- Kronenberg RS, Safar P, Lee J, Wright F, Noble W, et al. (1971) Pulmonary Artery Pressure and Alveolar Gas Exchange in Man during Acclimatization to 12,470 ft. J Clin Invest 50(4): 827-837.
- Singh I, Khanna PK, Srivastava MC, Lal M, Roy SB, et al. (1969) Acute Mountain Sickness. New England Journal of Medicine 280: 175-184.
- Dickinson J, Heath D, Gosney J, Williams D (1983) Altitude-related deaths in seven trekkers in the Himalayas. Thorax 38(9): 646-656.
- Houston, CharlesS, Dickinson J (1975) CEREBRAL FORM OF HIGH-ALTITUDE ILLNESS. Lancet 2(7938): 758-761.
- Gabry A.L, Ledoux X, Mozziconacci M, Martin C (2003) High-Altitude Pulmonary Edema at Moderate Altitude 2,400 m; 7,870 feet). Chest 123(1): 49-53.
- Hackett PH, Yarnell PR, Hill R, Reynard K, Heit J, et al. (1998) High-Altitude Cerebral Edema Evaluated With Magnetic Resonance Imaging. JAMA 280(22): 1920-1925.
- Yarnell PR, Heit J, Hackett PH (2000) High-Altitude Cerebral Edema (HACE): The Denver/ Front Range Experience. Semin Neurol 20(2): 209-218.
- Turner REF, Gatterer H, Falla M, Lawley JS (2021) High-altitude cerebral edema: its own entity or end-stage acute mountain sickness? J Appl Physiol 131(1): 313-325.
- Poh Agin P (2017) Measuring ultraviolet A protection in sunscreen products. J Am Acad Dermatol 77(3): e81.
- Gorman S, de Courten B, Lucas R (2019) Systematic Review of the Effects of Ultraviolet Radiation on Markers of Metabolic Dysfunction. Clin Biochem Rev 40(3): 147-162.
- Tang X, Yang T, Yu D, Xiong H, Zhang S (2024) Current insights and future perspectives of ultraviolet radiation (UV) exposure: Friends and foes to the skin and beyond the skin. Environ Int 185: 108535.
- Schmucki DA, Philipona R (2002) UV radiation in the Alps: the altitude effect. in: Slusser JR, Herman JR, Gao W, (Eds.). pp. 234-239.
- Izadi M, Jonaidi-Jafari N, Pourazizi M, Alemzadeh-Ansari M, Hoseinpourfard M (2018) Photokeratitis induced by ultraviolet radiation in travelers. J Postgrad Med 64(1): 40-46.
- Singh G (2017) High-altitude dermatology. Indian J Dermatol 62(1): 59.
- Nishigori C, Hattori Y, Toyokuni S (2004) Role of Reactive Oxygen Species in Skin Carcinogenesis. Antioxid Redox Signal 6(3): 561-570.
- Nishigori C, Yarosh DB, Ullrich SE, Vink AA, Bucana CD, et al. (1996) Evidence that DNA damage triggers interleukin 10 cytokine production in UV-irradiated murine keratinocytes. Proc Natl Acad Sci 93(19): 10354-10359.
- Wei M, He X, Liu N, Deng H (2024) Role of reactive oxygen species in ultraviolet-induced photodamage of the skin. Cell Div 19(1): 1.
- Chen HI, Hu WS, Hung MY, Ou HC, Huang SH, et al. (2020) Protective effects of luteolin against oxidative stress and mitochondrial dysfunction in endothelial cells. Nutr Metab Cardiovasc Dis 30(6): 1032-1043.
- Baier J, Maisch T, Maier M, Engel E, Landthaler M, Bäumler W (2006) Singlet Oxygen Generation by UVA Light Exposure of Endogenous Photosensitizers. Biophys J 91(4): 1452-1459.
- Yadav RB, Yadav AK, Pathak DP, Arora R (2024) Cold Injury Prevention and Management in High-altitude Extreme Environments Pharmacological and Therapeutical Interventions. Def Life Sci J 9: 106-113.
- Hallam MJ, Cubison T, Dheansa B, Imray C (2010) Managing frostbite. BMJ 341: c5864-c5864.
- Harirchi I, Arvin A, Vash JH, Zafarmand V (2005) Frostbite: incidence and predisposing factors in mountaineers. Br J Sports Med 39(12): 898-901.
- Gaur P, Prasad S, Kumar B, Sharma SK, Vats P (2021) High-altitude hypoxia induced reactive oxygen species generation, signaling, and mitigation approaches. Int J Biometeorol 65(4): 601-615.
- Lobo V, Patil A, Phatak A, Chandra N (2010) Free radicals, antioxidants, and functional foods: Impact on human health. Pharmacogn Rev 4(8): 118-126.
- Bruick R.K, McKnight S.L (2001) A Conserved Family of Prolyl-4-Hydroxylases That Modify HIF. Science 294(5545): 1337-1340.
- Bae T, Hallis SP, Kwak MK (2024) Hypoxia, oxidative stress, and the interplay of HIFs and NRF2 signaling in cancer. Exp Mol Med 56: 501-514.
- Lee FS (2024) Hypoxia Inducible Factor pathway proteins in high-altitude mammals. Trends Biochem Sci 49(1): 79-92.
- Olson N, van der Vliet A (2011) Interactions between nitric oxide and hypoxia-inducible factor signaling pathways in inflammatory disease. Nitric Oxide 25(2): 125-137.
- Xiao D, Bird IM, Magness RR, Longo LD, Zhang L (2001) Upregulation of eNOS in pregnant ovine uterine arteries by chronic hypoxia. Am J Physiol Heart Circ Physiol 280(2): H812-H820.
- Janaszak-Jasiecka A, Siekierzycka A, Bartoszewska S, Serocki M, Dobrucki LW, et al. (2018) eNOS expression and NO release during hypoxia is inhibited by miR-200b in human endothelial cells. Angiogenesis 21(4): 711-724.
- Ting KKY (2024) Revisiting the role of hypoxia-inducible factors and nuclear factor erythroid 2-related factor 2 in regulating macrophage inflammation and metabolism. Front Cell Infect Microbiol 14: 1403915.
- Bakonyi T, Radak Z (2004) High-altitude and free radicals. J Sports Sci Med 3(2): 64-69.
- Radk Z, Lee K, Choi W, Sunoo S, Kizaki T (1994) Oxidative stress induced by intermittent exposure at a simulated altitude of 4000 m decreases mitochondrial superoxide dismutase content in soleus muscle of rats. Eur J Appl Physiol Occup Physiol 69(5): 392-395.
- Imai H, Kashiwazaki H, Suzuki T, Kaeuto M, Himeno S (1995) Selenium Levels and Glutathione Peroxidase Activities in Blood in an Andean High-Altitude Population. J Nutr Sci Vitaminol (Tokyo) 41(3): 349-361.
- Ilavazhagan G, Bansal A, Prasad D, Thomas P, Sharma SK (2001) Effect of vitamin E supplementation on hypoxia-induced oxidative damage in male albino rats. Aviat Space Environ Med 72(10): 899-903.
- Schmidt MC, Askew EW, Roberts DE, Prior RL, Ensign WY et al. (2002) Oxidative stress in humans training in a cold, moderate altitude environment and their response to a phytochemical antioxidant supplement. Wilderness Environ Med 13(2): 94-105.
- Kaur C, Srinivasan KN, Singh J, Peng CM, Ling EA (2002) Plasma melatonin, pinealocyte morphology, and surface receptors/antigen expression on macrophages/microglia in the pineal gland following a high‐altitude exposure. J Neurosci Res 67(4): 533-543.
- Halliwell B (1995) How to characterize an antioxidant: an update. Biochem Soc Symp 61: 73-101.
- Shi H, Noguchi N, Niki E (1999) Comparative study on dynamics of antioxidative action of α-tocopheryl hydroquinone, ubiquinol, and α-tocopherol against lipid peroxidation. Free Radic Biol Med 27(3-4): 334-346.
- Levine M (1999) Criteria and Recommendations for Vitamin C Intake. JAMA 281(15): 1415-1423.
- Imai H, Nakagawa Y (2003) Biological significance of phospholipid hydroperoxide glutathione peroxidase (PHGPx, GPx4) in mammalian cells. Free Radic Biol Med 34(2): 145-169.
- Niki E (1991) Action of ascorbic acid as a scavenger of active and stable oxygen radicals. Am J Clin Nutr 54(6): 1119S-1124S.
- Ames BN, Cathcart R, Schwiers E, Hochstein P (1981) Uric acid provides an antioxidant defense in humans against oxidant- and radical-caused aging and cancer: Proc Natl Acad Sci 78: 6858-6862.
- Stocker R, Yamamoto Y, McDonagh AF, Glazer AN, Ames BN (1987) Bilirubin Is an Antioxidant of Possible Physiological Importance. Science 235(4792): 1043-1046.
- Roche M, Rondeau P, Singh NR, Tarnus E, Bourdon E (2008) The antioxidant properties of serum albumin. FEBS Lett 582(13): 1783-1787.
- Ernster L, Forsmark-Andre P (1993) Ubiquinol: an endogenous antioxidant in aerobic organisms. Clin Investig 71(8): 60-65.
- Ahmad Y, Sharma NK, Ahmad MF, Sharma M, Garg I, et al. (2015) The proteome of Hypobaric Induced Hypoxic Lung: Insights from Temporal Proteomic Profiling for Biomarker Discovery. Sci Rep 5: 10681.
- Tyagi T, Ahmad S, Gupta N, Sahu A, Ahmad Y, et al. (2014) Altered expression of platelet proteins and calpain activity mediate hypoxia-induced prothrombotic phenotype. Blood 123(8): 1250-1260.
- Samanta K, Ahel I, Kar P (2025) Deciphering of the reactive oxygen species (ROS) induced calpain activation in cancer progression and its therapeutic potential. Advances in Redox Research 15: 100124.
- Gangwar A, Paul S, Ahmad Y, Bhargava K (2020) Intermittent hypoxia modulates redox homeostasis, lipid metabolism associated inflammatory processes and redox post-translational modifications: Benefits at high-altitude. Sci Rep 10(1): 7899.
- Ahmad Y, Sharma NK, Garg I, Ahmad MF, Sharma M, Bhargava K, et al.(2013) An Insight into the Changes in Human Plasma Proteome on Adaptation to Hypobaric Hypoxia. PLoS One 8(7): e67548.
- Li Y, Chen R, Wang C, Deng J, Luo S (2023) Double-edged functions of hemopexin in hematological-related diseases: from basic mechanisms to clinical application. Front Immunol 14: 1274333.
- Hahl P, Hunt R, Bjes ES, Skaff A, Keightley A, et al. (2017) Identification of oxidative modifications of hemopexin and their predicted physiological relevance. J Biol Chem 292(33): 13658-13671.
- Chun RF, Shieh A, Gottlieb C, Yacoubian V, Wang J (2019) a. Vitamin D Binding Protein and the Biological Activity of Vitamin D. Front Endocrinol (Lausanne) 10: 718.
- Awadallah SM, Ramadan AR, Nusier MK (2013) Haptoglobin polymorphism in relation to antioxidative enzymes activity in type 2 diabetes mellitus. Diabetes Metab Syndr 7(1): 26-31.
- Emoto T, Sawada T, Morimoto N, Tenjin T, Wakimoto T (2013) The Apolipoprotein B/A1 Ratio is Associated with Reactive Oxygen Metabolites and Endothelial Dysfunction in Statin-Treated Patients with Coronary Artery Disease. J Atheroscler Thromb 20(7): 623-629.
- Sharma M, Khan S, Rahman S, Sing LR (2019) The Extracellular Protein, Transthyretin Is an Oxidative Stress Biomarker. Front Physiol 10: 5.
- Schuster R, Motola-Kalay N, Baranovski BM, Bar L, Tov N (2020) Distinct anti-inflammatory properties of alpha1-antitrypsin and corticosteroids reveal unique underlying mechanisms of action. Cell Immunol 356: 104177.
- Paul S, Gangwar A, Bhargava K, Ahmad Y (2018) STAT3-RXR-Nrf2 activates systemic redox and energy homeostasis upon steep decline in pO2 gradient. Redox Biol 14: 423-438.
- Checa J, Aran JM (2020) Reactive Oxygen Species: Drivers of Physiological and Pathological Processes. J Inflamm Res 13: 1057-1073.
- Michiels C (2004) Physiological and Pathological Responses to Hypoxia. Am J Pathol 164(6): 1875-1882.
- Sharma P, Mohanty S, Ahmad Y (2025) Decoding Proteomic cross-talk between hypobaric and normobaric hypoxia: Integrative analysis of oxidative stress, cytoskeleton remodeling, and inflammatory pathways. Life Sci 371: 123611.
- Zhou Q, Wang D, Liu Y, Yang X, Lucas R et al. (2017) Solnatide Demonstrates Profound Therapeutic Activity in a Rat Model of Pulmonary Edema Induced by Acute Hypobaric Hypoxia and Exercise. Chest 151(3): 658-667.
- Schmid B, Kredel M, Ullrich R, Krenn K, Lucas R et al. (2021) Safety and preliminary efficacy of sequential multiple ascending doses of solnatide to treat pulmonary permeability edema in patients with moderate-to-severe ARDS—a randomized, placebo-controlled, double-blind trial. Trials 22(1): 643.
- Hsu SW, Chang TC, Wu YK, Lin KT, Shi LS, et al. (2017) Rhodiola crenulata extract counteracts the effect of hypobaric hypoxia in rat heart via redirection of the nitric oxide and arginase 1 pathway. BMC Complement Altern Med 17(1): 29.
- Lafuente J, Bermudez G, Camargo-Arce L, Bulnes S (2016) Blood-Brain Barrier Changes in High-altitude. CNS Neurol Disord Drug Targets 15(9): 1188-1197.
- Lee SY, Li MH, Shi LS, Chu H, Ho CW, et al. (2013) Rhodiola crenulata Extract Alleviates Hypoxic Pulmonary Edema in Rats. Evid Based Complement Alternat Med 1-9.
- Kosanovic D, Tian X, Pak O, Lai Y, Hsieh Y, et al. (2013) Rhodiola : An Ordinary Plant or a Promising Future Therapy for Pulmonary Hypertension? A Brief Review. Pulm Circ 3(3): 499-506.
- Nan X, Su S, Ma K, Ma X, Wang X, et al. (2018) Bioactive fraction of Rhodiola algida against chronic hypoxia-induced pulmonary arterial hypertension and its anti-proliferation mechanism in rats. J Ethnopharmacol 216: 175-183.
- Chu B, Chen C, Li J, Chen X, Li Y (2017) Effects of Tibetan turnip (Brassica rapa L.) on promoting hypoxia-tolerance in healthy humans. J Ethnopharmacol 195: 246-254.
- Paul S, Geng C, Yang T, Yang Y, Chen J (2019) Phytochemical and Health‐Beneficial Progress of Turnip (Brassica rapa). J Food Sci 84(1): 19-30.
- Paredes A, Leyton Y, Riquelme C, Morales G (2016) A plant from the altiplano of Northern Chile Senecio nutans, inhibits the Vibrio cholerae pathogen. Springerplus 5(1): 1788.
- Cifuentes F, Paredes A, Palacios J, Muñoz F, Carvajal L (2016) Hypotensive and antihypertensive effects of a hydroalcoholic extract from Senecio nutans Sch. Bip. (Compositae) in mice: Chronotropic and negative inotropic effect, a nifedipine-like action. J Ethnopharmacol 179: 367-374.
- Biondich AS, Joslin JD (2016) Coca: The History and Medical Significance of an Ancient Andean Tradition. Emerg Med Int 1-5.
- Manetti L, Cavagnini F, Martino E, Ambrogio A (2014) Effects of cocaine on the hypothalamic–pituitary–adrenal axis. J Endocrinol Invest 37(8): 701-708.
- Skała E, Wysokińsk H (2005) Tanshinone Production in Roots of Micropropagated Salvia przewalskii Maxim. Z Naturforsch C J Biosci 60(7-8): 583-586.
- Wang Y, Duo D, Yan Y, He R, Wang S, et al. (2020) Bioactive constituents of Salvia przewalskii and the molecular mechanism of its antihypoxia effects determined using quantitative proteomics. Pharm Biol 58(1): 469-477.
- Li L, Lin L, Wen B, Zhao P, Liu D, et al. (2023) Promising Natural Medicines for the Treatment of High-Altitude Illness. High Alt Med Biol 24(3): 175-185.
- Chun R.F, Shieh A, Gottlieb C, Yacoubian V, Wang J, Hewison M (2019) b. Vitamin D Binding Protein and the Biological Activity of Vitamin D. Front Endocrinol (Lausanne) 10: 718.
- Dale Wilson B, Moon S, Armstrong F (2012) Comprehensive review of ultraviolet radiation and the current status on sunscreens. J Clin Aesthet Dermatol 5(9): 18-23.
- Frid M.G, McKeon B.A, Thurman J.M, Maron B.A, Li M (2020) Immunoglobulin-driven Complement Activation Regulates Proinflammatory Remodeling in Pulmonary Hypertension. Am J Respir Crit Care Med 201(2): 224-239.
- Mallet R.T, Burtscher J, Pialoux V, Pasha Q, Ahmad Y et al. (2023) Molecular Mechanisms of High-Altitude Acclimatization. Int J Mol Sci 24(2): 1698.
- Reguera S, Zamora-Camacho F.J, Melero E, García-Mesa S, Trenzado C.E (2015) Ultraviolet radiation does not increase oxidative stress in the lizard Psammodromus algirus along an elevational gradient. Comp Biochem Physiol a Mol Integr Physiol 183: 20-26.
- Rytz C.L, Pun M, Mawhinney J.A, Mounsey C.A, Mura M et al. (2022) Differential effects of high-altitude exposure on markers of oxidative stress, antioxidant capacity, and iron profiles. Am J Physiol Regul Integr Comp Physiol 323(4): R445-R456.
-
Poornima Sharma and Yasmin Ahmad*. Catalytic Role of Antioxidants in Mitigating the Physiological Consequences of High- Altitude-Associated Environmental Stressors. Arch Biomed Eng & Biotechnol. 6(3): 2025. ABEB.MS.ID.000687.
-
High-altitude; Environmental Stressors; Hypoxia; ROS; Anti-Oxidant Supplementation
-

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.
- Abstract
- Introduction
- High-altitude Environment Stressors
- Oxidative strain and Elevation exposure
- Free Radical Generation at High-altitude
- ROS-Associated Signaling Pathways in Response to Hypobaric Hypoxia
- Impact of high-altitude on antioxidant systems
- Antioxidants and their protective role
- Levels of Antioxidant Action
- Reactive Oxygen Species (ROS) Generation and Cellular ROS-Associated Proteome Disruptions Driven by High-Altitude Hypobaric Hypoxia
- Implications for Tailoring High-altitude specific Antioxidant interventions:
- Conclusion
- Declaration of competing interest
- Acknowledgement
- References






